Search found 74 matches
- Sat Mar 16, 2019 7:02 pm
- Forum: Arrhenius Equation, Activation Energies, Catalysts
- Topic: 7th Edition 7D.5
- Replies: 3
- Views: 434
Re: 7th Edition 7D.5
Please post the entire problem. Question: The rate constant of the reaction between CO2 and H2O in aqueous solution to give the HCO3- ion is 1.5 x 10^10 L mol^-1 s^-1 at 25 degrees Celsius. Determine the rate constant at human body temperature (37 degrees Celsius), given that the activation energy ...
- Sat Mar 16, 2019 1:40 pm
- Forum: Arrhenius Equation, Activation Energies, Catalysts
- Topic: 7th Edition 7D.5
- Replies: 3
- Views: 434
7th Edition 7D.5
Does anyone know why the solutions manual subtracted .59 from the Arrhenius equation expression?
- Sat Mar 16, 2019 1:30 pm
- Forum: Student Social/Study Group
- Topic: Final Jitters
- Replies: 457
- Views: 382425
Re: Final Jitters
I also have test anxiety, it's not fun. Usually just forcing myself to do lots of practice problems and having a routine (eating breakfast, sitting in the front, having a certain pen/calculator, etc whatever works for you) helps give me some comfort so I can relax some at least. Good luck and know y...
- Sat Mar 16, 2019 10:34 am
- Forum: Work, Gibbs Free Energy, Cell (Redox) Potentials
- Topic: Ecell
- Replies: 3
- Views: 2020
Re: Ecell
The Ecell being positive means that the reaction is favorable (it's spontaneous). Think of it more as energy the cell gives off (that could be used to do work) rather than the overall charge.
- Wed Mar 13, 2019 4:17 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Homework problem 7D1
- Replies: 5
- Views: 907
Re: Homework problem 7D1
I noticed the same thing. The equation is another form of the Arrhenius equation where the natural log is taken and the reverse reaction is taken into account I believe.
- Wed Mar 13, 2019 4:10 pm
- Forum: Appications of the Nernst Equation (e.g., Concentration Cells, Non-Standard Cell Potentials, Calculating Equilibrium Constants and pH)
- Topic: homework
- Replies: 1
- Views: 295
Re: homework
The natural log of 1/something is the same as the natural log of that something times negative one. This is because of the log properties ln(1) = 0 and ln(A/B) = ln(A) - ln(B).
- Sun Mar 10, 2019 5:28 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Microscopic reversibility
- Replies: 3
- Views: 413
Re: Microscopic reversibility
I believe that Lavelle said that we can assume microscopic reversibility for the purposes of our class, however, I am not 100% positive.
- Sun Mar 10, 2019 5:23 pm
- Forum: General Rate Laws
- Topic: Rate constant k
- Replies: 5
- Views: 611
Re: Rate constant k
The rate constant is a coefficient of proportionality relating the rate of a chemical reaction at a given temperature to the concentration of reactants.
- Sun Mar 10, 2019 5:17 pm
- Forum: Method of Initial Rates (To Determine n and k)
- Topic: Reaction Constant k
- Replies: 2
- Views: 431
Re: Reaction Constant k
k does change with temperature as it is temperature dependent like K and yes k is also different for different reactions.
- Sun Mar 03, 2019 5:06 pm
- Forum: Student Social/Study Group
- Topic: Post All Chemistry Jokes Here
- Replies: 9651
- Views: 3706331
Re: Post All Chemistry Jokes Here
some jokes for you
- Sun Mar 03, 2019 5:04 pm
- Forum: Method of Initial Rates (To Determine n and k)
- Topic: Order of the reactant (n)
- Replies: 4
- Views: 509
Re: Order of the reactant (n)
The order of a reactant tells us to what degree a reactant's concentration affects the rate of reaction. We determine it by using the formula below and a set of experimental data.
- Sun Mar 03, 2019 2:17 pm
- Forum: General Rate Laws
- Topic: Units of K
- Replies: 4
- Views: 506
Re: Units of K
Here's a handy table for the units of k that I like to refer to. This one is in dm^3 but this is the same as liters (L).
- Sun Feb 24, 2019 6:17 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Inert Conductors
- Replies: 3
- Views: 449
Re: Inert Conductors
Inert conductors are nonreactive solid metals that allow for electron transfer in the reaction between the oxidizing and reducing agents.
- Sun Feb 24, 2019 6:15 pm
- Forum: Balancing Redox Reactions
- Topic: Oxygen Oxidation Number
- Replies: 3
- Views: 438
Re: Oxygen Oxidation Number
Oxygen is usually assigned an oxidation state of ‒2 in its covalent compounds, such as CO, CO2, SO2, and SO3. Exceptions to this rule include peroxides (compounds containing the O2-2 group), where each oxygen is assigned an oxidation state of ‒1, as in hydrogen peroxide (H2O2), and OF2 in which oxyg...
- Sun Feb 24, 2019 6:07 pm
- Forum: Balancing Redox Reactions
- Topic: Balancing redox reactions in basic solutions
- Replies: 2
- Views: 400
Re: Balancing redox reactions in basic solutions
Here some steps for balancing redox reactions that I found helpful 1. Divide the equation into oxidation and reduction half reactions. 2. Balance all elements besides hydrogen and oxygen. 3. Balance O by adding H2O to the appropriate side of each equation. 4. Balance H by adding H+. 5. Balance the c...
- Sun Feb 17, 2019 1:17 pm
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: Calculating G if H and S are gven
- Replies: 3
- Views: 480
Re: Calculating G if H and S are gven
We aren't assuming that the Gibbs Free Energy is equal to 0, rather we are putting in 0 for its value to see when T\Delta S and \Delta H are equal to one another in order to determine at what temperatures the Gibbs Free Energy would be negative (spontaneous reaction) or positive (non spontaneous rea...
- Sun Feb 17, 2019 1:14 pm
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: calculating delta G
- Replies: 4
- Views: 450
Re: calculating delta G
Yes, standard conditions assume 298.15 K or 25 degrees Celsius, which one you use in your calculations just depends on the kind that you are doing.
- Sun Feb 17, 2019 1:12 pm
- Forum: Entropy Changes Due to Changes in Volume and Temperature
- Topic: DeltaS= nCln(T2/T1)
- Replies: 4
- Views: 1290
Re: DeltaS= nCln(T2/T1)
You know to use Cv or Cp depending on what the givens are in the question. If the system is at constant pressure then use Cp and if it is at constant volume then you use Cv.
- Sun Feb 17, 2019 1:09 pm
- Forum: Work, Gibbs Free Energy, Cell (Redox) Potentials
- Topic: Gibbs Free Energy in relation to H and S
- Replies: 3
- Views: 456
Re: Gibbs Free Energy in relation to H and S
you can solve for 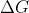 using
using  - \sum \Delta G(reactants)) because Gibbs Free Energy is defined by the state functions enthalpy and entropy, and it too is a state function.
because Gibbs Free Energy is defined by the state functions enthalpy and entropy, and it too is a state function.
- Sun Feb 10, 2019 1:24 pm
- Forum: Phase Changes & Related Calculations
- Topic: Phase Changes
- Replies: 11
- Views: 1450
Re: Phase Changes
Understanding it would definitely make problem solving easier so I would try to as much as you can. I took some notes in my discussion with the graph labeled, a picture of it is shown below. Hope it helps!
- Sun Feb 10, 2019 1:17 pm
- Forum: Concepts & Calculations Using Second Law of Thermodynamics
- Topic: Value of Variable Kb
- Replies: 4
- Views: 924
Re: Value of Variable Kb
K B is the Boltzmann constant and it provides a measure of the amount of energy (i.e., heat) corresponding to the random thermal motions of the particles making up a substance. The link below sends you to a video if you want to learn more about it. https://www.khanacademy.org/science/in-in-class11th...
- Sat Feb 09, 2019 2:08 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: methods
- Replies: 8
- Views: 963
Re: methods
If you're dealing with bond enthalpies then you use the equation: -\sum H(bonds formed))
- Sat Feb 09, 2019 2:02 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: midterm [ENDORSED]
- Replies: 4
- Views: 557
Re: midterm [ENDORSED]
The equations he will give us on the midterm can be found on his website or on your first test on the front page. Here's a link to the constants and equation sheet! https://lavelle.chem.ucla.edu/wp-content/supporting-files/Chem14B/Constants_Equations.pdf You can find this on the left hand side of hi...
- Sat Feb 02, 2019 12:57 pm
- Forum: Thermodynamic Systems (Open, Closed, Isolated)
- Topic: pressure
- Replies: 3
- Views: 414
Re: pressure
We can also assume it is at 1 atm when the question says that the reaction is occurring under standard conditions.
- Sat Feb 02, 2019 12:56 pm
- Forum: Concepts & Calculations Using First Law of Thermodynamics
- Topic: Homework sections
- Replies: 2
- Views: 269
Re: Homework sections
Yeah I also found it slightly confusing but my TA mentioned that we were doing the middle part of 4 first then moving to the beginning. We have covered 4C, 4D, 4E, and on Friday I believe we finished 4A so we are moving into 4B. After that we're going on to end of Topic 4 in the book for the thermod...
- Sat Feb 02, 2019 12:49 pm
- Forum: Phase Changes & Related Calculations
- Topic: w = -nRTln(v2/v1)
- Replies: 6
- Views: 10884
Re: w = -nRTln(v2/v1)
Also the \ln (\frac{V2}{V1}) part comes from the properties of logarithms because when you take a definite integral its F(b) - F(a) so for \int_{V1}^{V2} (\frac{1}{V})dV you get \ln (V2) - \ln (V1) which by log properties is the same as \ln(\frac{V2}{V1}) , th...
- Sat Feb 02, 2019 12:41 pm
- Forum: Concepts & Calculations Using First Law of Thermodynamics
- Topic: Friday Lecture Notes
- Replies: 2
- Views: 292
Re: Friday Lecture Notes
In my notes I have - reaction at constant P: that involve changes in the number of moles of gas then P\Delta V is significant - reaction at constant P: that involve solids and liquids, then P\Delta V is insignificant so you're right, it's not significant when solids and liquids are involved because ...
- Thu Jan 24, 2019 2:16 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: heat capacity
- Replies: 7
- Views: 792
Re: heat capacity
Specific heat capacity is the amount of heat required to raise 1 gram of a substance by one degree (i.e. 1 degree Kelvin). It is a state property because it is a property independent of how the substance was prepared (in this case heated).
- Thu Jan 24, 2019 2:12 pm
- Forum: Polyprotic Acids & Bases
- Topic: 6E.3 7th Edition
- Replies: 2
- Views: 1027
Re: 6E.3 7th Edition
Here's a link to an example of a pH calculation where you do have to include Ka2, might help you out more than my explanation. https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_(Physical_and_Theoretical_Chemistry)/Acids_and_Bases/Monoprotic...
- Thu Jan 24, 2019 2:03 pm
- Forum: Polyprotic Acids & Bases
- Topic: 6E.3 7th Edition
- Replies: 2
- Views: 1027
Re: 6E.3 7th Edition
If it is not justified then you would have to do another ICE table, calculate the equilibrium concentration of [H3O+] for the second deprotonation, and then to get the pH you add the [H3O+]s and take the -log of the over all concentration.
- Thu Jan 24, 2019 1:48 pm
- Forum: Applying Le Chatelier's Principle to Changes in Chemical & Physical Conditions
- Topic: 5J.11 (b) 7th edition
- Replies: 2
- Views: 277
Re: 5J.11 (b) 7th edition
You can tell that this reaction is endothermic because in order for a diatomic halogen to become two atoms the bond between the atoms needs to be broken and this requires energy, so you can predict that equilibrium will shift toward the products (this is because the extra energy that the heat gives ...
- Fri Jan 18, 2019 2:43 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: An Equation with both Weak Acids and Salt
- Replies: 1
- Views: 197
Re: An Equation with both Weak Acids and Salt
We never really change X to zero because if X is 0 then that means that some of the concentrations for reactant/products at equilibrium are zero which doesn't make sense, but we do neglect the X if it is added or subtracted from a concentration in our K expression if the K value is less than 10^-3.
- Fri Jan 18, 2019 2:37 pm
- Forum: Ideal Gases
- Topic: The Difference between Q and Kc [ENDORSED]
- Replies: 18
- Views: 8802
Re: The Difference between Q and Kc [ENDORSED]
Q is different from Kc because Kc indicates the ratio of products to reactants at equilibrium while Q indicates the ratio of products to reactants at any time during the reaction (such as when it has not yet reached equilibrium) we can then use Q to figure out which direction a reaction is sitting t...
- Thu Jan 17, 2019 9:50 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: ICE box
- Replies: 2
- Views: 333
Re: ICE box
yes, just be careful because some problems will give concentrations/values for product and reactant and in those cases you have to solve for Q to find out which direction the reaction is favoring before finishing the ice table to solve the actual problem.
- Sun Jan 13, 2019 11:00 am
- Forum: Ideal Gases
- Topic: K and Q rules
- Replies: 3
- Views: 419
Re: K and Q rules
I definitely think that memorizing those rules is the best way to go because it's hard to know which reaction is being favored without them if you are given concentrations/partial pressures for both products and reactants in a problem. The way that I remember them is actually the opposite of how Lav...
- Wed Jan 09, 2019 7:42 pm
- Forum: Student Social/Study Group
- Topic: New to Lavelle
- Replies: 32
- Views: 5330
Re: New to Lavelle
I definitely understand, I also found his websites to be slightly confusing to navigate at first. Trust me though you'll get the hang of it after the two weeks or so. As for outside of lecture prep, I personally found doing all the homework problems he assigns, going to Lyndon's review sessions (he'...
- Wed Jan 09, 2019 7:35 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: 6th edition, 11.23
- Replies: 4
- Views: 332
Re: 6th edition, 11.23
Two sig figs is actually right because the value 0.031 only has two sig figs since neither of the zeros are significant. If the number were .0310 then it would have three sig figs though because 0s that come after non-zeros in decimals are significant.
- Wed Jan 09, 2019 7:20 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Temperature and Equilibrium
- Replies: 3
- Views: 369
Re: Temperature and Equilibrium
In general how can you tell if determine if a reaction is exothermic or endothermic? You would have to calculate the enthalpy or \Delta H of the reaction to be able to know if a reaction is endothermic or exothermic. If the enthalpy is negative then it is exothermic and if it is positive then it is...
- Fri Dec 07, 2018 12:44 pm
- Forum: Naming
- Topic: Ligand Naming: -ido vs. -o
- Replies: 2
- Views: 293
Re: Ligand Naming: -ido vs. -o
My TA said we can use either and both naming conventions are on the ligand names in coordination compounds sheet on Lavelle's website! I guess you could consider "cyanido-kN" as "more" correct since it's the new form that IUPAC decided on, but for the final we can use whichever w...
- Fri Dec 07, 2018 12:41 pm
- Forum: Acidity & Basicity Constants and The Conjugate Seesaw
- Topic: Kw
- Replies: 1
- Views: 256
Re: Kw
Kw is the equilibrium constant for water. The constant varies depending on temperature but I don't think we'll ever be asked to calculate it because it's a constant that we use to solve problems. It's given on our formula sheet!
- Fri Dec 07, 2018 12:34 pm
- Forum: Naming
- Topic: Coordination Compounds on the Final
- Replies: 2
- Views: 327
Re: Coordination Compounds on the Final
I think memorizing them and their charges is a good idea because the names/formulas and charges aren't given in the homework problems so I think questions on the final about coordination compounds are going to be formatted similarly
- Thu Dec 06, 2018 8:26 pm
- Forum: Bond Lengths & Energies
- Topic: bond strength
- Replies: 3
- Views: 802
Re: bond strength
A molecule with a double bond will not always have a higher bowling point then one with more single bonds. This is because the physical properties like boiling and melting points are determined by intermolecular forces not intramolecular forces. Intermolecular forces are bonding forces between molec...
- Thu Dec 06, 2018 8:16 pm
- Forum: Naming
- Topic: Chemical Formulas
- Replies: 2
- Views: 362
Re: Chemical Formulas
A metal could be outside the brackets if it is bound to a coordination compound but the metal with the ligands attached to it that makes up the coordination compound is always inside the brackets.
- Tue Dec 04, 2018 9:09 pm
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: Polydentate and Coordination Number?
- Replies: 2
- Views: 766
Re: Polydentate and Coordination Number?
Being polydentate is different than a coordination compound's coordination number. If something has a coordination number of two then this means the metal center has 2 ligands attached to it, which doesn't necessarily mean that the ligands themselves are bidentate, tridentate, etc. the same goes for...
Re: Cyanide
I was wondering the same thing but Patricia Poths talked about this in her review session on Monday. She said they are the same it is just that IUPAC changed the way that we name some of the ligands recently to ending in ido rather than o. She also said that we can use either on the final and not lo...
- Tue Dec 04, 2018 9:03 pm
- Forum: Acidity & Basicity Constants and The Conjugate Seesaw
- Topic: K
- Replies: 3
- Views: 424
Re: K
K is an equilibrium constant which is a number that expresses the relationship between the amounts of products and reactants present at equilibrium in a reversible chemical reaction at a given temperature. K a values are equilibrium constants that correspond to acids in their reactions with water an...
- Tue Dec 04, 2018 8:57 pm
- Forum: Bronsted Acids & Bases
- Topic: 6th edition J.11
- Replies: 1
- Views: 402
Re: 6th edition J.11
You can determine which is the acid and which is the base from the Bronsted-Lowry definition of a base an acid. According to Bronsted-Lowry, an acid is a species that donates a proton (H+) and since HI has a H in it, it is the only one of those species that can donate an H+ via breaking the H-I bond...
- Tue Dec 04, 2018 8:51 pm
- Forum: Identifying Acidic & Basic Salts
- Topic: Acids and Bases
- Replies: 4
- Views: 782
Re: Acids and Bases
Remember that a salt is a metal bonded to a non-metal and so typically you can figure out what salt is made by combining the metal in the base (like K in KOH) with the nonmetal in the acid (like Cl in HCl). In this example, the salt in the products would be KCl and the OH- and H+ left over combine t...
- Tue Nov 27, 2018 12:33 pm
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: ligands and dentates
- Replies: 3
- Views: 422
Re: ligands and dentates
A ligand is a species that has at least one set of unpaired electrons that it uses to form a coordinate covalent bond with a metal atom. Monodentate means that it has one set of unpaired electrons that it donates to create the bond whereas bidentate means that it has two sets of unpaired electrons t...
- Tue Nov 27, 2018 12:21 pm
- Forum: Naming
- Topic: Naming complexes and coordination compounds
- Replies: 4
- Views: 460
Re: Naming complexes and coordination compounds
There are lots of possible ligands out there so I doubt he expects us to memorize the names for all of them, but it would be helpful for you to know the common ones like amine, chloride (and other halides), aqua, hydroxide, and cyanide.
- Tue Nov 27, 2018 12:11 pm
- Forum: Naming
- Topic: Naming Ligands
- Replies: 2
- Views: 125
Re: Naming Ligands
Ammonia changes to amine when ammonia bonds. It's similar to how non metals have different names when bonded to cations (NaCl is sodium chloride rather than sodium chlorine). We're going to learn more about naming in Wednesday lecture so that should help! We'll also learn more about ligand naming in...
- Tue Nov 27, 2018 12:07 pm
- Forum: Ionic & Covalent Bonds
- Topic: MIDTERM WORKED OUT ANSWERS?
- Replies: 2
- Views: 336
Re: MIDTERM WORKED OUT ANSWERS?
The worked out answers are on the class website, it's on the right side underneath exam information!
- Sun Nov 18, 2018 9:16 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Trigonal Bipyramidal
- Replies: 3
- Views: 420
Re: Trigonal Bipyramidal
The bond angles in trigonal bipyramidal are 180 degrees, 90 degrees, and 120 degrees. The 180 degrees refers to the bond angle between the axial atoms, the 90 degrees refers to the bond angles between an axial atom and an equatorial atom, and the bond angle 120 degrees refers to that between the equ...
- Sun Nov 18, 2018 9:11 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Multiple different bond angles
- Replies: 6
- Views: 904
- Sun Nov 18, 2018 9:10 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Multiple different bond angles
- Replies: 6
- Views: 904
Re: Multiple different bond angles
If you click on this link you can download a software that shows molecules in 3D and you can move them around so you have a better idea of the shapes and angles! If that doesn't help you with remembering molecule bond angles for different shapes then I would just memorize them.
- Sat Nov 10, 2018 10:18 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Bond Angle
- Replies: 2
- Views: 126
Re: Bond Angle
Yep you're right the bond angle would change because the new electrons being introduced to the molecular structure would interest and repel the electrons already there.
- Sat Nov 10, 2018 10:12 pm
- Forum: Dipole Moments
- Topic: Dipole Moments [ENDORSED]
- Replies: 10
- Views: 1008
Re: Dipole Moments [ENDORSED]
I'm pretty sure don't have to calculate them!
- Sun Nov 04, 2018 5:35 pm
- Forum: Quantum Numbers and The H-Atom
- Topic: Number of Electrons based on principle quantum number
- Replies: 2
- Views: 2047
Re: Number of Electrons based on principle quantum number
I believe the maximum number of electrons that would be present if n=2 would be 8 electrons because when n=2, l could be 0 or 1 (s or p), therefore there is an s orbital and a p orbital in the 2nd shell and s can hold 2 electrons and p can hold 6 so 2 + 6 = 8. This would mean that the maximum number...
- Sun Nov 04, 2018 5:26 pm
- Forum: Trends in The Periodic Table
- Topic: Which Ion is larger?
- Replies: 2
- Views: 295
Re: Which Ion is larger?
O^- would still be smaller because S^2- has a third shell which is significantly larger than O's 2nd shell.
- Sun Nov 04, 2018 5:13 pm
- Forum: Lewis Structures
- Topic: Lewis Structure(s)
- Replies: 3
- Views: 233
Re: Lewis Structure(s)
I believe that the exam will specify when to draw resonance structures. When it does ask you to draw resonance structures, it wants you to draw out the different possibilities for electron/atom arrangement where formal charges are closest to zero. This is kind of hard to put in words though so I att...
- Tue Oct 30, 2018 9:44 am
- Forum: Sigma & Pi Bonds
- Topic: What each bond refers to
- Replies: 3
- Views: 402
Re: What each bond refers to
^^ the first image is an example of a pi bond and the second shows some examples of possible sigma bonds
- Tue Oct 30, 2018 9:43 am
- Forum: Sigma & Pi Bonds
- Topic: What each bond refers to
- Replies: 3
- Views: 402
Re: What each bond refers to
I think that Lavelle is going to go over this soon in class so maybe he can make it more clear, but basically a sigma bond is a single bond and pi bonds are any additional bonds between the same two atoms following the single bond. For example a double bond between two carbons would have 1 sigma bon...
- Sun Oct 28, 2018 10:36 pm
- Forum: Ionic & Covalent Bonds
- Topic: Difference Between Ionic and Covalent Bonds
- Replies: 4
- Views: 575
Re: Difference Between Ionic and Covalent Bonds
Ionic bonds are bonds in which one atom (the nonmetal) takes an electron from another atom (the metal) so that both have an octet whereas covalent bonds are where nonmetals share electrons to get to their octets. https://www.google.com/url?sa=i&rct=j&q=&esrc=s&source=images&cd=&a...
- Sun Oct 28, 2018 10:30 pm
- Forum: Trends in The Periodic Table
- Topic: s-block and p-block reactivity?
- Replies: 6
- Views: 1159
Re: s-block and p-block reactivity?
The elements in the s-block are significantly more reactive than those in the p-block and this is because of their very low ionization energies whereas the p-block elements do have high electron affinities but still are not comparable to s-block's ionization energies. To see how reactive group 1 met...
- Sun Oct 28, 2018 10:19 pm
- Forum: Ionic & Covalent Bonds
- Topic: Cation/Anion Size
- Replies: 3
- Views: 698
Re: Cation/Anion Size
Electrons get pulled in when there is a higher positive charge (there are more protons) which is why they get smaller. This explains why cations are smaller than anions. Cations have lost some amount of electrons but still have the same number of protons as the neutral atom (this is why it's still t...
- Sun Oct 28, 2018 10:00 pm
- Forum: Ionic & Covalent Bonds
- Topic: Atomic Radius Decreasing
- Replies: 2
- Views: 228
Re: Atomic Radius Decreasing
Atomic radius decreases across a period because as you go across a period the effective nuclear charge (# protons) increases bringing the electrons closer to the nucleus. While more electrons are also being added, the radius still decreases because additional electrons added across a period are in t...
- Sun Oct 21, 2018 11:36 pm
- Forum: Electron Configurations for Multi-Electron Atoms
- Topic: Nodal planes
- Replies: 1
- Views: 274
Re: Nodal planes
A nodal plane is a part of a wave function where the probability of finding an electron is zero. You can see that p and d orbitals have nodal planes by looking at the shapes of those wave functions and where they narrow. The number of nodal planes is equal to angular momentum quantum number (l).
- Sun Oct 21, 2018 11:22 pm
- Forum: Dipole Moments
- Topic: Requirements
- Replies: 1
- Views: 425
Re: Requirements
The presence of a dipole moment is dependent on electronegativity and the structure of a compound. Personally I like to determine if a dipole moment occurs by drawing the lewis structure of a compound then looking at electronegativity values of the elements in it and comparing them. Lots of times yo...
- Sun Oct 21, 2018 10:56 pm
- Forum: Properties of Light
- Topic: Test 2 Equations
- Replies: 14
- Views: 1090
Re: Test 2 Equations
All of the equations we have gone over in lecture will be given. Here's a link to the equations and constants we're given! https://lavelle.chem.ucla.edu/wp-content/supporting-files/Chem14A/constants_equations.pdf Make sure you know c = \lambda \nu , E = h\nu , \lambda = \frac{h}{mv} , and En = \frac...
- Fri Oct 12, 2018 4:26 pm
- Forum: Photoelectric Effect
- Topic: Photoelectric Effect
- Replies: 2
- Views: 277
Re: Photoelectric Effect
Electrons aren't always ejected. They are only ejected if the light hitting the metal has a sufficient amount of energy (it's wavelength has to be small enough i.e. it has to have high frequency). A really important thing to remember is that the intensity (amplitude) of the light wave does not have ...
- Fri Oct 12, 2018 4:18 pm
- Forum: Accuracy, Precision, Mole, Other Definitions
- Topic: molar masses on the test
- Replies: 7
- Views: 936
Re: molar masses on the test
You'll also find that after doing chemistry for a while you'll just naturally memorize the molar masses of elements and compounds. After doing IB chemistry I had the molar masses of carbon, nitrogen, water, hydrogen, sodium hydroxide, and many more memorized without having to even study them because...
- Fri Oct 12, 2018 4:07 pm
- Forum: DeBroglie Equation
- Topic: Week 3 Textbook Assignment
- Replies: 2
- Views: 131
Re: Week 3 Textbook Assignment
You can choose any 7 you like! It would make the most sense to do quantum mechanics questions though since that is what we are working on right now. Don't worry though you don't have to do questions on things we haven't learned yet.
- Fri Oct 12, 2018 3:58 pm
- Forum: Significant Figures
- Topic: sig fig calculations
- Replies: 3
- Views: 659
Re: sig fig calculations
Yes, there are a variety of sig fig rules for calculations based off of division, addition, subtraction, and even for when you're taking logarithms (I think we'll do that in our acid base unit). If you're answering a question, your sig figs should be based off of your least precise measurement, that...
- Fri Oct 12, 2018 3:39 pm
- Forum: DeBroglie Equation
- Topic: Negative Energy?
- Replies: 2
- Views: 153
Re: Negative Energy?
The energy itself is not negative. The negative sign just tells you that that is lost rather than gained. It's kind of like velocity being directional. Negative velocity doesn't mean that the item moving is moving negatively it just indicates what direction it is moving in. Hopefully that helps :)

