Search found 62 matches
- Mon Mar 11, 2019 10:09 pm
- Forum: General Rate Laws
- Topic: Homogeneous vs Heterogeneous Reactions
- Replies: 2
- Views: 382
Re: Homogeneous vs Heterogeneous Reactions
"Homogeneous reactions are chemical reactions in which the reactants and products are in the same phase, while heterogeneous reactions have reactants in two or more phases. Reactions that take place on the surface of a catalyst of a different phase are also heterogeneous. A reaction between two...
- Mon Mar 11, 2019 10:00 pm
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: Test 2: Gibb's Free Energy Question
- Replies: 1
- Views: 346
Re: Test 2: Gibb's Free Energy Question
I don't know the answer and am confused on those as well so I'm going to go to the review of Test 2 in Haines 39 at 11 am. I'm sure the TA will go over the answers and explain it there.
- Mon Mar 11, 2019 9:58 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Pre-Equilibrium Steps
- Replies: 2
- Views: 302
Pre-Equilibrium Steps
Are we expected to know whether a reaction is fast or slow in order to use the pre-equilibrium approach? For example, in lecture today for the pre-equilibrium approach, step 1 was a fast bimolecular dimerization and step 2 was a slow bimolecular reaction.
- Mon Mar 11, 2019 9:51 pm
- Forum: General Rate Laws
- Topic: Avg Rate vs Instantaneous Rate
- Replies: 2
- Views: 335
Re: Avg Rate vs Instantaneous Rate
Average rate is an approximation and therefore isn't very accurate. I think most problems will want us to find instantaneous rates because they are more accurate
- Mon Mar 11, 2019 9:48 pm
- Forum: General Rate Laws
- Topic: unique rate [ENDORSED]
- Replies: 3
- Views: 680
Re: unique rate [ENDORSED]
Unique rate of reaction looks like -\frac{1}{C}*\frac{\Delta \left \lfloor R1 \right \rfloor}{\Delta t} and instantaneous reaction has the form \lim_{x\rightarrow 0} \frac{\Delta \left [ concentration \right ]}{dt} or \frac{d\left [ concentration \right ]}{dt} the instantaneous rate is more accurate.
- Mon Mar 11, 2019 9:37 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Pre-Equilibrium Approach
- Replies: 1
- Views: 255
Pre-Equilibrium Approach
How will we know when to use the pre-equilibrium approach? Will we always use this approach instead of the other two ways?
- Mon Mar 11, 2019 9:29 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Final Exam Topics
- Replies: 8
- Views: 838
Re: Final Exam Topics
The final will include all material from the entire quarter including what he goes over on Wednesday. Friday is review of past exam questions.
- Mon Feb 25, 2019 9:31 pm
- Forum: Work, Gibbs Free Energy, Cell (Redox) Potentials
- Topic: Pt inert electrode
- Replies: 9
- Views: 1823
Re: Pt inert electrode
In the solutions manual for the 6th edition 14.13, it states that "Pt is necessary when both oxidized and reduced species are in the same solution" and that it is necessary for "gas/ion electrode reactions"
- Mon Feb 25, 2019 9:24 pm
- Forum: Student Social/Study Group
- Topic: Fridays lecture notes
- Replies: 1
- Views: 550
Fridays lecture notes
Hi, I had to miss a couple of Friday lectures :( Does anyone have Friday week 6 and week 7 lecture notes they would be willing to share?
Thank you!
Thank you!
- Mon Feb 25, 2019 9:20 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Reducing agent/oxidizing agent
- Replies: 5
- Views: 562
Re: Reducing agent/oxidizing agent
A reducing agent is a substance that is oxidized, which means it loses electrons.
- Mon Feb 25, 2019 9:19 pm
- Forum: Balancing Redox Reactions
- Topic: about redox reaction
- Replies: 5
- Views: 794
Re: about redox reaction
Since a redox reaction is a reduction/oxidation reaction, I thought oxygen had to be present but when I searched it up it says that "Oxygen does not have to be present in a reaction for it to be a redox-reaction. Oxidation is the loss of electrons." https://simple.wikipedia.org/wiki/Oxidat...
- Mon Feb 25, 2019 9:13 pm
- Forum: Appications of the Nernst Equation (e.g., Concentration Cells, Non-Standard Cell Potentials, Calculating Equilibrium Constants and pH)
- Topic: Nernst equation with K and Q
- Replies: 2
- Views: 396
Re: Nernst equation with K and Q
When you change the log of Q to the log of K, you know that the reaction has reached equilibrium. I also am not sure how we determine which way the reaction will proceed.
- Mon Feb 25, 2019 9:08 pm
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: Value of K in Gibbs free energy equation
- Replies: 3
- Views: 706
Re: Value of K in Gibbs free energy equation
When a chemical reaction is at equilibrium, the Gibbs free energy is, indeed, equal to zero, but that doesn't mean that the Gibbs free energy under standard conditions is (the little circle to the top right is what you're most likely missing)... Can you explain more about what the standard conditio...
- Mon Feb 25, 2019 9:05 pm
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: Gibbs free energy at equilibrium
- Replies: 2
- Views: 276
Re: Gibbs free energy at equilibrium
When a reaction is at equilibrium, no more work can be done and the Gibbs free energy or delta G is zero.
- Mon Feb 25, 2019 8:57 pm
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: Vant Hoff Equation
- Replies: 3
- Views: 392
Re: Vant Hoff Equation
When do we know when to use the Vant Hoff equation?
- Mon Feb 25, 2019 8:55 pm
- Forum: Work, Gibbs Free Energy, Cell (Redox) Potentials
- Topic: Gibbs At Equilibrium
- Replies: 3
- Views: 538
Re: Gibbs At Equilibrium
When the reaction is at equilibrium the Gibbs Free energy will be zero
- Mon Feb 25, 2019 8:53 pm
- Forum: Balancing Redox Reactions
- Topic: Test #2
- Replies: 9
- Views: 938
Re: Test #2
SydBenedict2H wrote:Will he ask us any acid base questions, or is he only asking Gibbs and electrochemistry?
I don't think we'll be tested on acid base questions since it was on the midterm already. It'll just be Gibbs free energy and up to what we learned in class today (but not including it)
- Mon Feb 25, 2019 8:48 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Cell Diagrams Example 14.4 in 6th Edition Book
- Replies: 2
- Views: 825
Re: Cell Diagrams Example 14.4 in 6th Edition Book
Pt(s) | H2(g) | HCl(aq) | Hg2Cl2(s) | Hg(l)
From Step 1:
Cl-|Hg2Cl2|Hg
From Step 2:
Pt|H2|H+
To me, it looks like they divide the cell Pt(s) | H2(g) | HCl(aq) | Hg2Cl2(s) | Hg(l) in the middle and separate HCl into H+ and Cl-
From Step 1:
Cl-|Hg2Cl2|Hg
From Step 2:
Pt|H2|H+
To me, it looks like they divide the cell Pt(s) | H2(g) | HCl(aq) | Hg2Cl2(s) | Hg(l) in the middle and separate HCl into H+ and Cl-
- Mon Feb 25, 2019 8:44 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Cell Diagrams Example 14.4 in 6th Edition Book
- Replies: 2
- Views: 825
Re: Cell Diagrams Example 14.4 in 6th Edition Book
I'm confused about this diagram as well, but maybe it is just assumed Cl- will be a separate component?
- Tue Feb 12, 2019 1:36 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Saying Thank You to Dr. Lavelle
- Replies: 490
- Views: 633189
Re: Saying Thank You to Dr. Lavelle
Valentine's Song for Dr. Lavelle - 2/11/2019
- Wed Jan 30, 2019 4:16 pm
- Forum: Phase Changes & Related Calculations
- Topic: Heat Capacity
- Replies: 10
- Views: 967
Re: Heat Capacity
The variable that represents heat capacity is I think just "C", please correct me if I'm wrong.
- Wed Jan 30, 2019 4:11 pm
- Forum: Phase Changes & Related Calculations
- Topic: Phase changes
- Replies: 14
- Views: 1444
Re: Phase changes
Why is it that the phase change for water from a liquid to a gas than from a solid to a liquid has more energy? I think when water changes from a liquid to a gas there is more energy because the particles need to be moving faster as a gas and the all bonds in the liquid need to be broken for it to ...
- Wed Jan 30, 2019 4:03 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: Isolated systems
- Replies: 10
- Views: 823
Re: Isolated systems
How could the volume change in an isolated system? Would the system just expand on its own?
- Wed Jan 30, 2019 3:47 pm
- Forum: Calculating Work of Expansion
- Topic: Units for Work
- Replies: 6
- Views: 717
Re: Units for Work
Rachel-Weisz3C wrote:I think for pressure we use atm and for volume it should be Liters/milliliters, depending on the problem provides.
Is the SI unit for pressure Pascal? Should we be using the SI unit or just atm?
- Wed Jan 16, 2019 6:29 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: 11.57 6th edition
- Replies: 1
- Views: 209
Re: 11.57 6th edition
Did you divide the concentrations by the volume of 10 L? Also, there is a coefficient of 3 for H2, so you would need to raise the concentration of H2 to the 3rd power.
- Wed Jan 16, 2019 5:45 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Ice table coefficients [ENDORSED]
- Replies: 6
- Views: 966
Re: Ice table coefficients [ENDORSED]
I'm not sure if I'm correct but to get the change row in the ICE table, isn't it always going to be either -x or +x? I haven't encountered a problem where there are coefficients in front of it. But I am also confused on this topic.
- Wed Jan 16, 2019 5:14 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Solids and liquids in K
- Replies: 6
- Views: 2194
Re: Solids and liquids in K
You don't include liquids in your equilibrium expressions because they occur in such large amounts that they are considered the solvents of the chemical reaction. Any reaction that occurs using a liquid will not provide a large enough change to that liquids concentration so as to require us to incl...
- Wed Jan 16, 2019 5:10 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Acids
- Replies: 6
- Views: 523
Re: Acids
A weak acid does not fully dissociate in water and therefore, would require an equilibrium equation. A strong acid fully dissociates in water and so it would be written as just a forward reaction. You just have to memorize the strong acids and every other acid is a weak acid. The strong acids are H...
- Wed Jan 16, 2019 5:01 pm
- Forum: Non-Equilibrium Conditions & The Reaction Quotient
- Topic: Percentage Ionization
- Replies: 4
- Views: 408
Re: Percentage Ionization
Also, what does it mean to be "completely ionized"?
In class Dr. Lavelle stated that "Ba(OH)2 (aq) is a strong base, essentially completely ionized."
In class Dr. Lavelle stated that "Ba(OH)2 (aq) is a strong base, essentially completely ionized."
- Wed Jan 16, 2019 4:56 pm
- Forum: Non-Equilibrium Conditions & The Reaction Quotient
- Topic: Percentage Ionization
- Replies: 4
- Views: 408
Percentage Ionization
Can someone explain to me what percentage ionization means? In the example Dr. Lavelle did in class today, he asked what is the pH and percentage ionization of acetic acid in 0.10 M Ch3COOH?
- Wed Jan 16, 2019 4:51 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Taking x away in a Ka calculation
- Replies: 5
- Views: 442
Re: Taking x away in a Ka calculation
In the example Dr. Lavelle did in class today with kA to find the pH, can someone help explain to me how he was able to approximate it to 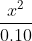 ?
?
- Wed Jan 16, 2019 4:46 pm
- Forum: Non-Equilibrium Conditions & The Reaction Quotient
- Topic: Weak Acids and Bases
- Replies: 6
- Views: 584
Re: Weak Acids and Bases
How can you tell if you have a weak/strong acid or base?
- Sun Jun 10, 2018 1:01 am
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: Porphyrin Ligand
- Replies: 4
- Views: 689
Re: Porphyrin Ligand
On the final, are we supposed to memorize/be able to recognize all of the ligands?
- Sun Jun 10, 2018 12:59 am
- Forum: Conjugate Acids & Bases
- Topic: Conjugate Acids/Bases
- Replies: 8
- Views: 1253
Re: Conjugate Acids/Bases
I believe it is the opposite on the other side of the reaction. Attached is a picture of my worksheet from discussion. So from the image, I think HCl is a Bronsted acid and its conjugate base is Cl - . H 2 O is a Bronsted Base in this reaction and its conjugate acid is H 3 O + I'm not sure if I am e...
- Sun Jun 10, 2018 12:36 am
- Forum: Accuracy, Precision, Mole, Other Definitions
- Topic: Molar Mass
- Replies: 5
- Views: 861
Re: Molar Mass
When finding the molar mass of Fe2, you would look on the periodic table to see the molar mass of just Fe which is 55.845, and multiply it by 2 to get 111.69 g.mol-1 of Fe2.
- Sun Jun 03, 2018 11:19 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Molecular Shape
- Replies: 6
- Views: 783
Re: Molecular Shape
But if you have more lone pairs, they would push the other bonds/molecules even further away from them correct? Thank you:)
- Sun Jun 03, 2018 11:05 pm
- Forum: *Molecular Orbital Theory (Bond Order, Diamagnetism, Paramagnetism)
- Topic: AXE formula
- Replies: 32
- Views: 12318
Re: AXE formula
I believe if they give you the molecular formula and the AXE formula you could determine the shape. Say if you were given H2O: AX2E2... then you know there are 2 bonding atoms and 2 lone pairs. Then once you draw the molecule out you could figure out its shape.
- Sun Jun 03, 2018 10:58 pm
- Forum: Formal Charge and Oxidation Numbers
- Topic: Formal charge purpose
- Replies: 40
- Views: 7262
Re: Formal charge purpose
If you had a molecule with three different lewis structures (resonance) and they had formal charges each with multiple zeros, would you pick the lewis structure with the most zeros/lowest formal charge to be the most stable lewis structure of that molecule?
- Thu May 24, 2018 10:18 pm
- Forum: Lewis Structures
- Topic: chapter 3 hw #57 question
- Replies: 5
- Views: 736
Re: chapter 3 hw #57 question
Could someone please explain to me how we know that we are supposed to use oxygen for this problem? Am I missing something in the question where it says to use oxygen, or is it because it's a certain type of ion?
Thank you:)
Thank you:)
- Thu May 24, 2018 8:50 pm
- Forum: Lewis Structures
- Topic: 3.41 part c
- Replies: 4
- Views: 606
Re: 3.41 part c
When I added up the total amount of valence e- I should have, I got 30 valence e-. In the solutions manual, they started with NH2 first, but in my lewis structure I started with H2C first. I still have the same amount of total valence e-'s as the lewis structure in the solutions manual. Does it matt...
- Wed May 23, 2018 7:06 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Bond Angle
- Replies: 10
- Views: 1166
Re: Bond Angle
I think what determines the bond angles is that each atom has to be equally spaced from one another. The exact numbers are calculated experimentally, so we don't have to know them.
- Sun May 20, 2018 11:16 pm
- Forum: Properties of Electrons
- Topic: Midterm #2 question 4a,b
- Replies: 5
- Views: 824
Re: Midterm #2 question 4a,b
I'm not sure if this is correct, but I think part b is asking if the wavelength is too small to be detected. So you would just have to see if your final answer is a very very small number.
- Sun May 20, 2018 11:13 pm
- Forum: Lewis Structures
- Topic: Lone pair electrons
- Replies: 5
- Views: 807
Re: Lone pair electrons
I think you would use the line only when it is bonding with something else, so it wouldn't be a lone pair.
- Sun May 20, 2018 11:08 pm
- Forum: Dipole Moments
- Topic: the r power
- Replies: 2
- Views: 365
Re: the r power
r is the distance between the atoms or molecules, and in the textbook on page 193 it states that "It turns out that the potential energy varies as the sixth power of the distance between the molecules... Doubling the separation of polar molecules reduces the strength of the interaction by a fac...
- Sun May 13, 2018 11:17 pm
- Forum: Octet Exceptions
- Topic: OCTET RULE [ENDORSED]
- Replies: 10
- Views: 1099
Re: OCTET RULE [ENDORSED]
When atoms are combining covalently, meaning that they are sharing electrons, they want to get a full outer orbital of 8 electrons and become stable.
- Sun May 13, 2018 11:06 pm
- Forum: Lewis Structures
- Topic: resonance
- Replies: 6
- Views: 709
Re: resonance
I'm not sure if this is correct, but what I understand resonance to be is when you can draw lewis structures of atoms bonding in different, yet equivalent ways. With the example in lecture of NO3-, we can draw it with a double bond connecting an O and the N on the left side, right side, or from the ...
- Wed May 09, 2018 1:17 am
- Forum: Administrative Questions and Class Announcements
- Topic: Missed lecture [ENDORSED]
- Replies: 1
- Views: 325
Re: Missed lecture [ENDORSED]
Lecture on Monday covered part of chapter 3. We talked about covalent bonds, octet rule, how to draw lewis structures, and resonance.
- Sun May 06, 2018 1:44 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: Question about HW#2.37
- Replies: 1
- Views: 366
Re: Question about HW#2.37
When it says an electron in the s-orbital can penetrate to the nucleus of an atom, penetration is describing how close an electron can get to the nucleus. On this website, https://chem.libretexts.org/Core/Physical_and_Theoretical_Chemistry/Quantum_Mechanics/10%3A_Multi-electron_Atoms/Multi-Electron_...
- Sun May 06, 2018 1:30 pm
- Forum: Electron Configurations for Multi-Electron Atoms
- Topic: Unpaired Electrons from Electron Configurations [ENDORSED]
- Replies: 2
- Views: 1266
Re: Unpaired Electrons from Electron Configurations [ENDORSED]
If you are given the electron configuration of an element, you can determine the number of unpaired electrons by drawing out the orbital diagram. Then when you fill in the levels, you can see which subshells do not have paired electrons. I found this video helpful when I was stuck on this too: https...
- Sun May 06, 2018 1:18 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: Switching 3d and 4s
- Replies: 6
- Views: 1290
Re: Switching 3d and 4s
Just to clarify, this means that for any element with an electron configuration that has both s and d orbitals, the d orbital would come before the s and followed by p. But for elements without the d orbital it would be the s orbital first. Correct me if I am wrong. And also s orbitals have higher ...
- Sun Apr 29, 2018 11:07 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: HELP hw problem 2.17 [ENDORSED]
- Replies: 5
- Views: 503
Re: HELP hw problem 2.17 [ENDORSED]
When l=2, m can be -2, -1, 0, 1, 2 which corresponds to 5 orbitals.
I got this from the chart we wrote in lecture on 4/25.
Hope this helps :)
I got this from the chart we wrote in lecture on 4/25.
Hope this helps :)
- Sun Apr 29, 2018 10:58 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: orbitals
- Replies: 3
- Views: 498
Re: orbitals
In class when the professor gave an example to find the orbitals for Bromine, he used shorthand and wrote [Ar] instead of repeating the same orbitals. I believe he said when we use shorthand to use the elements that are noble gases (group 18), but I am not sure exactly why that is the case. When you...
- Sun Apr 29, 2018 10:38 pm
- Forum: Photoelectric Effect
- Topic: Test 2 final question [ENDORSED]
- Replies: 15
- Views: 1930
Re: Test 2 final question [ENDORSED]
I'm confused about the concept of photons versus energy. are photons the energy unit and therefore the more photons present the more electrons released? or are photons different from energy? I think photons *have* energy, but photons are not the energy unit (?). In the textbook it says, "It is...
- Sun Apr 22, 2018 10:26 pm
- Forum: Balancing Chemical Reactions
- Topic: Q4
- Replies: 3
- Views: 575
Re: Q4
I'm not sure if everyone's tests are in the same order but my Q4 uses Phenanthrene. I wrote a balanced equation for the combustion of Phenanthrene (C14H10). So first I wrote C14H10 + O2 ---> CO2 + H20 Then I wrote the elements in a column and how much the elements had on each side of the equation, a...
- Sun Apr 22, 2018 10:10 pm
- Forum: Properties of Light
- Topic: Memorizing formulas test 2
- Replies: 16
- Views: 1776
Re: Memorizing formulas test 2
I don't have any fun/easy ways to memorize the formulas, but I believe Professor Lavelle said that all formulas will be given to us on tests/exams. We just have to know how to apply them.
- Sun Apr 22, 2018 10:06 pm
- Forum: Properties of Light
- Topic: Wavelength/frequency
- Replies: 11
- Views: 1319
Re: Wavelength/frequency
Increasing the wavelength decreases the kinetic energy of ejected electrons because when you have E=hv, and you substitute in the frequency (v) as c/(lambda) you get.. E= h * c/(lambda). So when you increase the wavelength, lambda, it makes the fraction c/(lambda) a smaller number overall which mean...
- Sun Apr 15, 2018 11:26 pm
- Forum: Photoelectric Effect
- Topic: Photoelectric Effect Diagram [ENDORSED]
- Replies: 2
- Views: 418
Re: Photoelectric Effect Diagram [ENDORSED]
DET stands for the detector that detects the kinetic energy of the ejected electron.
- Sun Apr 15, 2018 11:24 pm
- Forum: SI Units, Unit Conversions
- Topic: Avogadro #
- Replies: 9
- Views: 1209
Re: Avogadro #
When a question asks you to find how many molecules there are of something, you would use Avogadro's number in your calculations. For example in E.29 (c) "E.29 A chemist measured out 8.61 g of copper(II) chloride tetrahydrate, CuCl2*4H2O. (a) How many moles of CuCl24H2O were measured out? (b) H...
- Sun Apr 15, 2018 11:14 pm
- Forum: Properties of Electrons
- Topic: Geiger -Marsden experiment [ENDORSED]
- Replies: 5
- Views: 660
Re: Geiger -Marsden experiment [ENDORSED]
The Geiger-Marsden experiment showed the nuclear model of an atom. Because some of positively charged alpha particles deflected more than 90 degrees and some bounced straight back, this suggested that the alpha particle was repelled by the positive charge of a nucleus. Protons and neutrons in the nu...
- Sun Apr 15, 2018 10:45 pm
- Forum: Balancing Chemical Reactions
- Topic: Whole numbers
- Replies: 7
- Views: 931
Re: Whole numbers
Subscripts must never be changed because that would change the entire formula. If you have a fraction like 33/2 for one coefficient, you can just multiply the whole equation by 2 in this case to get whole numbers (which is preferable).
- Sun Apr 15, 2018 10:41 pm
- Forum: Photoelectric Effect
- Topic: Example 1.5 [ENDORSED]
- Replies: 8
- Views: 990
Re: Example 1.5 [ENDORSED]
Emely Reyna 1F wrote:You should look at the constants worksheet that value is given.
I was confused about where the book got this number too. So for all electrons, their mass will always be the same?
- Wed Apr 11, 2018 5:38 pm
- Forum: Balancing Chemical Reactions
- Topic: Another Way to Balance
- Replies: 7
- Views: 1075
Re: Another Way to Balance
This might still be guessing and checking, but when I balance equations I write down each element in a column. Then, I write how many there are on the left side of the equation to the left of the element & how many there are on the right side of the equation to the right of the element. Then whe...

