Search found 27 matches
- Wed Jun 13, 2018 11:31 pm
- Forum: Calculating pH or pOH for Strong & Weak Acids & Bases
- Topic: French Toast Number 27 Part B
- Replies: 4
- Views: 608
Re: French Toast Number 27 Part B
you just need to do 14-(-log(1.1)) since you already know that the concentration of the new mixture is .55 - just multiply that by 2 moles of HO- to get the concentration
- Sun Jun 10, 2018 10:36 pm
- Forum: Amphoteric Compounds
- Topic: Amphoteric and amphiprotic
- Replies: 6
- Views: 1458
Re: Amphoteric and amphiprotic
all amphoteric substances (can act as both acids and bases) are amphiprotic, but amphiprotic substances specifically can donate and accept hydrogen ions. not all acid/base reaction make use of hydrogen ion exchange
- Sun Jun 10, 2018 10:29 pm
- Forum: Bronsted Acids & Bases
- Topic: 12.15
- Replies: 1
- Views: 388
Re: 12.15
Kw is a water constant, so if you know the concentration of OH- you can find the concentration of H3o+
- Sun Jun 10, 2018 9:02 pm
- Forum: Properties of Electrons
- Topic: Wave-like Properties of Electron
- Replies: 5
- Views: 674
Re: Wave-like Properties of Electron
i think it's typically like 1x10^-18
- Sat Jun 02, 2018 9:52 pm
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: Significance of Coordination Number?
- Replies: 7
- Views: 2576
Re: Significance of Coordination Number?
i guess it could help with figuring out the shape of a molecule? unsure if it has any other important values that we've talked about
- Sat Jun 02, 2018 9:48 pm
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: Chelate? [ENDORSED]
- Replies: 2
- Views: 403
Re: Chelate? [ENDORSED]
just a compound with two or more ligands on the central atom
- Wed May 30, 2018 11:23 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Problem 4.9
- Replies: 3
- Views: 454
Problem 4.9
Why is the shape of ICl3 T-shaped instead of trigonal planar or trigonal pyramidal?
- Sun May 27, 2018 11:07 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: lone pairs and polarity
- Replies: 2
- Views: 503
Re: lone pairs and polarity
molecules don't need to have an odd number of lone pairs to be polar (h2o only has 2 lone pairs on the oxygen), but basically the lone pairs push the electrons on the other atoms away from their atom, which makes a molecule polar
- Thu May 24, 2018 8:26 am
- Forum: Lewis Structures
- Topic: 3.45 quick question
- Replies: 3
- Views: 491
Re: 3.45 quick question
nitrogen has a lower electronegativity since it's closer to the left (or further away from fluorine), so it goes in the center
- Wed May 23, 2018 10:15 pm
- Forum: Formal Charge and Oxidation Numbers
- Topic: Formal charge with ions
- Replies: 4
- Views: 805
Formal charge with ions
when calculating formal charges of ions, do we add/subtract the indicated charges on the atoms that they influence? or do we just use the typical number of valence electrons?
- Sun May 20, 2018 11:41 pm
- Forum: Ideal Gases
- Topic: PV=nRT
- Replies: 3
- Views: 673
Re: PV=nRT
i think you might not be able to use them since PV=nRT is for gas and m1v1=m2v2 is for liquid solutions - not 100% sure though
- Sun May 20, 2018 11:32 pm
- Forum: Bond Lengths & Energies
- Topic: Bond Length
- Replies: 9
- Views: 1135
Re: Bond Length
I'm not sure if there's any formula for bond length (at least not one that we've covered) but generally triple bonds are shorter than double bonds which are shorter than single bonds. it has to do with the fact that when you share more electrons, they're more strongly drawn to the nuclei of the atom...
- Sun May 06, 2018 11:06 pm
- Forum: Bohr Frequency Condition, H-Atom , Atomic Spectroscopy
- Topic: midterm Q.7b [ENDORSED]
- Replies: 3
- Views: 520
Re: midterm Q.7b [ENDORSED]
the negative sign just means that bound electrons have lower energy than free electrons. the E(infinity) indicates that, since a free electron could be thought of as just being in a very far-off shell (high n value)
- Sun May 06, 2018 10:56 pm
- Forum: Molarity, Solutions, Dilutions
- Topic: Test 1. Q7.
- Replies: 1
- Views: 372
Re: Test 1. Q7.
my TA told us that there wasn't enough information to answer this question, so they only checked for he amount of C and H that we found
- Sun May 06, 2018 10:55 pm
- Forum: General Science Questions
- Topic: Solutions for tests
- Replies: 3
- Views: 555
Re: Solutions for tests
probably go to office hours
- Sun Apr 29, 2018 8:52 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: 2.13
- Replies: 3
- Views: 334
Re: 2.13
the cartesian axis is just the 3D axis used to describe the shape of the orbital, like in math classes how you can draw vectors on 3D graphs. the lobe orientation is based on where the lobe lies on this axis
- Sun Apr 29, 2018 8:51 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: Why does 4s fill up before 3d?
- Replies: 4
- Views: 482
Re: Why does 4s fill up before 3d?
s, p, d, and f are short for sharp, principle, diffuse, and fundamental based on the type of spectral lines the orbitals make
- Fri Apr 27, 2018 3:24 pm
- Forum: DeBroglie Equation
- Topic: does everything have a wavelength
- Replies: 10
- Views: 1541
does everything have a wavelength
just kinda forgot this in my notes, but while im on the topic is there any kind of cutoff for when we really consider an object having a wavelength?
- Sun Apr 22, 2018 10:15 pm
- Forum: Balancing Chemical Reactions
- Topic: Q4
- Replies: 3
- Views: 571
Re: Q4
Organic compounds pretty much alway combust into water and co2, so the balances equation is 2C13H10 + 31O2 -> 26CO2 + 10H20
- Sun Apr 22, 2018 10:13 pm
- Forum: Heisenberg Indeterminacy (Uncertainty) Equation
- Topic: ∆P and ∆X
- Replies: 5
- Views: 666
Re: ∆P and ∆X
I find it easier to think of them more like values with margins of error, so delta P and delta X are ranges of values where the true value is somewhere in the interval.
- Sun Apr 22, 2018 10:11 pm
- Forum: Properties of Light
- Topic: Memorizing formulas test 2
- Replies: 16
- Views: 1755
Re: Memorizing formulas test 2
all the formulas and constants for this course were on the front cover of the last test, I don't think you have to worry about memorizing them
- Sun Apr 15, 2018 8:48 pm
- Forum: Photoelectric Effect
- Topic: Example 1.5 in the textbook [ENDORSED]
- Replies: 2
- Views: 401
Re: Example 1.5 in the textbook [ENDORSED]
we divide the velocity of the electron by planck's constant to isolate the frequency (v) and then plug this frequency equation in to c=v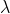
to find the wavelength of the radiation.
to find the wavelength of the radiation.
- Sun Apr 15, 2018 8:42 pm
- Forum: Photoelectric Effect
- Topic: Kinetic Energy [ENDORSED]
- Replies: 2
- Views: 381
Re: Kinetic Energy [ENDORSED]
pretty much! the kinetic energy is just the energy that the electrons have from the photons after they've been knocked off the metal.
- Sun Apr 15, 2018 8:41 pm
- Forum: Properties of Light
- Topic: Calculating the energy of a photon [ENDORSED]
- Replies: 2
- Views: 374
Re: Calculating the energy of a photon [ENDORSED]
h=6.626 x 10^-34, which is planck's constant. v is the frequency of blue light, which is the 6.4 x 10^14 that you'd found before
- Sun Apr 08, 2018 10:23 pm
- Forum: Significant Figures
- Topic: Why are sig figs important?
- Replies: 13
- Views: 16202
Re: Why are sig figs important?
I guess sig figs are used to just so that we have a system to base our calculations off of such that we'll all get the same answer as long as we do it correctly. It limits the variation in how frequently we round and to which decimal place, which i guess helps to keep answers consistent across the b...
- Sun Apr 08, 2018 10:16 pm
- Forum: Empirical & Molecular Formulas
- Topic: Molecular/Empirical Formulas
- Replies: 2
- Views: 312
Re: Molecular/Empirical Formulas
Generally carbon comes first, then hydrogen, then the remaining elements in alphabetical order.
- Sun Apr 08, 2018 10:15 pm
- Forum: Balancing Chemical Reactions
- Topic: Balancing the Equation
- Replies: 9
- Views: 1220
Re: Balancing the Equation
I used to write out the number of each element below the equations so that I could visualize it better and then look for common factors to balance chemical equations. You could also set up a system of equations with variable coefficients before each molecule in the reaction and balance it that way, ...

