Search found 8 matches
- Sun Jul 09, 2017 11:30 pm
- Forum: Trends in The Periodic Table
- Topic: Electron Affinity of Beryllium & Magnesium
- Replies: 1
- Views: 1912
Electron Affinity of Beryllium & Magnesium
Why are the electron affinity values for beryllium (Be) and magnesium (Mg)  0?
0?
- Sun Jul 09, 2017 11:05 pm
- Forum: Electron Configurations for Multi-Electron Atoms
- Topic: Exercise 2.51
- Replies: 1
- Views: 550
Re: Exercise 2.51
I had a bit of trouble figuring out the electron configuration for tantalum (Ta), so I had to look it up and it is [Xe] 4f145d36s2
The d-subshell isn't filled entirely, and there's an odd number of electrons present (3). That is why 3 unpaired electrons are predicted. Hope this helps
The d-subshell isn't filled entirely, and there's an odd number of electrons present (3). That is why 3 unpaired electrons are predicted. Hope this helps
- Sun Jul 09, 2017 10:32 pm
- Forum: Trends in The Periodic Table
- Topic: Electron Affinity Trend
- Replies: 3
- Views: 710
Re: Electron Affinity Trend
504754253 wrote:more gaseous elements are on the right side of the periodic table and they are more reactive. Hope this helps!
If they are more reactive, does this mean they release more or less energy when an electron is added?
- Sun Jul 09, 2017 10:13 pm
- Forum: Trends in The Periodic Table
- Topic: Electron Affinity Trend
- Replies: 3
- Views: 710
Electron Affinity Trend
In the course reader it says there is “generally high electron affinity for elements in the top right of the periodic table.” What does this mean?
- Sun Jul 02, 2017 11:20 pm
- Forum: Bohr Frequency Condition, H-Atom , Atomic Spectroscopy
- Topic: Atomic Spectra & Energy Levels
- Replies: 2
- Views: 621
Atomic Spectra & Energy Levels
Can someone please explain the following:
As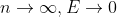
This was noted in class on Friday (6/30) with regard to changes in energy levels, and some clarification would be greatly appreciated!
As
This was noted in class on Friday (6/30) with regard to changes in energy levels, and some clarification would be greatly appreciated!
- Sun Jul 02, 2017 10:55 pm
- Forum: Properties of Electrons
- Topic: Wave Properties of Electrons
- Replies: 1
- Views: 391
Wave Properties of Electrons
I remember Dr. Lavelle going over the Dual Slit Experiment during lecture on Friday (6/30).
Will we ever be asked on a test to explain/describe the details of that experiment, or is it more important to remember the results and their significance?
Will we ever be asked on a test to explain/describe the details of that experiment, or is it more important to remember the results and their significance?
- Sun Jul 02, 2017 10:33 pm
- Forum: Limiting Reactant Calculations
- Topic: Reactant vs. Reagent [ENDORSED]
- Replies: 23
- Views: 34888
Reactant vs. Reagent [ENDORSED]
I’ve noticed both of these terms, on separate occasions, used in limiting reactant problems.
Are they used interchangeably, or is there an important distinction between the two that I should know?
Are they used interchangeably, or is there an important distinction between the two that I should know?
- Fri Jun 30, 2017 9:30 pm
- Forum: Empirical & Molecular Formulas
- Topic: Whole Number Coefficients
- Replies: 5
- Views: 1478
Whole Number Coefficients
When writing empirical formulas, is there a trick to knowing what to multiply your decimals by in order to get whole numbers, or do I have to guess and check? For example, I got coefficients of 1, 1, and 2.381 in a problem I was working through. What should I multiply 2.381 by in order to get a whol...

