Search found 101 matches
- Sat Mar 14, 2020 8:05 pm
- Forum: First Order Reactions
- Topic: 7B.3 Determining the order of a reaction
- Replies: 2
- Views: 256
Re: 7B.3 Determining the order of a reaction
I thought molecularity and order went together? If not, what is the purpose of molecularity if not to go along with the order of a reaction?
- Sat Mar 14, 2020 8:03 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: Cvm/Cpm
- Replies: 3
- Views: 408
Re: Cvm/Cpm
We use those if we are talking about monoatomic ideal gases. Dr. Lavelle said that we would not be tested on diatomic or linear gases.
- Sat Mar 14, 2020 7:59 pm
- Forum: Arrhenius Equation, Activation Energies, Catalysts
- Topic: HW 7E.5
- Replies: 2
- Views: 238
Re: HW 7E.5
The difference between an intermediate and a catalyst is how they occur in a reaction. An intermediate is formed in one of the steps and used in a later step. As a result, it is a product in one step and then a reactant in a later step. A catalyst is used up in one step and formed in a later step. A...
- Sat Mar 14, 2020 7:55 pm
- Forum: *Enzyme Kinetics
- Topic: 7E.3
- Replies: 1
- Views: 381
Re: 7E.3
The textbook does an example in the reading, and it uses the equation ln(\frac{k2}{k1})=\frac{Ea}{R}(\frac{1}{T1}-\frac{1}{T2}) Ea is the activation energy. Substituting the known values should result in the correct answer for part a. For part b, you use the same equation but with a ...
- Sat Mar 14, 2020 7:54 pm
- Forum: Arrhenius Equation, Activation Energies, Catalysts
- Topic: topic 7E problem 3
- Replies: 1
- Views: 220
Re: topic 7E problem 3
The textbook does an example in the reading, and it uses the equation ln(\frac{k2}{k1})=\frac{Ea}{R}(\frac{1}{T1}-\frac{1}{T2}) Ea is the activation energy. Substituting the known values should result in the correct answer for part a. For part b, you use the same equation but with a ...
- Sat Mar 14, 2020 7:50 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: rate determining step
- Replies: 4
- Views: 447
Re: rate determining step
The slowest step is the rate determining step because the reaction will not proceed faster or slower if you change the speed at which the fast step occurs.
- Sat Mar 14, 2020 1:25 pm
- Forum: First Order Reactions
- Topic: 7B.3 Determining the order of a reaction
- Replies: 2
- Views: 256
7B.3 Determining the order of a reaction
For part c of 7B.3, I know that it says the reaction is first-order, but I thought that if there are two molecules in the reactants, it is a second-order reaction. Is it based on the notation (2A vs A+A) that tells us that it's not a second-order reaction?
- Sun Mar 08, 2020 10:46 pm
- Forum: Ideal Gases
- Topic: When do we change PV=NRT into deltas?
- Replies: 4
- Views: 482
Re: When do we change PV=NRT into deltas?
It essentially depends on the problem. Whichever factor is held constant, you can turn the others into deltas. For example, in an isothermal, irreversible reaction, the temperature stays the same, so you can use  V and
V and  P since they change.
P since they change.
- Sun Mar 08, 2020 10:42 pm
- Forum: Appications of the Nernst Equation (e.g., Concentration Cells, Non-Standard Cell Potentials, Calculating Equilibrium Constants and pH)
- Topic: Purpose of Nernst Equation
- Replies: 5
- Views: 413
Re: Purpose of Nernst Equation
The Nernst Equation is primarilly used to determine the cell potential under non-standard conditions. This is why you use Q combined with the standard cell potential to get the cell potential for any cell you are given, say one that is not at equilibrium.
- Sun Mar 08, 2020 10:40 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Molecularity and rate laws
- Replies: 5
- Views: 429
Re: Molecularity and rate laws
A unimolecular elementary step corresponds to a first order rate law. A bimolecular elementary step corresponds to a second order rate law, and so on and so on. As such, they are directly correlated with each other and can be used interchangeably to describe a reaction. Is there a good reason why w...
- Sun Mar 08, 2020 10:38 pm
- Forum: General Rate Laws
- Topic: 7C.11
- Replies: 1
- Views: 215
Re: 7C.11
The rate constant is affected by changes in temperature, but not changes in reactant concentrations.
- Sun Mar 08, 2020 10:36 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Rate determining step
- Replies: 2
- Views: 243
Re: Rate determining step
The rate determining step is the slowest elementary step in the reaction. We will either be given this value or instructed to calculate this using rate laws.
- Sun Mar 08, 2020 10:35 pm
- Forum: Interesting Applications: Rechargeable Batteries (Cell Phones, Notebooks, Cars), Fuel Cells (Space Shuttle), Photovoltaic Cells (Solar Panels), Electrolysis, Rust
- Topic: Test 2
- Replies: 4
- Views: 457
Re: Test 2
I'd assume it will be this week since it usually takes them about a week to grade. I'm sure Dr. Lavelle will announce it either tomorrow or on Wednesday.
- Sun Mar 08, 2020 10:07 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Molecularity and rate laws
- Replies: 5
- Views: 429
Re: Molecularity and rate laws
A unimolecular elementary step corresponds to a first order rate law. A bimolecular elementary step corresponds to a second order rate law, and so on and so on. As such, they are directly correlated with each other and can be used interchangeably to describe a reaction.
- Sun Mar 08, 2020 10:06 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Cell Diagrams
- Replies: 6
- Views: 435
Re: Cell Diagrams
When the states are the same, you use commas to separate them, whereas when they're different phases, use the vertical line to separate them.
- Sun Mar 08, 2020 10:03 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Termolecular Molecularity
- Replies: 5
- Views: 484
Re: Termolecular Molecularity
A termolecular elementary step is any combination of three molecules colliding. This is very unlikely, and termolecular reactions are rare.
- Sun Mar 08, 2020 10:01 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Bimolecular Molecularity
- Replies: 2
- Views: 243
Re: Bimolecular Molecularity
A bimolecular reaction is any reaction with two molecules involved. The two molecules can be the same or they can be different.
- Sun Mar 08, 2020 9:57 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Reaction Mechanism
- Replies: 1
- Views: 181
Re: Reaction Mechanism
It is the number of steps to form the products. In other words, reactions can be composed of multiple steps, and proposed reaction mechanisms are possible explanations for how these reactions occur. A reaction mechanism is the set of elementary steps as a whole. In this class, we are asked to determ...
- Sun Mar 08, 2020 9:55 pm
- Forum: Administrative Questions and Class Announcements
- Topic: 100 or 50 posts?
- Replies: 8
- Views: 622
Re: 100 or 50 posts?
I believe having 100 posts is acceptable. In fact, it is probably better to leave them up so future chemistry students can use them for reference.
- Sun Mar 08, 2020 9:54 pm
- Forum: Zero Order Reactions
- Topic: Identifying Zero vs. First vs. Second Order Reactions
- Replies: 7
- Views: 567
Re: Identifying Zero vs. First vs. Second Order Reactions
If given their graphs, you can see which graph provides a straight line based on the axes (ln [A] vs 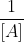 vs [A]). If given their rate laws or chemical equations, you can see how many molecules are involved in each reaction (molecularity).
vs [A]). If given their rate laws or chemical equations, you can see how many molecules are involved in each reaction (molecularity).
- Sun Mar 08, 2020 9:52 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Molecularity
- Replies: 3
- Views: 243
Re: Molecularity
It is the number of species involved in an elementary step. In other words, it is how many molecules are required to collide with each other in order for the reaction to proceed.
- Sun Mar 08, 2020 9:50 pm
- Forum: First Order Reactions
- Topic: first order reaction vs second order reaction
- Replies: 2
- Views: 239
Re: first order reaction vs second order reaction
If you have a graph, you can differentiate based on what graph shows a straight line (ln [A] vs 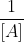 ). If you have the rate laws or chemical equations, you can determine which order the reactions are based on the numbers and ratios of the reactants.
). If you have the rate laws or chemical equations, you can determine which order the reactions are based on the numbers and ratios of the reactants.
- Sun Mar 08, 2020 9:46 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Final Exam Material Distribution
- Replies: 6
- Views: 424
Re: Final Exam Material Distribution
I'd assume it's going to be evenly distributed, but we will have to wait and see until Dr. Lavelle provides additional info closer to the exam date.
- Sun Mar 08, 2020 9:35 pm
- Forum: General Science Questions
- Topic: Slope
- Replies: 4
- Views: 404
Re: Slope
Depending on what order your reaction is, your rate law will be different. For zero-order and first-order reactions, the slope is equal to -k, but for second-order reactions, the slope is just k. The trick is just distinguishing which value is k when using a rate law. How can you tell the differenc...
- Sun Mar 08, 2020 9:32 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: 7.23b
- Replies: 1
- Views: 175
Re: 7.23b
H20 is not included in the reaction since it is a liquid, and liquids are not included in equilibrium constant equations. The rate of the forward reaction would be rate = k1[ClO-] , and the rate of the reverse reaction would be rate = k1'[HClO][OH-] . Since the rate of the forward reaction is eq...
- Sun Mar 08, 2020 9:17 pm
- Forum: General Science Questions
- Topic: Slope
- Replies: 4
- Views: 404
Re: Slope
Depending on what order your reaction is, your rate law will be different. For zero-order and first-order reactions, the slope is equal to -k, but for second-order reactions, the slope is just k. The trick is just distinguishing which value is k when using a rate law.
- Sun Feb 09, 2020 10:24 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: Specific Heat Capacity
- Replies: 5
- Views: 187
Re: Specific Heat Capacity
Specific heat capacity is used with the mass of a substance to determine the q of a reaction. Molar heat capacity is used with moles, and heat capacity is the amount of heat required to raise the temperature of an object by 1 degree Celsius. To calculate the specific/molar heat capacity, you divide ...
- Sun Feb 09, 2020 10:22 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: m and n in heat capacity
- Replies: 4
- Views: 198
Re: m and n in heat capacity
Specific heat capacity and molar heat capacity are calculated by dividing the heat capacity (heat required to raise temperature by 1 degree Celsius) by the mass or moles of substance present. As such, it will not matter which one you use as long as you use the correct specific/molar heat capacity.
- Sun Feb 09, 2020 10:19 pm
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: Midterm Material
- Replies: 10
- Views: 541
Re: Midterm Material
Based on what he said in class and Outline 4, the midterm should cover everything that we have learned in class so far (up to last Fri 2/7).
- Sun Feb 09, 2020 9:49 pm
- Forum: Calculating Work of Expansion
- Topic: 4A.3
- Replies: 4
- Views: 190
Re: 4A.3
Adding on to the previous replies, the equation 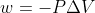 outputs an answer with units m^3
outputs an answer with units m^3  atm. To convert this answer to J, you need to convert to m^3
atm. To convert this answer to J, you need to convert to m^3  Pa, which would involve multiplying your original answer by 101.325.
Pa, which would involve multiplying your original answer by 101.325.
- Sun Feb 09, 2020 9:44 pm
- Forum: Concepts & Calculations Using First Law of Thermodynamics
- Topic: 4A.3
- Replies: 2
- Views: 153
Re: 4A.3
The change in internal energy ( \Delta U ) is equal to the sum of heat (q) and work (w). In this problem, there is no heat involved (that we know of), so we can assume that the change in internal energy is equal to the work done on the system, which is 28 J. The answer in the back of the book says 8...
- Sun Feb 09, 2020 9:37 pm
- Forum: Non-Equilibrium Conditions & The Reaction Quotient
- Topic: Ka Kb
- Replies: 11
- Views: 690
Re: Ka Kb
If a weak acid is dissociating in solution, then you use Ka, which is the K value for a reaction in which an acid gives off a proton and adopts its conjugate base form. Kb is the opposite in that it is the K value for a reaction in which a base accepts a proton and becomes its conjugate acid, produc...
- Sun Feb 09, 2020 9:19 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: Calorimeters
- Replies: 17
- Views: 988
Re: Calorimeters
Is there a difference between a regular calorimeter and a bomb calorimeter? Yes, if you're talking about the simple calorimeters that are used in high school labs, which basically consist of thermometers suspended in a container filled with water. The difference between these calorimeters and bomb ...
- Sat Feb 08, 2020 5:50 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: Calorimeters
- Replies: 17
- Views: 988
Re: Calorimeters
Is there a difference between a regular calorimeter and a bomb calorimeter? Yes, if you're talking about the simple calorimeters that are used in high school labs, which basically consist of thermometers suspended in a container filled with water. The difference between these calorimeters and bomb ...
- Thu Feb 06, 2020 9:27 pm
- Forum: Entropy Changes Due to Changes in Volume and Temperature
- Topic: 4F.5
- Replies: 1
- Views: 51
Re: 4F.5
You can use the equation ) . If V2 > V1,
. If V2 > V1, 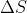 will be positive since gases will not spontaneously occupy a space with a smaller volume. Energy is required for that to occur.
will be positive since gases will not spontaneously occupy a space with a smaller volume. Energy is required for that to occur.
- Thu Feb 06, 2020 9:24 pm
- Forum: Third Law of Thermodynamics (For a Unique Ground State (W=1): S -> 0 as T -> 0) and Calculations Using Boltzmann Equation for Entropy
- Topic: Degeneracy
- Replies: 1
- Views: 66
Re: Degeneracy
There may be a small, simple question since Dr. Lavelle had a section in the notes dedicated to calculating W, but I wouldn't imagine it being very complex if it is on the midterm.
- Thu Feb 06, 2020 9:17 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: Calorimeters
- Replies: 17
- Views: 988
Re: Calorimeters
A bomb calorimeter maintains a constant V. Since V is constant, any increase in gas molecules would result in an increase in pressure since there would be more particles in the same V.
- Thu Feb 06, 2020 9:14 pm
- Forum: Concepts & Calculations Using Second Law of Thermodynamics
- Topic: Reversible vs. Irreversible Reactions
- Replies: 3
- Views: 220
Re: Reversible vs. Irreversible Reactions
Thanks for the replies. However, there is still one aspect that I am a little confused about. How does work tie in with these reactions? Why do reversible reactions have perform a greater amount of work? I can understand this based on the area under the curves, but can someone explain this conceptua...
- Thu Feb 06, 2020 9:10 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Midterm
- Replies: 7
- Views: 274
Re: Midterm
To expand on the previous response, Dr. Lavelle seems to be devoting lots of time to showing the related nature of the equations during the derivation process. This seems like prime material for a conceptual type question, so it may be prudent to understand the relation of the equations. Yes, there...
- Thu Feb 06, 2020 9:09 pm
- Forum: Entropy Changes Due to Changes in Volume and Temperature
- Topic: Keeping equations straight
- Replies: 2
- Views: 94
Re: Keeping equations straight
A lot of the equations tie in with each other and are used in junction to solve for certain values. I have found it helps to think of the P and V when reading a problem. Visualizing the system and asking yourself if the pressure or volume is changing is a key step to deciding which equation to use s...
- Wed Feb 05, 2020 9:03 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: Cp and Cv
- Replies: 10
- Views: 481
Re: Cp and Cv
On Dr. Lavelle's website, there equation sheet lists a few Cp values, and I highly doubt we will be required to memorize anything related to specific values.
- Wed Feb 05, 2020 9:01 pm
- Forum: Concepts & Calculations Using Second Law of Thermodynamics
- Topic: Reversible vs. Irreversible Reactions
- Replies: 3
- Views: 220
Reversible vs. Irreversible Reactions
I can understand the logic behind the varying amounts of work depending on what type of reaction is being discussed, but I'm still having trouble understanding the conceptual aspects of these terms. What separates reversible reactions from irreversible reactions?
- Wed Feb 05, 2020 8:57 pm
- Forum: Calculating Work of Expansion
- Topic: units
- Replies: 9
- Views: 237
Re: units
w is usually given as joules (J) or kilojoules (kJ). However, depending on the problem, you may need to convert from pressure/volume units such as Pa 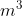 and L atm.
and L atm.
- Wed Feb 05, 2020 8:55 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Midterm
- Replies: 7
- Views: 274
Re: Midterm
My guess is they will announce it within the next 5 days, given that the review sessions have started to begin.
- Wed Feb 05, 2020 8:54 pm
- Forum: Third Law of Thermodynamics (For a Unique Ground State (W=1): S -> 0 as T -> 0) and Calculations Using Boltzmann Equation for Entropy
- Topic: Boltzmann Equation Notes
- Replies: 2
- Views: 160
Re: Boltzmann Equation Notes
Why did he write statistical? How does it relate to error in W?
- Wed Feb 05, 2020 8:50 pm
- Forum: Third Law of Thermodynamics (For a Unique Ground State (W=1): S -> 0 as T -> 0) and Calculations Using Boltzmann Equation for Entropy
- Topic: Microstates
- Replies: 7
- Views: 215
Re: Microstates
Adding on to the previous reply, you would use degeneracy to calculate a specific value of entropy using the Boltzman Equation, S = kBln W
- Wed Feb 05, 2020 8:47 pm
- Forum: Entropy Changes Due to Changes in Volume and Temperature
- Topic: Increasing entropy
- Replies: 1
- Views: 89
Re: Increasing entropy
Yes. This is because gas atoms will not occupy a volume that is smaller than their current space. To do so, another factor would be involved such as changes in temperature. It does not spontaneously shrink in volume.
- Wed Feb 05, 2020 8:45 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: 4D.3
- Replies: 1
- Views: 114
Re: 4D.3
Since the problem gives you the specific heat capacity of the calorimeter, you can calculate the q of the reaction by calculating mc \Delta T of the calorimeter, which equals the q of the reaction. Make sure to be mindful of the sign. Since you're calculating the q of the calorimeter, the q of the r...
- Wed Feb 05, 2020 8:43 pm
- Forum: General Science Questions
- Topic: Class before Midterm
- Replies: 2
- Views: 149
Re: Class before Midterm
Based on the structure of the class last quarter, I'd guess it'll be consist of review questions and topics.
- Thu Jan 30, 2020 10:10 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: Homework Problem 4A.3
- Replies: 1
- Views: 111
Homework Problem 4A.3
I'm a little confused about the units used to calculate the work done in compressing a piston. Is it atm and m? I am getting the correct number as the final answer, but the decimal place is different.
- Thu Jan 23, 2020 7:22 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: 6B.9
- Replies: 5
- Views: 171
Re: 6B.9
Alexis Webb 2B wrote:I think they multiplied the [OH-] over and then divided by Kw.
Is there a reason why this is done? Why do we reverse the fraction in this case?
- Sat Dec 07, 2019 7:41 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: Test 2, Question 7A
- Replies: 3
- Views: 301
Re: Test 2, Question 7A
The H bonded to the C atom on the right side of the molecule does not count as a hydrogen bonding site since the electronegativity difference between C and H is not enough for there to be a strong partial positive charge on the hydrogen. H bonding can only occur with F, O, or N atoms.
- Sat Dec 07, 2019 7:38 pm
- Forum: Lewis Structures
- Topic: Lewis for carbon monoxide
- Replies: 3
- Views: 1262
Re: Lewis for carbon monoxide
C and O have a combined total of 10 valence electrons available for bonding. This means that there must be a triple bond with two lone pairs on each atom to satisfy the octet rule, with each atom having 8 electrons. Having only a double bond would means there wouldn't be enough electrons to satisfy ...
- Sat Dec 07, 2019 7:34 pm
- Forum: Calculating the pH of Salt Solutions
- Topic: HW 6D.11
- Replies: 1
- Views: 218
Re: HW 6D.11
It seems that the Al3+ hybridizes its s, p, and d orbitals to form 6 hybridized orbitals. The reason why it's 6 is because that's the maximum number of H2O molecules that can fit around a Al3+ ion, and by having the maximum number of bonds, it is the most stable. Here is a good resource if you want ...
- Sat Dec 07, 2019 7:20 pm
- Forum: Naming
- Topic: -ate, -ide, -ite, -o
- Replies: 3
- Views: 305
Re: -ate, -ide, -ite, -o
According to the textbook, for any anion inside the [ ] (so any anion directly bonded to the central metal cation), the ending should be an -ato or an -ido or an -ito. For example, the Chloride anion is written as chlorido. The -ido is part of a newer change, but both the -ido and the -o suffixes a...
- Sat Dec 07, 2019 7:19 pm
- Forum: Properties & Structures of Inorganic & Organic Acids
- Topic: Strong vs. Weak Acids
- Replies: 3
- Views: 302
Re: Strong vs. Weak Acids
The structure of oxyacids involves the H atom bonding to the O atom and then to the Cl, I, or whichever atom is remaining. Since the H is not directly bonded to the Cl or I, the bond length is not relevant. Instead, the electronegativity is more important. This is because the Cl or I atom pulls elec...
- Sat Dec 07, 2019 7:13 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: Test 2, Question 7A
- Replies: 3
- Views: 301
Re: Test 2, Question 7A
Yes, you are correct. Each O atom counts as two H bonding sites due to the two lone electron pairs.
- Sat Dec 07, 2019 7:09 pm
- Forum: Naming
- Topic: -ate, -ide, -ite, -o
- Replies: 3
- Views: 305
Re: -ate, -ide, -ite, -o
The -ate suffix is used for the central metal cation is part a coordination compound that has an overall negative charge (K4[Fe(CN)6] is potassium hexacyanoferrate(II)). The -ide suffix is used for an anion that bonds to the coordination compound (anion that comes after the brackets). The -ite suffi...
- Sat Dec 07, 2019 1:32 am
- Forum: Naming
- Topic: Easy Way to Name/Remember
- Replies: 2
- Views: 207
Re: Easy Way to Name/Remember
Name the ligands and then the central metal cation in one word. The ligands should be in alphabetical order, ignoring the prefixes, and the central metal cation should be last with its oxidation state. If there is a cation in front of the brackets that bonds to the coordination compound, name the el...
- Sat Dec 07, 2019 1:28 am
- Forum: Properties & Structures of Inorganic & Organic Acids
- Topic: Identifying Acids and Bases
- Replies: 2
- Views: 308
Re: Identifying Acids and Bases
Yes, if a molecule has a lone pair, it could be used to bond with H+. By donating an electron to the H+, it acts as a Lewis base. For charges, we know that positive transition metal cations act as acids by weakening the bond between H and O in H2O molecules. We also know that negatively charged anio...
- Sat Dec 07, 2019 1:24 am
- Forum: Conjugate Acids & Bases
- Topic: Conjugate base of strong and weak acids
- Replies: 1
- Views: 516
Re: Conjugate base of strong and weak acids
If we think of it in terms of a reaction and using a chemical equation, it is easier to understand the logic behind this. The dissociation of an acid or base is a reversible reaction. This means that at equilibrium, both the forward and reverse reactions are occurring simultaneously. Let's say that ...
- Sat Dec 07, 2019 1:19 am
- Forum: Identifying Acidic & Basic Salts
- Topic: Weak acid and weak base
- Replies: 1
- Views: 149
Re: Weak acid and weak base
Since the weak acid and base both do not dissociate very much, the solution will contain mostly the weak acid, weak base, and very small amounts of the salt and its ions. A salt is formed, but the pH is determined by the weak acid and weak base, not the salt.
- Sat Dec 07, 2019 1:12 am
- Forum: Conjugate Acids & Bases
- Topic: 6A.4
- Replies: 1
- Views: 549
Re: 6A.4
Urea is actually a base, and it reacts with H2O to produce NH3 and CO2. The two H+ come from the H2O and bond to the NH2 groups, and the 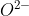 bonds to the remaining C=O atoms.
bonds to the remaining C=O atoms.
- Sat Dec 07, 2019 1:06 am
- Forum: Calculating pH or pOH for Strong & Weak Acids & Bases
- Topic: 6D.11 part d
- Replies: 1
- Views: 108
Re: 6D.11 part d
In both parts, the K+ cation is a Group 1 element, so we know that it does not affect the pH. The difference is in the anion. The F- anion is the conjugate base of a weak acid (HF), so that means it will lower the pH by acting as a base. For HBr, the Br- anion is the conjugate base of a strong acid ...
- Sat Dec 07, 2019 1:03 am
- Forum: Acidity & Basicity Constants and The Conjugate Seesaw
- Topic: 6C21
- Replies: 2
- Views: 280
Re: 6C21
I don't see any H2O components in 6C21. Are you sure you are referring to the correct question? Or do you mean 6D11?
- Sat Dec 07, 2019 1:00 am
- Forum: Lewis Acids & Bases
- Topic: Memorizing Charges of Transition Metals
- Replies: 9
- Views: 832
Re: Memorizing Charges of Transition Metals
Can't the same metal have different oxidation states, though? Like Fe can be +2 or +3 Yes, but to figure out which oxidation state the metal has, we use the ligands to figure out the charge that we must balance out. As such, memorizing the ligands essentially works towards knowing the oxidation sta...
- Sat Dec 07, 2019 12:58 am
- Forum: Acidity & Basicity Constants and The Conjugate Seesaw
- Topic: Ka and Kb
- Replies: 3
- Views: 381
Re: Ka and Kb
No, I don't think the book goes into detail about bases that accept multiple protons, but it should be the same as polyprotic acids, only in reverse order. The same trends apply.
- Sat Dec 07, 2019 12:57 am
- Forum: Amphoteric Compounds
- Topic: how to identify?
- Replies: 5
- Views: 402
Re: how to identify?
Yes, amphiprotic compounds fall in the category of amphoteric compounds. All amphiprotic compounds (I don't know about exceptions) are amphoteric, but not all amphoteric compounds are amphiprotic.
- Sat Dec 07, 2019 12:55 am
- Forum: Calculating the pH of Salt Solutions
- Topic: 6D11 E and F
- Replies: 1
- Views: 208
Re: 6D11 E and F
When the salts are in an aqueous solution, the cation and anion will react differently. In the case of AlCl3, the small, highly positive Al3+ cation will interact with the water molecules and will cause the water molecules to give up a proton, acting as an acid. The Cl- anion is the conjugate base o...
- Sat Dec 07, 2019 12:47 am
- Forum: Calculating pH or pOH for Strong & Weak Acids & Bases
- Topic: HW 6C.17.
- Replies: 1
- Views: 180
Re: HW 6C.17.
The presence of the nitrogen atom usually denotes a weaker base. Furthermore, HBrO is a weak acid, so its conjugate base (BrO-) will be a relatively strong base.
- Sat Dec 07, 2019 12:45 am
- Forum: Naming
- Topic: Naming Anionic Ligands
- Replies: 3
- Views: 249
Re: Naming Anionic Ligands
I think both are fine, it's just that -ido is part of a newer change, but the old system remains valid still.
- Sat Dec 07, 2019 12:43 am
- Forum: Properties & Structures of Inorganic & Organic Acids
- Topic: Acid Strength- Acetic vs Formic Acid
- Replies: 4
- Views: 532
Re: Acid Strength- Acetic vs Formic Acid
The acetic acid has a methyl group attached (CH3), which is an electron-donating compound. As such, the anionic form of the acetic acid is less stable since the CH3 does not delocalize the negative charge and instead adds to it. Meanwhile, the H atom attached to the formic acid is electron-withdrawi...
- Wed Dec 04, 2019 10:59 pm
- Forum: Lewis Acids & Bases
- Topic: Characteristics of Lewis acids
- Replies: 2
- Views: 203
Re: Characteristics of Lewis acids
What differentiates Bronsted acids from Lewis acids are the definitions of each. However, they are both acids, so they would still share the same properties that acids have, though their strengths may vary.
- Wed Dec 04, 2019 10:51 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: Hydrogen bonding
- Replies: 1
- Views: 255
Re: Hydrogen bonding
A hydrogen bonding site is where there are any lone electron pairs that can experience electrostatic interactions with an atom from another molecule. This is why the O atom in water can form 2 hydrogen bonds with two other hydrogen atoms. For hydrogen bonding to occur, there only needs to be a F, O,...
- Wed Dec 04, 2019 10:40 pm
- Forum: Acidity & Basicity Constants and The Conjugate Seesaw
- Topic: Ka and Kb
- Replies: 3
- Views: 381
Re: Ka and Kb
Yes I would assume so since polyprotic bases and Kb values act similar to acids other than accepting protons. Although it is in reverse, polyprotic bases would accept protons less and less readily the more protons they gain.
- Wed Dec 04, 2019 10:33 pm
- Forum: Properties & Structures of Inorganic & Organic Acids
- Topic: Resonance and Acid strength
- Replies: 1
- Views: 202
Re: Resonance and Acid strength
Resonance stabilizes the products of acid dissociation reactions by delocalizing the negative charge so it is spread out more evenly among all the atoms. Since the product is more stable, the equilibrium is shifted to the right, leading to a stronger acid since it will more readily dissociate.
- Wed Dec 04, 2019 10:31 pm
- Forum: Lewis Acids & Bases
- Topic: Memorizing Charges of Transition Metals
- Replies: 9
- Views: 832
Re: Memorizing Charges of Transition Metals
Can't the same metal have different oxidation states, though? Like Fe can be +2 or +3 Yes, but to figure out which oxidation state the metal has, we use the ligands to figure out the charge that we must balance out. As such, memorizing the ligands essentially works towards knowing the oxidation sta...
- Wed Dec 04, 2019 10:27 pm
- Forum: Identifying Acidic & Basic Salts
- Topic: amphoteric versus amphiprotic
- Replies: 1
- Views: 162
Re: amphoteric versus amphiprotic
Amphiprotic substances can donate or accept H+. This means they are also amphoteric since they can act as either an acid or a base. However, amphoteric substances make up a broader category than amphiprotic substances. This is because there are different definitions of acids and bases (Lewis, Bronst...
- Wed Dec 04, 2019 10:22 pm
- Forum: Naming
- Topic: Cation Order
- Replies: 1
- Views: 164
Re: Cation Order
I believe writing the cations and anions in the order they are named/written is fine. So pentaamminechloroplatinum(IV) bromide would be [Pt(NH3)5Cl]Br3. Alphabetical order is very important when naming the compounds. Otherwise, the order is not essential as long as the general arrangement is correct.
Re: Ligands
Yes, I believe my TA mentioned something about naming ligands likely being on the final, so it would be best to memorize the chart.
- Wed Dec 04, 2019 10:13 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: How do you tell if parts of a molecule are in the same plane?
- Replies: 2
- Views: 369
Re: How do you tell if parts of a molecule are in the same plane?
It mostly depends on the shape. I found looking at Google images of 3D representations of VSEPR theory shapes helped a lot. https://tse4.mm.bing.net/th?id=OIP.EZWgs8att7jBKyzxvarQ_gHaEC&pid=Api For example, in a seesaw-shaped molecule, there are two different planes that the atoms are in. The tw...
- Tue Dec 03, 2019 1:16 am
- Forum: Bronsted Acids & Bases
- Topic: Relative Acid Strength
- Replies: 1
- Views: 195
Re: Relative Acid Strength
The methyl group is electron releasing group, meaning that it is more likely to donate an electron. This is because the carbon is slightly more electronegative than the hydrogen, and its partial negative charge repulses its remaining electron. Since it is more likely to donate an electron compared t...
- Tue Dec 03, 2019 1:01 am
- Forum: Lewis Acids & Bases
- Topic: Memorizing Charges of Transition Metals
- Replies: 9
- Views: 832
Re: Memorizing Charges of Transition Metals
Yes, I think it is best to memorize the charges of the transition metals, especially since nitrite is relatively simple to memorize.
- Tue Dec 03, 2019 12:58 am
- Forum: Naming
- Topic: Mono for Ligands with only one
- Replies: 1
- Views: 119
Re: Mono for Ligands with only one
I don't think it is necessary, seeing as how the homework and lecture omit the mono prefix. It seems like it would be too redundant especially with the length of some ligand names.
- Mon Dec 02, 2019 11:29 pm
- Forum: Amphoteric Compounds
- Topic: how to identify?
- Replies: 5
- Views: 402
Re: how to identify?
Compounds containing multiple hydrogen atoms are amphoteric since they can donate and accept hydrogens. For compounds that don't contain multiple hydrogens, we know that certain elements form amphoteric compounds. These elements include copper, zinc, tin, lead, aluminium, and beryllium, and they for...
- Mon Dec 02, 2019 11:19 pm
- Forum: Naming
- Topic: Table 9C.1
- Replies: 6
- Views: 375
Re: Table 9C.1
I think to be safe, I would memorize them, especially since they are used in the homework problems. However, I would ask a TA or professor about this.
- Mon Nov 18, 2019 10:03 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Exact Bond Angles for Test 1
- Replies: 3
- Views: 163
Exact Bond Angles for Test 1
Do we need to know the exact bond angles for molecular shapes that have lone electron pairs or can we just say slightly less than 109.5 degrees for AX3E and AX2E2 (NH3 and H2O)?
- Sun Oct 20, 2019 10:56 pm
- Forum: Electron Configurations for Multi-Electron Atoms
- Topic: Sequence of Orbitals in a Singl
- Replies: 3
- Views: 242
Re: Sequence of Orbitals in a Singl
I believe that Dr. Lavelle said that it is random/arbitrary since px, py, and pz can all be generalized to the p subshell.
- Sun Oct 20, 2019 10:53 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: Nodal Planes
- Replies: 3
- Views: 184
Re: Nodal Planes
Nodal planes can tell us the properties of atoms in how they can interact with each other. Since atoms can form bonds with each other through their electrons, nodal planes are important since they are essentially regions where no electrons can be found. As such, this is related to orbitals since som...
- Sun Oct 20, 2019 10:49 pm
- Forum: *Shrodinger Equation
- Topic: Schrodinger's Equation
- Replies: 2
- Views: 215
- Sun Oct 20, 2019 10:45 pm
- Forum: Properties of Electrons
- Topic: What's the difference between a shell, a subshell, an energy level, and an orbital?
- Replies: 8
- Views: 617
Re: What's the difference between a shell, a subshell, an energy level, and an orbital?
The three quantum numbers (n, l, and ml) describe an orbital, which is the math function used to describe an electron's probable position. The principle quantum number (n) refers to the overall energy and size of the orbital. An energy shell and energy level are interchangeable terms. The angular mo...
- Sun Oct 20, 2019 10:32 pm
- Forum: Electron Configurations for Multi-Electron Atoms
- Topic: Electron Spin
- Replies: 2
- Views: 111
Re: Electron Spin
The arrows are different directions to indicate the opposite electron spins. Although the electrons aren't actually spinning up and down, we can indicate their spins with up and down arrows. As to why we write the up arrow before the down arrow, I believe it is arbitrary seeing as how the direction ...
- Sun Oct 13, 2019 2:07 pm
- Forum: Properties of Light
- Topic: Problem 1.31
- Replies: 1
- Views: 152
Re: Problem 1.31
To solve this, we must calculate the energy of the photons of each laser. First, we start with the high-intensity red ruby laser and calculate the frequency. c = \lambda v 3.00 \times 10^{8} m/s = (694 \times 10^{-9} m)(v) v = 4.32 \times 10^{14} Hz Then, we substitute the frequency ...
- Sun Oct 13, 2019 1:32 pm
- Forum: Bohr Frequency Condition, H-Atom , Atomic Spectroscopy
- Topic: Self-test 1B.4B
- Replies: 2
- Views: 130
Re: Self-test 1B.4B
For this, you would use the DeBroglie Equation to calculate the wavelength of a moving object with momentum.
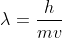
(2 \times 331 m/s)})
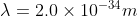
- Sat Oct 12, 2019 7:54 pm
- Forum: Properties of Light
- Topic: Equations We’ve Learned So Far
- Replies: 11
- Views: 687
Re: Equations We’ve Learned So Far
Is there a specific example or equation you're having trouble with? I'm not sure if I can describe all of them.
- Sat Oct 12, 2019 7:51 pm
- Forum: Properties of Light
- Topic: Absorption Spectra
- Replies: 1
- Views: 228
Re: Absorption Spectra
In absorption spectroscopy, light excites the electrons of a gas, causing them to jump to higher energy levels. The electrons are not as stable at higher energy levels, so they drop back down to their original energy level, releasing the energy in the form of light. Depending on the element, the abs...
- Sun Oct 06, 2019 10:03 pm
- Forum: Ionic & Covalent Bonds
- Topic: Ionic vs Covalent
- Replies: 32
- Views: 43735
Re: Ionic vs Covalent
covalent network are the strongest (ex. diamond and graphite) are stronger than ionic however covalent bonds themselves are usually weaker than ionic bonds. How much of knowledge like this will be required for the final in this class? Right now, we haven't gotten to covalent networks, but in genera...
- Fri Oct 04, 2019 12:26 pm
- Forum: Properties of Light
- Topic: Amplitude? [ENDORSED]
- Replies: 8
- Views: 550
Re: Amplitude? [ENDORSED]
Does that mean that a higher amplitude corresponds to a higher intensity of light?
- Fri Oct 04, 2019 12:24 pm
- Forum: Properties of Light
- Topic: CHEM 1A
- Replies: 2
- Views: 154
Re: CHEM 1A
When electrons transition from higher energy levels to lower energy levels, they go from a state of higher energy to a state of lower energy. During this process, energy is released in the form of light.
- Wed Oct 02, 2019 12:16 am
- Forum: Empirical & Molecular Formulas
- Topic: Clarification on the Vitamin C Example
- Replies: 6
- Views: 261
Re: Clarification on the Vitamin C Example
Elaborating on the rounding aspect, all the numbers provided have three significant figures. Since one of the numbers is less than 1, it makes sense for there to be some small mass lost in the calculations as the other numbers would be rounded.


