Search found 61 matches
- Fri Mar 15, 2019 12:51 pm
- Forum: Arrhenius Equation, Activation Energies, Catalysts
- Topic: 7th edition 7D.1
- Replies: 1
- Views: 228
7th edition 7D.1
The rate constant of the first-order reaction 2N2O (g) = 2N2 (g) + O2 (g) is 0.76 s^-1 at 1000 K and 0.87 s^-1 at 1030 K. How do you calculate the activation energy for this reaction?
- Thu Mar 14, 2019 10:20 pm
- Forum: Arrhenius Equation, Activation Energies, Catalysts
- Topic: catalyst vs intermediate
- Replies: 9
- Views: 895
catalyst vs intermediate
What is the difference between the catalyst and intermediate of the reaction?
- Sun Mar 10, 2019 4:54 pm
- Forum: First Order Reactions
- Topic: 7th edition 7B.7
- Replies: 1
- Views: 278
7th edition 7B.7
Substance A decomposes in a first-order reaction and its half life is 355 s. How much time must elapse for the concentration of A to decrease to (a) one-eighth of its initial concentration; (b) one-fourth of its initial concentration; (c) 15% of its initial concentration; (d) one-ninth of its initia...
- Sun Mar 10, 2019 3:58 pm
- Forum: First Order Reactions
- Topic: 7th edition 7B.3
- Replies: 1
- Views: 2177
7th edition 7B.3
Determine the rate constant for each of the following first order reactions, in each case expressed for the rate loss of A:
how do you determine the rate constant for part c) 2A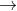 B + C, given that [Ao] = 0.153 mol/L and that after 115 s the concentration of B rises to 0.34 mol/L
B + C, given that [Ao] = 0.153 mol/L and that after 115 s the concentration of B rises to 0.34 mol/L
how do you determine the rate constant for part c) 2A
- Sun Mar 10, 2019 3:30 pm
- Forum: Method of Initial Rates (To Determine n and k)
- Topic: 7th edition 7A. 17
- Replies: 3
- Views: 730
7th edition 7A. 17
For part c) determine the value of the rate constant, I get 2.85 as the answer for K not 2.85 x 10^12. How do you get the correct answer and why are the units L^4 x mmol^4 x s^-1 as shown in the back of the book?
- Sun Mar 10, 2019 3:21 am
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: test 2
- Replies: 1
- Views: 242
test 2
Why is delta G not positive in photosynthesis?
- Sat Mar 02, 2019 4:33 pm
- Forum: Balancing Redox Reactions
- Topic: 7th edition 6L.5
- Replies: 1
- Views: 313
7th edition 6L.5
Write the half-reactions, the balanced equation for the cell reaction, and the cell diagram for each of following skeletal equations:
for part c) Cl2(g) + H2(g) = HCl(aq)
why do both the anode and cathode separate HCl to either H+ of Cl-?
for part c) Cl2(g) + H2(g) = HCl(aq)
why do both the anode and cathode separate HCl to either H+ of Cl-?
- Sat Mar 02, 2019 4:22 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: 7th edition 6M.5
- Replies: 1
- Views: 415
7th edition 6M.5
For each reaction that is spontaneous under standard conditions (K>1), write a cell diagram, determine the standard cell potential, and calculate deltaG for the reaction:
for part b) 2Hg^2+(aq) + 2Br-(aq) = Hg2^2+(aq) + Br2(l)
How do you determine that this reaction is not spontaneous?
for part b) 2Hg^2+(aq) + 2Br-(aq) = Hg2^2+(aq) + Br2(l)
How do you determine that this reaction is not spontaneous?
- Sat Mar 02, 2019 4:16 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: 7th edition 6M.5
- Replies: 1
- Views: 275
7th edition 6M.5
For each reaction that is spontaneous under standard conditions (K>1), write a cell diagram, determine the standard cell potential, and calculate deltaG for the reaction: For part a) 2NO3-(aq) + 8H+(aq) + 6Hg(l) = 2Hg2^2+(aq) +2NO(g) + 2H2O(l) why does the answer in the textbook use O(g) in the cell...
- Sun Feb 24, 2019 11:42 pm
- Forum: Work, Gibbs Free Energy, Cell (Redox) Potentials
- Topic: determining n
- Replies: 4
- Views: 524
determining n
When using this equation, delta G = -nFEcell, how do you determine the value of moles (n)?
- Sun Feb 24, 2019 12:42 pm
- Forum: Balancing Redox Reactions
- Topic: acidic vs basic solution
- Replies: 1
- Views: 243
acidic vs basic solution
In the book, each problem tells us whether the reaction takes place in an acidic or basic solution. What is the importance in knowing what type of solution the reaction occurs in?
- Sun Feb 24, 2019 12:39 pm
- Forum: Balancing Redox Reactions
- Topic: reducing/oxidizing agents
- Replies: 1
- Views: 216
reducing/oxidizing agents
What is the difference between being a reducing agent vs being reduced and being an oxidizing agent vs being oxidized?
- Sun Feb 24, 2019 11:54 am
- Forum: Balancing Redox Reactions
- Topic: 7th edition 6K.1
- Replies: 2
- Views: 356
7th edition 6K.1
The following redox reaction is used in acidic solution in the Breathalyzer test to determine the level of alcohol in blood: H+ + Cr2O7^2- + C2H5OH = Cr^3+ + C2H4O + H2O. When writing the balanced oxidation half-reaction, why can you add 2 H+ to the reaction to get C2H5OH = C2H4O + 2H+ + 2e- when th...
- Sun Feb 17, 2019 11:09 pm
- Forum: Entropy Changes Due to Changes in Volume and Temperature
- Topic: 7th edition 4I. 9
- Replies: 3
- Views: 547
7th edition 4I. 9
Initially an ideal gas at 323 K occupies 1.67 L at 4.95 atm. The gas is allowed to expand to 7.33 L by two pathways: (a) isothermal, reversible expansion; (b) isothermal, irreversible free expansion. Calculate delta S total, delta S, and delta S surroundings for each pathway. How would you solve for...
- Sun Feb 17, 2019 11:01 pm
- Forum: Entropy Changes Due to Changes in Volume and Temperature
- Topic: 7th edition 4I. 5
- Replies: 3
- Views: 452
7th edition 4I. 5
Suppose that 50.0 g of water at 20.0 C is mixed with 65.0 g of water at 50.0 C at constant atmospheric pressure in a thermally insulated vessel. Calculate delta S and delta S total for the process?
Why is there a difference between delta S and delta S total for this process?
Why is there a difference between delta S and delta S total for this process?
- Sun Feb 17, 2019 10:45 pm
- Forum: Calculating Standard Reaction Entropies (e.g. , Using Standard Molar Entropies)
- Topic: 4I.3
- Replies: 5
- Views: 10588
Re: 4I.3
In part (a), the equation you use to calculate the standard enthalpy of vaporization is delta S (vap) = delta H (vap) / T(boiling point in K). So you get delta H (vap) = (85 J/K)(80 + 273 K) = 30,005 J or 30.0 kJ. For part (b), the standard entropy change of the system is calculated by multiplying ...
- Sun Feb 10, 2019 5:25 pm
- Forum: Thermodynamic Definitions (isochoric/isometric, isothermal, isobaric)
- Topic: Reversible vs. Irreversible
- Replies: 2
- Views: 327
Re: Reversible vs. Irreversible
In a reversible process, the direction of the process can be "reversed" in incremental steps while irreversible processes can't be reversed and don't occur in discrete steps. Reversible expansion does more work while irreversible expansion does work against constant pressure.
- Sun Feb 10, 2019 5:17 pm
- Forum: Third Law of Thermodynamics (For a Unique Ground State (W=1): S -> 0 as T -> 0) and Calculations Using Boltzmann Equation for Entropy
- Topic: degeneracy
- Replies: 2
- Views: 360
degeneracy
Can someone explain the concept of W (degeneracy) and how to solve for it?
- Sun Feb 10, 2019 5:03 pm
- Forum: Calculating Work of Expansion
- Topic: work equations
- Replies: 4
- Views: 489
work equations
Can someone explain when we use this equation to solve for work w = - P ∆V versus when we use this equation w = -nRT ln V2/V1 ?
- Sun Feb 03, 2019 12:11 am
- Forum: Calculating Work of Expansion
- Topic: integral work equation
- Replies: 2
- Views: 354
integral work equation
Will we need to know how to use the work equation with the integral on a test or was Dr. Lavelle just explaining the background and giving more information for the other equations?
- Sun Feb 03, 2019 12:05 am
- Forum: Concepts & Calculations Using First Law of Thermodynamics
- Topic: 7th edition 4A.3
- Replies: 1
- Views: 174
7th edition 4A.3
Air in a bicycle pump is compressed by pushing in the handle. The inner diameter of the pump if 3.0 cm and the pump is depressed 20. cm with a pressure of 2.00 atm. (a) how much work is done in the compression? (b) is the work positive or negative with respect to the air in the pump? (c) what is the...
- Tue Jan 29, 2019 6:33 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: 7th edition 4C.1
- Replies: 1
- Views: 274
7th edition 4C.1
Which gaseous compound do you expect to have the higher molar heat capacity, NO or NO2? How would you determine which compound has the higher molar heat capacity?
- Sun Jan 27, 2019 12:56 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: 7th edition 4D.15
- Replies: 1
- Views: 269
7th edition 4D.15
Determine the reaction enthalpy for the hydrogenation of ethyne to ethane, C2H2 (g) + 2H2 (g) = C2H6 (g), from the following data: ΔHc(C2H2, g) = -1300 kJ/mol, ΔHc(C2H6, g) = -1560 kJ/mol, ΔHc(H2, g) = -286 kJ/mol. Why is the answer -312 kJ/mol and not +312 kJ/mol?
- Sun Jan 27, 2019 12:38 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: Accuracy of Enthalpy Calculation
- Replies: 3
- Views: 473
Re: Accuracy of Enthalpy Calculation
The actual bond enthalpy depends on the neighboring molecules as well, so that is why there are varying values for the other molecules and the average is given.
- Sun Jan 20, 2019 9:40 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: 7th edition 6A.19
- Replies: 1
- Views: 206
7th edition 6A.19
For part (c) the concentration of H30+ is 3.1 mol/L. Since [H30+] x [OH-] = 10^-14, I keep getting 3.2 x 10^-15 while the answer in the book is 3.2 x 10^-12. What am I doing different/wrong and how do you get the correct answer?
- Sun Jan 20, 2019 9:28 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: K=1
- Replies: 2
- Views: 193
Re: K=1
Because the equation for the equilibrium constant is K=products/reactants, when K is equal to 1 the amount of products is equal to the amount of reactants at equilibrium.
- Sun Jan 20, 2019 8:57 pm
- Forum: Applying Le Chatelier's Principle to Changes in Chemical & Physical Conditions
- Topic: pressure and volume
- Replies: 5
- Views: 511
pressure and volume
Can someone explain the relationship between pressure and volume for Le Chatelier's Principle.
- Sun Jan 20, 2019 8:38 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: ice table mols/molarity
- Replies: 3
- Views: 214
ice table mols/molarity
In 5I.23 7th edition, the problem states that "a reaction mixture consisting of 2.00 mol CO and 3.00 mol H2 is placed in a reaction vessel of volume 10.0 L and heated at 1200 K. At equilibrium, 0.478 mol Ch4 was present in the system. Determine the value of Kc for the reaction CO(g) + 3H2(g) = ...
- Sun Jan 13, 2019 7:41 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: 7th edition 5I.13
- Replies: 1
- Views: 146
7th edition 5I.13
Part B of the question says that "if 2.0 mmol F2 was placed into the reaction vessel instead of the chlorine, what would be its equilibrium composition at 1000K." I was able to solve correctly for part A, but I can't seem to get the correct answer for part B. Can someone explain the steps ...
- Sun Jan 13, 2019 6:36 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: 7th edition 5I.3
- Replies: 2
- Views: 265
7th edition 5I.3
"In a gas phase equilibrium mixture of H2, I2, and HI at 500 K, [HI] = 2.21 x 10^-3 mol/L and [I2] = 1.46 x 10^-3 mol/L. Given the value of the equilibrium constant in table 5G.2, calculate the equilibrium molar concentration of H2." How do you know what the products and reactants are betw...
- Sun Jan 13, 2019 5:18 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Kp Units
- Replies: 6
- Views: 557
Re: Kp Units
Equilibrium constants (Kp) will not have units for your final answer, so while you are evaluating with partial pressure the units do not matter as long as all the partial pressures have the same units.
- Sat Dec 08, 2018 11:57 pm
- Forum: Calculating pH or pOH for Strong & Weak Acids & Bases
- Topic: sig figs
- Replies: 4
- Views: 521
sig figs
In high school, I was taught to add a sig fig when you take the -log to calculate the pH; however, there have been different things said about sig figs for this class regarding this topic. What is the expected rule for sig figs when taking the log that I should follow?
- Sat Dec 08, 2018 11:47 pm
- Forum: Biological Examples
- Topic: coordination compounds
- Replies: 2
- Views: 465
coordination compounds
What specific coordination compounds are necessary to know their biological importance?
- Sat Dec 08, 2018 11:44 pm
- Forum: Naming
- Topic: 7th edition 9C.1 table
- Replies: 1
- Views: 312
7th edition 9C.1 table
Do we have to memorize all the common ligands in the table on pg. 724?
- Sun Dec 02, 2018 9:40 pm
- Forum: Lewis Acids & Bases
- Topic: lewis acids & bases
- Replies: 3
- Views: 519
lewis acids & bases
I know that lewis acids accept electrons while lewis bases donate electrons, but how can you determine whether the species is able to accept or donate the electron pairs? Do you have to draw the lewis structures to determine this?
- Sun Dec 02, 2018 9:28 pm
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: 7th edition 9C.7
- Replies: 1
- Views: 200
7th edition 9C.7
What is a chelating complex and how can you determine whether or not one will form between the three isomers of diaminobenzene given?
- Sun Dec 02, 2018 9:21 pm
- Forum: Naming
- Topic: 7th edition 9C.1
- Replies: 1
- Views: 252
7th edition 9C.1
For part a of the question the name of [Fe(CN)6]4- is hexacyanoferrate(ii) ion. Why is "ferrate" used for the naming of Fe when the element is iron?
- Sun Nov 25, 2018 10:18 pm
- Forum: Hybridization
- Topic: 7th edition 2F.1
- Replies: 1
- Views: 286
7th edition 2F.1
State the relative orientation of each of the following hybrid orbitals: (a) sp3; (b) sp; (c) sp3d2; (d) sp2
How do you determine the orientation of these hybrid orbitals?
How do you determine the orientation of these hybrid orbitals?
- Sun Nov 25, 2018 10:03 pm
- Forum: Hybridization
- Topic: hybridization
- Replies: 2
- Views: 336
hybridization
Is there another way to determine the hybridization of an atom without drawing the molecule's lewis structure or is that the only way to determine hybridization?
- Sun Nov 25, 2018 9:57 pm
- Forum: Hybridization
- Topic: sigma/pi bonds
- Replies: 7
- Views: 711
sigma/pi bonds
Besides that a single bond = 1 sigma bond, a double bond = 1 sigma + 1 pi bond, and a triple bond = 1 sigma + 2 pi bonds, what else is important about these types of bonds? What is the concept behind them?
- Sun Nov 18, 2018 6:16 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: 7th edition 2E. 21
- Replies: 4
- Views: 364
7th edition 2E. 21
For part d of the question where it asks for the Lewis structure and bond angle N2H4, the book gives the exact bond angle of 107. I know in class Dr. Lavelle said for trigonal pyramidal structures we only have to know that the angle is less than 109.5, so for our answer would it be okay to say less ...
- Sun Nov 18, 2018 5:43 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: 7th edition 2E.13
- Replies: 3
- Views: 397
7th edition 2E.13
For part (a) I3- the lewis structure in the book is vertical. Is it important to draw the linear structure vertically rather than horizontally?
- Sun Nov 18, 2018 5:24 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: molecular shape and bond angles
- Replies: 3
- Views: 382
molecular shape and bond angles
What molecular shapes do we need to know the exact bond angles of and what molecular shapes do we need know that are less than 120 or 109.5 and not the exact angles?
- Sun Nov 11, 2018 7:52 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: 7th edition 2E. 5
- Replies: 2
- Views: 256
7th edition 2E. 5
For the CH2F2 structure, the answer in the book shows more of a 3D model indicated by the filled in wedge and striped line wedge for the bonds between the carbon and hydrogens. Do we need to draw the lewis structure like that or can we draw it how we have been doing, in the 2D model?
- Sun Nov 11, 2018 6:08 pm
- Forum: Ionic & Covalent Bonds
- Topic: 7th edition 2D.3
- Replies: 2
- Views: 310
7th edition 2D.3
Which of these compounds has bonds that are primarily ionic? (a) BBr3; (b) BaBr2; (c) BeBr2
Why is BeBr2 not considered to have primarily ionic bonds as the compound is between a metal and nonmetal, meaning it has ionic bonds?
Why is BeBr2 not considered to have primarily ionic bonds as the compound is between a metal and nonmetal, meaning it has ionic bonds?
- Sun Nov 11, 2018 5:52 pm
- Forum: Bond Lengths & Energies
- Topic: 7th edition 2D. 13 [ENDORSED]
- Replies: 2
- Views: 459
7th edition 2D. 13 [ENDORSED]
Place the following molecules or ions in order of decreasing bond length: the CO bond in CO, CO2, CO3^2-... How do you determine the bond lengths for these molecules/ions in order to determine the order? Do you need to draw the lewis structures?
- Sun Nov 04, 2018 4:05 pm
- Forum: Lewis Structures
- Topic: 7th edition 2B.9
- Replies: 4
- Views: 399
7th edition 2B.9
For these compounds, ammonium chloride, potassium phosphide, and sodium hypochlorite, why do you separate them into their cations and anions to draw the Lewis structures?
- Sun Nov 04, 2018 3:57 pm
- Forum: Lewis Structures
- Topic: 7th edition 2B.7
- Replies: 2
- Views: 251
7th edition 2B.7
In the given lewis structure, how are you able to identify the unknown element?
- Sun Nov 04, 2018 3:50 pm
- Forum: Lewis Structures
- Topic: Lewis Structures with Br
- Replies: 1
- Views: 308
Re: Lewis Structures with Br
If there were no lone pairs on Br and you fulfilled the octet rule for F then you would not have used all valence electrons. For BrF3 there are 28 valence electrons, meaning two lone pairs need to be placed on Br as just fulfilling the octet rule for the the fluorines uses 24 electrons.
- Sun Oct 28, 2018 5:29 pm
- Forum: Electron Configurations for Multi-Electron Atoms
- Topic: 7th edition 2A.9 and 2A.11
- Replies: 2
- Views: 378
7th edition 2A.9 and 2A.11
I am a little confused how to predict the ions for these two problems. 2A.9 Which M^2+ ions (M=metal) are predicted to have the following ground-state electron configurations: (a) [Ar]3d7 (b) [Ar]3d6 2A.11 Which M^3+ ions (M=metal) are predicted to have the following ground-state electron configurat...
- Sun Oct 28, 2018 5:11 pm
- Forum: Quantum Numbers and The H-Atom
- Topic: Degeneracy
- Replies: 5
- Views: 487
Re: Degeneracy
Degeneracy refers to when an orbital has equal energy. For example, the p orbital would have a degeneracy of 3 and the d orbital a degeneracy of 5.
- Sun Oct 28, 2018 4:51 pm
- Forum: Trends in The Periodic Table
- Topic: Topic 1F.11c electron affinity
- Replies: 2
- Views: 325
Re: Topic 1F.11c electron affinity
I am also confused on how sulfur was the answer for 11c. I was also wondering how to know that the electron affinity for (b) beryllium and magnesium and (d) gallium and indium are equal.
- Sun Oct 21, 2018 11:29 pm
- Forum: Heisenberg Indeterminacy (Uncertainty) Equation
- Topic: About Uncertainty in position
- Replies: 3
- Views: 391
Re: About Uncertainty in position
The uncertainty in position would be 350. pm, but in order to apply it to the equation you need to convert this to meters so the units will align with one another.
- Sun Oct 21, 2018 11:02 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: 7th edition 1D.25
- Replies: 3
- Views: 266
7th edition 1D.25
Can someone explain the reason the 2d and 4g subshells cannot exist in an atom?
- Sun Oct 21, 2018 10:38 pm
- Forum: Heisenberg Indeterminacy (Uncertainty) Equation
- Topic: HW Question Regarding 1B.27
- Replies: 6
- Views: 623
Re: HW Question Regarding 1B.27
I think for this question the answer in the back of the book is wrong. On the chemistry 14A webpage in the solution manual errors page it shows this problem is one of them. So instead of the answer being 1.3 x 10^-36 it is actually 6.7 x 10^-37. This is because the Δv should be 10, not 5 in the equa...
- Sat Oct 13, 2018 11:40 pm
- Forum: Properties of Electrons
- Topic: 7th edition 1A.11
- Replies: 3
- Views: 422
7th edition 1A.11
Will we also have to know the Paschen series (lower energy level is n=3) and the Brackett series (lower energy level is n=4) for assignments/tests if so far in class we have only discussed the Lyman series and Balmer series?
- Sat Oct 13, 2018 11:23 pm
- Forum: Einstein Equation
- Topic: 7th edition 1B. 7
- Replies: 2
- Views: 263
7th edition 1B. 7
In part B of this question, it asks for how much energy is emitted by 5.00 mg of sodium atoms and in part c is asks for how much energy is emitted by 1.00 mol of sodium atoms, both emitting light at a wavelength of 589 nm. How are you able to go from grams and mols of sodium atoms to joules?
- Sat Oct 13, 2018 11:12 pm
- Forum: Einstein Equation
- Topic: 7th edition 1B.5
- Replies: 5
- Views: 634
7th edition 1B.5
In this question, the energy is given in keV and we are given a conversion from eV to joules. What is keV and what is the difference between this unit of measurement and joules?
- Sun Oct 07, 2018 11:05 pm
- Forum: Balancing Chemical Reactions
- Topic: 7th edition L.35
- Replies: 5
- Views: 703
7th edition L.35
"Sodium bromide, NaBr, which is used to produce AgBr for use in photographic film, can itself be prepared as follows: Fe + Br2 --> FeBr2 FeBr2 + Br2 --> Fe3Br8 FeBr2 + Na2CO3 --> NaBr + CO2 + Fe3O4 What mass of iron, in kilograms, is needed to produce 2.50 t of NaBr? Note that these equations m...
- Sun Oct 07, 2018 9:48 pm
- Forum: Limiting Reactant Calculations
- Topic: different ways to determine limiting reactant
- Replies: 3
- Views: 311
different ways to determine limiting reactant
Is it possible to determine the limiting reactant a different way than how it was taught in class? I think in high school I was taught to convert the moles of each reactant to moles of product and then compare the product moles instead of just comparing the moles of the reactants to determine the li...
- Sun Oct 07, 2018 7:23 pm
- Forum: Accuracy, Precision, Mole, Other Definitions
- Topic: E15 7th Edition
- Replies: 1
- Views: 220
Re: E15 7th Edition
First you need to figure out what the metal (M) is in the metal hydroxide M(OH)2. Since you have the molar mass of the metal hydroxide you can subtract the molar mass of (OH)2 to find the molar mass of the metal. 74.10-34.02=40.08 g/mol 40.08 is the molar mass of calcium (Ca) so the sulfide would ju...

