Search found 104 matches
- Sat Mar 14, 2020 4:55 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Grading Scale
- Replies: 16
- Views: 1123
Re: Grading Scale
It seems the same as 14A, and I think the only thing that might change is how he adjusts the grades based on the class's performance this quarter.
- Thu Mar 12, 2020 8:24 am
- Forum: Kinetics vs. Thermodynamics Controlling a Reaction
- Topic: final
- Replies: 3
- Views: 357
Re: final
I have heard his final covers pretty much all the topics in equal amounts, so I would expect about 1-2 questions on kinetics. Hope this helps!
- Thu Mar 12, 2020 8:23 am
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Endo VS Exo
- Replies: 4
- Views: 436
Re: Endo VS Exo
You have to look at the activation energies (not the rate constants). If the fwd reaction has a lower activation energy than the rev reaction, you know that there is a smaller "hump" going forward and a larger one going backward. If you draw the reaction profile, this corresponds to an exo...
- Mon Mar 09, 2020 11:21 am
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Rate laws for reaction mechanisms
- Replies: 3
- Views: 311
Re: Rate laws for reaction mechanisms
If you read the section above Example 7C.1, the book talks about steady-state approximation, which Professor Lavelle said we would not be doing. I think we will not be expected to know this, but I would just confirm after class or during office hours in order to make sure. If you do so, would it be ...
- Mon Mar 09, 2020 11:17 am
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Calculating K
- Replies: 6
- Views: 433
Re: Calculating K
K is actually measured by the activities of all the reactants, which have no units. Using concentrations is technically an approximation technique, and in order to account for this, we remove the units. This was covered in one of the equilibrium video lectures on his website. Hope this helps!
- Mon Mar 09, 2020 11:15 am
- Forum: Thermodynamic Definitions (isochoric/isometric, isothermal, isobaric)
- Topic: Isothermal Irreversible Free Expansion
- Replies: 3
- Views: 2512
Re: Isothermal Irreversible Free Expansion
Isothermal means a constant temperature, so we can use the formula ΔS = nR ln(V 2 /V 1 ). Since entropy is a state function, only the initial and final states matter, not the pathway taken to get there. This means that for entropy change of the system, there is no difference between reversible and i...
- Mon Mar 09, 2020 11:07 am
- Forum: Student Social/Study Group
- Topic: Studying for Final
- Replies: 7
- Views: 765
Re: Studying for Final
Going over the outlines that Professor Lavelle posts on his website helped me last quarter as well. Lyndon's review session and doing the end of chapter textbook problems were also nice, as some of those problems were very similar to/exactly the same as what was on the actual final.
- Mon Mar 09, 2020 10:59 am
- Forum: First Order Reactions
- Topic: Pseudo-First Order Reaction
- Replies: 4
- Views: 380
Re: Pseudo-First Order Reaction
You can assume/make it a pseudo-first order reaction by making the initial concentration of one reactant extremely large. When we do this, we can essentially say that the change in reactant concentration during the reaction is so tiny that it is insignificant, i.e. that reaction concentration remain...
- Sat Mar 07, 2020 2:32 pm
- Forum: Arrhenius Equation, Activation Energies, Catalysts
- Topic: Elementary reactions
- Replies: 5
- Views: 481
Re: Elementary reactions
Adding on to what @Amanda Lin 2I said, an overall reaction can be a sum of several elementary steps, which is helpful for kinetic analysis. Hope this helps!
- Sat Mar 07, 2020 2:08 pm
- Forum: Method of Initial Rates (To Determine n and k)
- Topic: Rate of reaction
- Replies: 3
- Views: 343
Re: Rate of reaction
As @705302428 mentioned, you would only see species in the denominator if they have a negative order. Professor Lavelle didn't cover this in lecture, so I don't think we'll be expected to know this. Hope this helps!
- Sat Mar 07, 2020 2:06 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Molecularity
- Replies: 4
- Views: 414
Re: Molecularity
Like everyone is saying, I wouldn't worry too much about 4 or more molecules colliding, and if you know how to identify uni-, bi-, and termolecular reactions, you should be fine. Hope this helps!
- Sat Mar 07, 2020 2:03 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Arrhenius Equation
- Replies: 6
- Views: 430
Re: Arrhenius Equation
you use it to find the equilibrium constant K. It defines the relationship between activation energy, the equilibrium constant, temperature, and A which is the frequency factor. How do we get A or the frequency factor? I'm assuming that it's experimentally determined and will probably be given to u...
- Sat Mar 07, 2020 2:02 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Concentration cell
- Replies: 8
- Views: 625
Re: Concentration cell
For a concentration cell, the species in the anode and the species in the cathode are the exact same. In a general galvanic cell, they can be different. Hope this helps!
- Mon Feb 24, 2020 2:42 pm
- Forum: Balancing Redox Reactions
- Topic: half reaction
- Replies: 10
- Views: 707
Re: half reaction
Pages 538-543 in the textbook were really helpful for me, and it explains each step clearly and gives examples to follow along with. Hope this helps!
- Mon Feb 24, 2020 2:39 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Hw 8
- Replies: 10
- Views: 717
Re: Hw 8
It may also be helpful to email your TA and clarify what's cool with them. Hope this helps!
- Mon Feb 24, 2020 2:36 pm
- Forum: Appications of the Nernst Equation (e.g., Concentration Cells, Non-Standard Cell Potentials, Calculating Equilibrium Constants and pH)
- Topic: What does the nitrate do in a concentration cell?
- Replies: 6
- Views: 479
Re: What does the nitrate do in a concentration cell?
Like everyone is saying, nitrate diffuses through the porous disk to balance the charges on both sides. The actual reaction that is occurring is for Ag+(aq) and Ag(s), but NO3- is important for the concentration cell as well.
- Mon Feb 24, 2020 2:34 pm
- Forum: Balancing Redox Reactions
- Topic: Platinum in cell diagram
- Replies: 10
- Views: 787
Re: Platinum in cell diagram
In addition to what everyone is saying, there was a specific example done in lecture. In his example last Friday, Professor Lavelle showed a cell with copper and iron in it. In the copper half reaction, Cu(s) was present, so we can use the solid itself as an electrode. However, in the iron half reac...
- Mon Feb 24, 2020 2:29 pm
- Forum: Appications of the Nernst Equation (e.g., Concentration Cells, Non-Standard Cell Potentials, Calculating Equilibrium Constants and pH)
- Topic: Ion-Selective Electrodes
- Replies: 2
- Views: 213
Re: Ion-Selective Electrodes
In addition to what @Brian J Cheng 1I mentioned, the ion concentration inside the glass meter is different form the ion concentration in the solution, creating an electrochemical gradient, and therefore, a e- current.
- Sun Feb 23, 2020 12:39 am
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Anode vs Cathode
- Replies: 6
- Views: 405
Re: Anode vs Cathode
In addition to what @Jasmine Fendi 1D mentioned, the anode is where the oxidation takes place and the cathode is where reduction occurs. Hope this helps!
- Sun Feb 23, 2020 12:38 am
- Forum: Appications of the Nernst Equation (e.g., Concentration Cells, Non-Standard Cell Potentials, Calculating Equilibrium Constants and pH)
- Topic: cell potention in nernst
- Replies: 3
- Views: 263
Re: cell potention in nernst
Eo is the cell potential under standard conditions, and E is the cell potential at any other condition. Hope this helps!
- Sun Feb 23, 2020 12:36 am
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: inert conductor
- Replies: 2
- Views: 184
Re: inert conductor
We use an inert conductor when one of the redox couples does not have a solid in it. In his example on Friday, Professor Lavelle showed a cell with copper and iron in it. In the copper half reaction, Cu(s) was present, so we can use the solid itself as an electrode. However, in the iron half reactio...
- Sun Feb 23, 2020 12:34 am
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Salt Bridge
- Replies: 1
- Views: 225
Re: Salt Bridge
Yes, the salt bridge prevents a buildup of charge in the galvanic cell by letting ions from one electrode flow to the other side. Professor Lavelle mentioned the flow of Cl- ions in the galvanic cell example on Friday, so referring to that might help.
- Thu Feb 20, 2020 11:02 pm
- Forum: Student Social/Study Group
- Topic: What Song Was Playing During Lecture?
- Replies: 2
- Views: 310
What Song Was Playing During Lecture?
When he showed the midterm average, Professor Lavelle started playing a song, and it was really catchy and helped me smile through the pain. Now the tune's stuck in my head and I don't know what it is – can someone please tell me the name of the song so I can bump it after getting destroyed by Test ...
- Sat Feb 15, 2020 12:21 am
- Forum: Entropy Changes Due to Changes in Volume and Temperature
- Topic: Entropy in reversible and irreversible
- Replies: 3
- Views: 293
Re: Entropy in reversible and irreversible
Since entropy is a state function and the path between initial and final states doesn't matter, the entropy of the system is nRln( \frac{V_{2}}{V_{1}} ) for both reversible and irreversible expansion. However, as @Samuel G Rivera - Discussion 4I mentioned, the main difference is what happens to the ...
- Sat Feb 15, 2020 12:12 am
- Forum: Applying Le Chatelier's Principle to Changes in Chemical & Physical Conditions
- Topic: Pressure and Moles
- Replies: 7
- Views: 456
Re: Pressure and Moles
Count the moles of gas on each side by counting the coefficients, which will help you determine the correct way equilibrium shifts.
- Sat Feb 15, 2020 12:08 am
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: Constant Pressure and Volume
- Replies: 6
- Views: 463
Re: Constant Pressure and Volume
Adding on to what was said above, in constant pressure systems, we typically have to account for expansion work, but in constant volume systems, we don't (the gas can't expand if there's no volume change). Hope this helps!
- Thu Feb 13, 2020 1:50 pm
- Forum: Ideal Gases
- Topic: STP
- Replies: 12
- Views: 1645
Re: STP
STP, which is standard temperature and pressure, is 1 atm and 273 K (273.15, to be specific).
This is often confused with standard state/standard conditions, which is 298 K, This is also referred to as room temperature.
Hope this helps!
This is often confused with standard state/standard conditions, which is 298 K, This is also referred to as room temperature.
Hope this helps!
- Thu Feb 13, 2020 1:29 pm
- Forum: Administrative Questions and Class Announcements
- Topic: grading curve
- Replies: 10
- Views: 695
Re: grading curve
He doesn't curve tests, but he said that he looks at the point distributions at the end of the quarter and adjusts grades as necessary. Last quarter for 14A, I think it helped people by a couple points, but I wouldn't bank on it to save a grade. Hope this helps!
- Mon Feb 03, 2020 10:28 am
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: units for reaction enthalpy and enthalpy of formation
- Replies: 3
- Views: 240
Re: units for reaction enthalpy and enthalpy of formation
You can use both interchangeably. According to my TA (Dane), the per mol part simply clarifies that we are calculating the energy absorbed/release if the entire reaction occurred 1 mol times. Hope this helps!
- Mon Feb 03, 2020 10:20 am
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: work equations
- Replies: 5
- Views: 224
Re: work equations
What about when we are calculating the change in internal energy in a system under constant pressure? Work is involved, do we always use the negative sign in the expression for work? We don't just put the negative sign because work is involved; rather, we add the negative sign because the system is...
- Mon Feb 03, 2020 10:08 am
- Forum: Student Social/Study Group
- Topic: Midterm study
- Replies: 8
- Views: 507
Re: Midterm study
I also found Lyndon and Matthew's 3 hour session to be super helpful last quarter. It's on Monday, February 10 this time from 6-9 P.M., and they give a super helpful worksheet and talk through the problem solving process, so I highly recommend attending if you have the time!
- Mon Feb 03, 2020 10:04 am
- Forum: Calculating Work of Expansion
- Topic: Midterm Question Involving Integrals
- Replies: 3
- Views: 254
Re: Midterm Question Involving Integrals
We may cover something in lecture this week that involves integrals, so I would review the equation sheet and integral rules just in case. Hope this helps!
- Mon Feb 03, 2020 10:03 am
- Forum: Administrative Questions and Class Announcements
- Topic: week 5 homework
- Replies: 4
- Views: 205
Re: week 5 homework
Since we are also incorporating a lot of thermochemistry and First Law concepts into our lectures for the Second and Third Laws, I'm pretty sure it would be fine to have some problems from the previous unit for Week 5 HW. However, I would include some problems from the current unit as well. Hope thi...
- Thu Jan 30, 2020 9:09 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: Heat Capacity
- Replies: 2
- Views: 157
Re: Heat Capacity
Divide by the number of moles, i.e. amount of substance.
- Thu Jan 30, 2020 9:09 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: Hess's Law vs. Standard Enthalpies of Formation
- Replies: 5
- Views: 260
Re: Hess's Law vs. Standard Enthalpies of Formation
Adding on to what @Zoya Mulji 1K said, if we're given a table of standard enthalpies of formation, the problem will want us to use enthalpies of product minus enthalpies of reactants. Most of the time, it should be pretty clear what method the problem wants you to use. Hope this helps!
- Thu Jan 30, 2020 8:59 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: Bond Enthalpies
- Replies: 5
- Views: 234
Re: Bond Enthalpies
A measured bond enthalpy is the average value of a bond that was calculated from many different molecules, so it is less accurate. A C–H bond demonstrates this perfectly. When we look at CH 4 , C 2 H 2 , and CHCl 3 , the bond enthalpy for each C–H bond will be different for each molecule. So, in ord...
- Thu Jan 30, 2020 8:47 pm
- Forum: Concepts & Calculations Using First Law of Thermodynamics
- Topic: Equipartition Theorem
- Replies: 2
- Views: 173
Re: Equipartition Theorem
Adding on to what @Goyama_2A mentioned, understanding the basics of the theorem will help when deriving the formulas mentioned later in the chapter. Hope this helps!
- Thu Jan 30, 2020 8:45 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Test #1// #5
- Replies: 5
- Views: 259
Re: Test #1// #5
If you go through the acid-base examples that we did in class (on a Friday, I believe), there was a problem that had a similar structure to #5. This should help clarify the steps as well. Hope this helps!
- Wed Jan 22, 2020 4:11 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Percentage Ionization
- Replies: 2
- Views: 96
Re: Percentage Ionization
Adding on to @KeyaV1C, you calculate [concentration of deprotonated species] with an ICE table and the K value. Hope this helps!
- Wed Jan 22, 2020 4:07 pm
- Forum: Phase Changes & Related Calculations
- Topic: Enthalpy of vaporization
- Replies: 2
- Views: 188
Re: Enthalpy of vaporization
In order to vaporize a substance, you need to break all the IMFs holding the sample together (because a gas, for our purposes, exhibits no IMFs). This takes a lot of energy compared to melting (where you only need to break a few IMFs), which is why the enthalpy of vaporization is so high. Hope this ...
- Wed Jan 22, 2020 4:04 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Acids and Bases pka and pkb
- Replies: 8
- Views: 349
Re: Acids and Bases pka and pkb
Adding on to what @Hui Qiao Wu 1I mentioned, the lower the pka of an acid, the higher the pKb of its conjugate base. This is because of the equation pka + pKb = 14. Hope this helps!
- Wed Jan 22, 2020 4:02 pm
- Forum: Phase Changes & Related Calculations
- Topic: Phase changes: endothermic vs exothermic
- Replies: 12
- Views: 1303
Re: Phase changes: endothermic vs exothermic
Yes, the reverse reactions (condensation, freezing, and deposition) would be exothermic because they release energy, forming a more stable phase of matter.
- Wed Jan 22, 2020 4:00 pm
- Forum: Phase Changes & Related Calculations
- Topic: Temp in sublimation
- Replies: 2
- Views: 207
Re: Temp in sublimation
While a substance is undergoing sublimation, all the energy (from the heat) is being used to break the IMFs that hold the solid together. Therefore, the temperature (which is the average kinetic energy of the molecules) does not change. During sublimation, the heat doesn't make the molecules move fa...
- Sat Jan 18, 2020 5:29 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: pH sig figs
- Replies: 5
- Views: 193
Re: pH sig figs
This link on the Chem 14B website (titled "Everything you want to know about Significant Figures") was really helpful for me: https://lavelle.chem.ucla.edu/wp-content/supporting-files/Chem14B/EVERYTHING_YOU_WANTED_TO_KNOW_ABOUT_SF.pdf The second page has the rules for logarithms, and I hop...
- Sat Jan 18, 2020 5:23 pm
- Forum: *Making Buffers & Calculating Buffer pH (Henderson-Hasselbalch Equation)
- Topic: Buffer questions
- Replies: 4
- Views: 744
Re: Buffer questions
Professor Lavelle said that everything up until the end of Friday was fair game for the test, and since we had a buffer example in class that day, there's a chance that it may be on the test. Hope this helps!
- Sat Jan 18, 2020 5:10 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Fall Quarter Final
- Replies: 2
- Views: 137
Re: Fall Quarter Final
Like @asannajust_1J mentioned, there might be an email sent out, but if not, I would just drop by on Tuesday or later to check it out. Hope this helps!
- Sat Jan 18, 2020 5:07 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Exercise 6A.19
- Replies: 4
- Views: 178
Re: Exercise 6A.19
I believe the correct answer is 3.2 x 10-15 M.
In the solutions manual, the [H3O+] = 3.1 x 10-3 M, but in our book, it's just 3.1 M, leading to a different answer. Hope this helps!
In the solutions manual, the [H3O+] = 3.1 x 10-3 M, but in our book, it's just 3.1 M, leading to a different answer. Hope this helps!
- Sat Jan 18, 2020 5:00 pm
- Forum: Applying Le Chatelier's Principle to Changes in Chemical & Physical Conditions
- Topic: Test 1
- Replies: 6
- Views: 349
Re: Test 1
Professor Lavelle mentioned at the end of class on Friday that it was only chemical equilibrium + acids and bases, so basically everything we covered in the first two weeks. Hope this helps!
- Thu Jan 09, 2020 4:08 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Equilibrium Constants with Ionic Compounds
- Replies: 2
- Views: 188
Re: Equilibrium Constants with Ionic Compounds
When the book says the "activity", it simply means the concentration/partial pressures. As stated in lecture, we approximate the activity by using the concentrations or partial pressures of the species in the reaction. So, all we need to do is write the expression for K like you would norm...
- Thu Jan 09, 2020 4:04 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Why is K unitless?
- Replies: 10
- Views: 667
Re: Why is K unitless?
We use reactivity to calculate K, and concentrations/partial pressures are just an approximation of the reactivities. Reactivities have no units, so K will therefore not have any units. This was covered in the modules and lectures in further detail. Hope this helps!
- Thu Jan 09, 2020 4:01 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Kp v. Kc
- Replies: 5
- Views: 221
Re: Kp v. Kc
After asking this question in office hours yesterday, Professor Lavelle said that if the problem simply asks you to write the expression for K for a reaction with only gases, you can use K p . However, he did make it explicitly clear that K is just a general term for either K c or K p , so it doesn'...
- Thu Jan 09, 2020 3:56 pm
- Forum: Applying Le Chatelier's Principle to Changes in Chemical & Physical Conditions
- Topic: determining shift in equilibrium
- Replies: 4
- Views: 247
Re: determining [censored] in equilibrium
Yes, you need to take the ΔH into account. Since ΔH < 0, the forward reaction is exothermic and the reverse reaction is endothermic. In other words, the forward reaction gives off heat and the reverse reaction uses up heat. Since heat is added, Le Chatelier's Principle states that the system will tr...
- Thu Jan 09, 2020 3:49 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: When to assume x=0 for ICE box problems
- Replies: 1
- Views: 97
Re: When to assume x=0 for ICE box problems
You can make this assumption whenever K < 10 -4 , as stated in Professor Lavelle's modules. Since the value of K is extremely small, we know that only a very, very tiny amount of product will be formed. In other words, only a very, very tiny amount of reactant is used up. So, when subtracting x (the...
- Wed Dec 04, 2019 12:25 pm
- Forum: Amphoteric Compounds
- Topic: amphoteric oxides
- Replies: 3
- Views: 293
Re: amphoteric oxides
As everyone is mentioning, it would probably be useful to know the amphoteric oxides themselves, which is something you have to memorize. It's in the textbook (page 450), and he may ask us questions about them on the final. Hope this helps!
- Wed Dec 04, 2019 12:10 pm
- Forum: Air Pollution & Acid Rain
- Topic: Acid rain eqn
- Replies: 1
- Views: 267
Re: Acid rain eqn
i. SO2 (aq) + H2O (l) 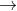 H2SO3 (aq)
H2SO3 (aq)
ii. CO2 (aq) + H2O (l)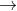 H2CO3 (aq)
H2CO3 (aq)
I believe these are the two reactions we need to know for acid rain. Hope this helps!
ii. CO2 (aq) + H2O (l)
I believe these are the two reactions we need to know for acid rain. Hope this helps!
- Wed Dec 04, 2019 9:33 am
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: week 9 hw problems
- Replies: 4
- Views: 295
Re: week 9 hw problems
I think Week 9 HW should include at least some coordination compound problems (Week 9 was mainly when we covered this topic), but I also did some acid-base problems for Week 9. Week 10 should definitely be acids and bases, though. Hope this helps!
- Wed Dec 04, 2019 9:29 am
- Forum: Biological Examples
- Topic: Cisplatin
- Replies: 6
- Views: 471
- Wed Dec 04, 2019 9:18 am
- Forum: Coordinate Covalent Bonds
- Topic: When do we use the prefixes bis, tris, tetrakis, etc?
- Replies: 4
- Views: 655
Re: When do we use the prefixes bis, tris, tetrakis, etc?
When a ligand is polydentate and you have more than one of these ligands, you use bis-, tris-, etc. This is done because some polydentate ligands already have the prefixes di-, tri-, etc. in them (ex: di ethylene tri amine). By using bis-, tris-, and the alternate prefixes, you can clearly show how ...
- Thu Nov 28, 2019 11:59 am
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Test #2 handback
- Replies: 11
- Views: 730
Re: Test #2 handback
I had my discussion on Wednesday and got my test back, so I'm assuming that everyone who had discussion on Monday, Tuesday, and Wednesday received their tests. If you have discussion on Thursday or Friday, you'll most likely get it back in discussion next week or after a lecture (either Monday or We...
- Thu Nov 28, 2019 11:54 am
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Week 9 homework
- Replies: 11
- Views: 758
Re: Week 9 homework
Most TAs are collecting Week 9 and Week 10 HW together during their discussions next week. As @NRobbins_1K also mentioned, Professor Lavelle sent out an email saying that we would have the option of turning in Week 9 HW during Week 10 discussion.
- Thu Nov 28, 2019 11:51 am
- Forum: Properties & Structures of Inorganic & Organic Acids
- Topic: Final Exam
- Replies: 10
- Views: 726
Re: Final Exam
The final will be cumulative, but it will probably focus a bit more on the second half of the quarter because the first half was tested extensively during the midterm. However, I would still study everything carefully to be prepared. Hope this helps!
- Thu Nov 28, 2019 11:48 am
- Forum: Calculating pH or pOH for Strong & Weak Acids & Bases
- Topic: Strength and Weakness
- Replies: 12
- Views: 1134
Re: Strength and Weakness
Would the pKa and Ka values be given or would we have to calculate that ourselves? You only need one value to make the comparison, so if you're given either the K a or pK a , you can tell which one is the stronger acid. However, sometimes you may not be given either of these values. If this is the ...
- Thu Nov 28, 2019 11:38 am
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: Test Questions?
- Replies: 2
- Views: 218
Re: Test Questions?
In addition to naming, Professor Lavelle also could ask us about the shape and coordination number of the coordination complex. He may ask us which ligands are polydentate, and as @Vincent Leong 1A mentioned, he could also ask us about their biological importance.
- Wed Nov 20, 2019 3:21 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: intermolecular repulsions
- Replies: 2
- Views: 202
Re: intermolecular repulsions
I took my test today and nothing related to intermolecular repulsions was there, so you should be good.
- Wed Nov 20, 2019 3:14 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: London forces vs. dipole-dipole for halogen-containing molecules
- Replies: 3
- Views: 215
Re: London forces vs. dipole-dipole for halogen-containing molecules
To expand upon @Brittney Hun 1K and clarify a couple of things, both CHF 3 and CHI 3 exhibit dipole-dipole forces, so this doesn't matter in determining the melting/boiling points. What matters, therefore, is the LDFs of each molecule. CHI 3 is a larger molecule/has a higher molar mass, and this cor...
- Wed Nov 20, 2019 3:07 pm
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: Compound Arrangement
- Replies: 2
- Views: 141
Re: Compound Arrangement
Like @Caitlyn Tran 1G mentioned, there's no specific reason or time where you need to write the ammonia backwards. It's just a clarification thing, so you won't need to memorize when you would need to write a molecule backwards.
- Wed Nov 20, 2019 1:48 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Do we need to know the pentagonal bipyramidal model for Test 2?
- Replies: 6
- Views: 417
Re: Do we need to know the pentagonal bipyramidal model for Test 2?
After asking my TA and also taking the test, I can confidently say that no, we won't need to know it. Hope this helps!
- Wed Nov 20, 2019 1:46 pm
- Forum: Lewis Structures
- Topic: Strength of bonds
- Replies: 3
- Views: 436
Re: Strength of bonds
HO- is more tightly bound because the oxygen has a smaller atomic radius, so the nuclear forces of attraction are much stronger and pull the electrons more tightly. The high electronegativity of oxygen also leads to this conclusion.
- Thu Nov 14, 2019 10:54 am
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: Electronegativity and Diplole Moments
- Replies: 2
- Views: 120
Re: Electronegativity and Diplole Moments
If two different atoms are covalently bonded together, the electrons are shared unequally. The more electronegative atom pulls all the electrons closer to itself, making the atom partially negative. Since the electrons are being pulled away most of the time from the less electronegative atom, the le...
- Thu Nov 14, 2019 10:48 am
- Forum: Octet Exceptions
- Topic: existence of radicals
- Replies: 1
- Views: 281
Re: existence of radicals
Yes, it is extremely unstable for radicals to exist, which is why they're highly reactive and don't last for a long time. For example the CH3 molecule is a radical, and it easily reacts with another H atom to form CH4, a much more stable molecule.
- Thu Nov 14, 2019 10:42 am
- Forum: Determining Molecular Shape (VSEPR)
- Topic: bond angles
- Replies: 10
- Views: 513
Re: bond angles
I'm pretty sure it's just something you have to memorize. For some molecules it's pretty intuitive (ex: since CO 2 is a linear molecule, the bond angle is 180 degrees), but once you get to the more complicated molecules, you just have to memorize them. There's a chart in the textbook/online to help ...
- Thu Nov 14, 2019 10:35 am
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: Hydrogen Bonding
- Replies: 2
- Views: 104
Re: Hydrogen Bonding
This model wouldn't be able to hydrogen bond even if a carbon had a lone pair. This is because for hydrogen bonding, 1) the H needs to be bonded to a highly electronegative atom, specifically N, O, or F and 2) the N, O, or F has to have a lone pair. Since C 2 H 4 doesn't have an H bonded to an N, O,...
- Thu Nov 14, 2019 10:32 am
- Forum: Administrative Questions and Class Announcements
- Topic: TEST 2
- Replies: 8
- Views: 472
Re: TEST 2
Test 2 will most likely cover intermolecular forces, VSEPR and Lewis structures, and whatever we learn during lecture on Friday/Monday. There is a chance that it could be a cumulative test, so I would just ask the TAs to be sure. Hope this helps!
- Fri Nov 08, 2019 3:05 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: Test 2
- Replies: 6
- Views: 430
Re: Test 2
For future reference, the test and exam schedule is on the Class Website. It's a link titled "Test and Exam Schedule" and it's pretty easy to find. Hope this helps!
- Fri Nov 08, 2019 3:01 pm
- Forum: Lewis Structures
- Topic: Midterm grades
- Replies: 26
- Views: 1487
Re: Midterm grades
He just announced in the 2 P.M. lecture that we're getting our midterms back on Wednesday during our lectures. Good luck!
- Thu Nov 07, 2019 10:23 pm
- Forum: Administrative Questions and Class Announcements
- Topic: What homework to turn in per week
- Replies: 7
- Views: 398
Re: What homework to turn in per week
@Cassandra_1K If you're unsure, you can ask your TA for clarification and I'm sure they'd be happy to answer. Chemistry Community is also a great resource!
- Thu Nov 07, 2019 10:21 pm
- Forum: Bond Lengths & Energies
- Topic: dissociation energy
- Replies: 6
- Views: 429
Re: dissociation energy
Like @Daniel Honeychurch 1B mentioned, for Chem 14A, we'll be given the experimental values of the dissociation energies. Professor Lavelle mentioned that we will be calculating dissociation energies in Chem 14B, though.
- Thu Nov 07, 2019 10:15 pm
- Forum: Lewis Structures
- Topic: Formal Charges
- Replies: 15
- Views: 1012
Re: Formal Charges
The +/- charge doesn't really depend on the central atom. Instead, it depends on the electronegativity of the atoms. You want the most electronegative atom to have a - charge on in, so for something like PO 4 3- , it wouldn't be the P that has a negative formal charge, but rather the oxygens around ...
- Sun Nov 03, 2019 12:51 am
- Forum: Bond Lengths & Energies
- Topic: bond length
- Replies: 6
- Views: 304
Re: bond length
As everyone has mentioned, there is probably a way to find the exact bond length (in angstroms) based on its strength or vice versa, but I believe that's beyond the scope of this course. I think the main thing we need to know is the relationship between bond length and strength – the longer the bond...
- Sun Nov 03, 2019 12:47 am
- Forum: Properties of Light
- Topic: Equations
- Replies: 4
- Views: 204
Re: Equations
The reference sheet is probably going to be exactly the same as the one we got for Test 1, so you could look at that for reference while preparing. Hope this helps!
- Sun Nov 03, 2019 12:41 am
- Forum: Dipole Moments
- Topic: Dipole Moments on Lewis Structures
- Replies: 2
- Views: 111
Re: Dipole Moments on Lewis Structures
As @AGulati_4A mentioned, drawing the "slightly negative" and "slightly positive" symbols along with the vectors indicate a dipole moment. The vector should start from the slightly positive atom and point toward the slightly negative atom.
- Sun Nov 03, 2019 12:38 am
- Forum: Bohr Frequency Condition, H-Atom , Atomic Spectroscopy
- Topic: Atomic Spectra Midterm Q
- Replies: 2
- Views: 139
Re: Atomic Spectra Midterm Q
Like @Sebastian Lee 1H said, you probably won't need to memorize the exact color, but memorizing the wavelengths for different types of light (UV, visible, X-ray, etc.) would be helpful.
- Sun Nov 03, 2019 12:34 am
- Forum: Octet Exceptions
- Topic: Expanded Octets
- Replies: 1
- Views: 111
Re: Expanded Octets
Elements with electrons in the 3p subshell can expand their octets because they have empty 3d orbitals, allowing them to have more than 8 electrons. This is true for the p block in Period 3 and for all elements below Period 3, and we typically see expanded octets occurring in the halogen and noble g...
- Fri Oct 25, 2019 9:22 pm
- Forum: Properties of Light
- Topic: Hz vs. frequency
- Replies: 7
- Views: 655
Re: Hz vs. frequency
Hertz (or s-1) is simply the unit for frequency, just like the kilogram is the unit for mass. Every time it asks for the frequency, your response will be with the unit Hz (or MHz, GHz, etc).
- Fri Oct 25, 2019 9:21 pm
- Forum: Ionic & Covalent Bonds
- Topic: ion charge
- Replies: 3
- Views: 144
Re: ion charge
@Katherine Wu: Ga is a weird one because it's in Column/Family 3 of the periodic table, meaning it tends to form a 3+ charge. It confused me at first, but then I realized there's two ways to look at this: 1. Look at the family Ga is in. Ga is in Family 13 (or 3A). Then, I looked at the elements abov...
- Fri Oct 25, 2019 9:13 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: Determining values of ml
- Replies: 1
- Views: 104
Re: Determining values of ml
As Professor Lavelle mentioned during lecture, the values of ml and what orbitals they correspond to are somewhat arbitrary. Typically, if ml = -1, 0, 1 for the p subshell, we make px = -1, py = 0, and pz = 1 because it's convenient and intuitively makes sense. However, I don't think the numbers nee...
- Fri Oct 25, 2019 9:10 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: Calculating Subshells
- Replies: 3
- Views: 150
Re: Calculating Subshells
The subshells specifically refer to s, p, d, and f, which is what the angular momentum number gives you. Once you get to the magnetic quantum number, you're talking about orbitals, NOT subshells. So, for n = 4, l = 0, 1, 2, and 3, which correspond to s, p, d, and f. The answer is therefore 4s, 4p, 4...
- Fri Oct 25, 2019 9:05 pm
- Forum: Photoelectric Effect
- Topic: Increasing Light Intensity Photoelectric Effect
- Replies: 7
- Views: 923
Re: Increasing Light Intensity Photoelectric Effect
Yes – once a sufficient threshold energy is reached (by increasing frequency), the amplitude of the wave does matter. Increasing the amplitude proportionally increases the number of electrons ejected. So, if you double the amplitude, you double the number or electrons ejected, and so on. Hope this h...
- Sat Oct 19, 2019 10:49 pm
- Forum: Student Social/Study Group
- Topic: Grades
- Replies: 18
- Views: 1232
Re: Grades
Does anyone know if there is a way to calculate our grades ourselves? Is it point system? The grading system is point-based, but as Professor Lavelle states on the syllabus, letter grades aren't assigned until the very end. I don't think there's a way to know what letter you have, but you can estim...
- Sat Oct 19, 2019 10:37 pm
- Forum: Student Social/Study Group
- Topic: Homework for Week 4
- Replies: 4
- Views: 213
Re: Homework for Week 4
Since the material we're covering in class still relates to The Quantum World, yes, you can still do problems from this unit.
- Sat Oct 19, 2019 6:28 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: Tips for the Midterm
- Replies: 10
- Views: 383
Re: Tips for the Midterm
Like MTanikella_1C mentioned, the video modules, specifically the post-assessments, are pretty useful because they ask both conceptual questions and questions that require you to apply a formula. Reviewing them once or twice should help!
- Sat Oct 19, 2019 6:25 pm
- Forum: *Shrodinger Equation
- Topic: Review
- Replies: 4
- Views: 179
Re: Review
The Schrodinger wave function was covered briefly in the modules, but I believe that for topics we are now doing in class (quantum numbers, electron configurations, Lewis dot diagrams, etc), there are no modules for it on the Chem 14A website.
- Sat Oct 19, 2019 6:21 pm
- Forum: General Science Questions
- Topic: Midterm
- Replies: 3
- Views: 232
Re: Midterm
Adding onto this, the syllabus mentioned that one homework problem would be on the midterm exam, so doing all of these problems and reviewing them would probably help.
- Mon Oct 14, 2019 8:24 pm
- Forum: Accuracy, Precision, Mole, Other Definitions
- Topic: Test 1 [ENDORSED]
- Replies: 107
- Views: 27734
Re: Test 1 [ENDORSED]
@Keerthana Sivathasan 4G
Yeah, my TA said today that we'd get it back during our discussion this week.
Yeah, my TA said today that we'd get it back during our discussion this week.
- Sat Oct 12, 2019 6:06 pm
- Forum: Bohr Frequency Condition, H-Atom , Atomic Spectroscopy
- Topic: Frequency doesn't match energy difference
- Replies: 2
- Views: 210
Re: Frequency doesn't match energy difference
Yes, the light simply passes through the object. Professor Lavelle mentioned this briefly in class.
- Sat Oct 12, 2019 6:00 pm
- Forum: DeBroglie Equation
- Topic: Crossing out units
- Replies: 3
- Views: 196
Re: Crossing out units
As @Akshay Chellappa 1H mentioned, the numerator term for the equation has the units J * s. Although J is kg * m^2 * s^-2, the J will be multiplied by the s to cancel out part of the s^-2, making the numerator kg * m^2 * s^-1 (Note the s^-1 instead of the s^-2). Then, the overall fraction will be (k...
- Sat Oct 12, 2019 5:52 pm
- Forum: Trends in The Periodic Table
- Topic: Chemistry Community
- Replies: 8
- Views: 503
Re: Chemistry Community
@Matt Sanruk 4F: I believe Professor Lavelle and his TAs have probably figured a system out. Although I don't know how it works exactly, he's been using this system for quite a while and I trust that he knows what he's doing. I think as long as we do the 5 posts by every Sunday, we should be fine.
- Sat Oct 12, 2019 5:47 pm
- Forum: Properties of Electrons
- Topic: Wave Properties of Electrons
- Replies: 4
- Views: 166
Re: Wave Properties of Electrons
As many people here are stating, electrons were determined to have wave-like properties due to the diffraction patterns created during the double slit and screen experiment. It was known that diffraction patterns were a key characteristic of waves, so when the experiment resulted in diffraction patt...
- Sat Oct 12, 2019 5:37 pm
- Forum: Accuracy, Precision, Mole, Other Definitions
- Topic: Test 1 [ENDORSED]
- Replies: 107
- Views: 27734
Re: Test 1 [ENDORSED]
Like @Rebecca Remple 1J said, we should probably getting them back by the next discussion or by the end of next week (according to the TAs). Worst case, we get them during discussion in Week 4.
- Wed Oct 02, 2019 3:41 pm
- Forum: Balancing Chemical Reactions
- Topic: Combustion [ENDORSED]
- Replies: 3
- Views: 241
Re: Combustion [ENDORSED]
Like @megan3j said, there will always be carbon dioxide and water as products in combustion reactions. There will also always be oxygen (diatomic form) as a reactant. There may also be some additional elements in the combustion reaction (e.g. nitrogen). However, for these problems, you probably will...

