Search found 50 matches
- Thu Mar 12, 2020 4:47 pm
- Forum: Arrhenius Equation, Activation Energies, Catalysts
- Topic: activation energy units
- Replies: 3
- Views: 529
activation energy units
does activation energy depend on the number of moles of something or is it a constant value for a certain reaction?
- Thu Mar 12, 2020 4:38 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: comparing rates
- Replies: 3
- Views: 266
comparing rates
if a chemical equation is balanced and all the reactants and products have a stoichiometric coefficient of 1, for example, would the rates of formation for the products be the same as the rate of consumption of the reactants?
- Wed Mar 11, 2020 7:19 pm
- Forum: Arrhenius Equation, Activation Energies, Catalysts
- Topic: Temperature and Reaction Rate
- Replies: 6
- Views: 477
Re: Temperature and Reaction Rate
they are proportional in that if you increase temperature, k will increase as well and also vice versa if you decrease temperature
- Wed Mar 11, 2020 12:34 pm
- Forum: General Rate Laws
- Topic: Instantaneous rate
- Replies: 4
- Views: 351
Re: Instantaneous rate
i think so because there are much more applications with the instantaneous rate law, such as differential rate law, integrated rate law, and for things like finding the half life of species and such
- Wed Mar 11, 2020 12:30 pm
- Forum: First Order Reactions
- Topic: determining Kr
- Replies: 5
- Views: 379
Re: determining Kr
because for a first order rxn, the plot for a linear slope would be: ln[A]= -kt + ln[A(initial)]
When comparing it to the linear form y=mx+b, you'll see that -k is m, or the slope. so to find k, you would multiply the slope, -k, by -1
When comparing it to the linear form y=mx+b, you'll see that -k is m, or the slope. so to find k, you would multiply the slope, -k, by -1
- Thu Mar 05, 2020 9:35 pm
- Forum: Van't Hoff Equation
- Topic: delta G, pressure and. K
- Replies: 2
- Views: 412
Re: delta G, pressure and. K
it is temperature dependent because the equation delta G = -RTln(k) and also k is dependent on temperature too . also Delta G naught is when pressure is constant so there's that
- Wed Mar 04, 2020 11:38 pm
- Forum: Method of Initial Rates (To Determine n and k)
- Topic: units of k
- Replies: 9
- Views: 716
units of k
I know the units of k is dependent on the order of the rate equation, but how do we use that knowledge to determine the units of k?
- Wed Mar 04, 2020 11:34 pm
- Forum: General Rate Laws
- Topic: Independent Rates (7A.15)
- Replies: 2
- Views: 279
Re: Independent Rates (7A.15)
to find the order of C you want to find the experiments where [C] changes, but [A] and [B] remain constant, which would be experiments 1 and 4. to do this, you set up a rate equation but with fractions, with the numerator being the experiment where [C] is largest (so it's easier). You would get: (70...
- Wed Mar 04, 2020 11:30 pm
- Forum: General Rate Laws
- Topic: First vs Second Order Rate Laws
- Replies: 3
- Views: 324
Re: First vs Second Order Rate Laws
increasing the concentration of a reactant will have a greater effect on second order rxns because the rate = k[A]^2 for second order vs rate = k[A] for first order
- Wed Mar 04, 2020 11:28 pm
- Forum: General Rate Laws
- Topic: conceptual question (zero order)
- Replies: 3
- Views: 293
Re: conceptual question (zero order)
so for first order reactions, its typically one of the molecules thats first order reacting with the solvent molecules, so technically it is only one of the atoms/molecules in question. Zero order essentially renders a molecule "useless" with the overall rate as the slow step determines th...
- Fri Feb 28, 2020 12:28 am
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: 6M.5
- Replies: 2
- Views: 244
6M.5
for a, the answer says the cell diagram is Hg|Hg2 2+||NO3-, H+|O (g)| Pt
how did they get this as the cell diagram?
how did they get this as the cell diagram?
- Thu Feb 27, 2020 11:54 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: cell diagram
- Replies: 3
- Views: 231
cell diagram
when writing the cell diagram and the reduction reaction will require a Platinum electrode, does the diagram look like (for example) ||Cl-|Pt or is it ||Pt|Cl- or does it even matter
- Thu Feb 27, 2020 11:39 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Textbook question 6L.5
- Replies: 2
- Views: 253
Re: Textbook question 6L.5
since its starting with Au+, the oxidation reaction would be Au+-->Au3+ (bc losing e-) and the reduction would be Au+--->Au(s) (bc gaining e-). really for balancing you would just be adding electrons to each side, then multiply both half reactions by the coeffeicient that would give you an equal num...
- Thu Feb 27, 2020 11:36 pm
- Forum: Balancing Redox Reactions
- Topic: Oxygen's oxidation #
- Replies: 6
- Views: 462
Re: Oxygen's oxidation #
since O2 is in it's most stable form at standard conditions, its oxidation state would be 0 . Same applies for other elements in their standard state (ie. H2, Cl2, etc)
- Thu Feb 27, 2020 11:32 pm
- Forum: Work, Gibbs Free Energy, Cell (Redox) Potentials
- Topic: Work and Cell Potential Relationship
- Replies: 3
- Views: 252
Re: Work and Cell Potential Relationship
since the work is work max, and work max is = to 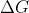 , which is = to nFE(cell), then work max= nFE(cell)
, which is = to nFE(cell), then work max= nFE(cell)
- Fri Feb 21, 2020 1:59 am
- Forum: Work, Gibbs Free Energy, Cell (Redox) Potentials
- Topic: 6L.1 (b)
- Replies: 1
- Views: 143
6L.1 (b)
did anyone else get a really large number? I got 2240kJ and I dont think this seems right
- Fri Feb 21, 2020 1:27 am
- Forum: Balancing Redox Reactions
- Topic: balancing half reactions in a basic solution
- Replies: 7
- Views: 449
balancing half reactions in a basic solution
When adding the OH- to balance the H+, do you have to add it to both sides?
- Fri Feb 21, 2020 1:20 am
- Forum: Balancing Redox Reactions
- Topic: 6K.5 part a
- Replies: 2
- Views: 215
Re: 6K.5 part a
when balancing by using stoichiometric coefficients, you do that to balance things that are not either Oxygen or Hydrogen
- Fri Feb 21, 2020 1:16 am
- Forum: Balancing Redox Reactions
- Topic: Reduction vs. oxidation
- Replies: 29
- Views: 1211
Re: Reduction vs. oxidation
you can calculate the oxidation numbers of all of the different elements constituting the molecules and if an oxidation number increases, then that atom/molecule is oxidized, and if the oxidation number decreases then that atom/molecule is reduced
- Thu Feb 20, 2020 9:44 pm
- Forum: Balancing Redox Reactions
- Topic: oxidation numbers
- Replies: 4
- Views: 312
oxidation numbers
when an oxidation number decreases, is it reduced? or is it oxidized because does the oxidation number lowering correlate to a loss of electrons?
- Thu Feb 13, 2020 10:53 pm
- Forum: Calculating Standard Reaction Entropies (e.g. , Using Standard Molar Entropies)
- Topic: Why is delta U = 0 for isothermal reactions?
- Replies: 11
- Views: 4370
Re: Why is delta U = 0 for isothermal reactions?
since its isothermal it means energy cannot come in or leave, so by nature the change in energy would be zero
- Thu Feb 13, 2020 10:19 pm
- Forum: Entropy Changes Due to Changes in Volume and Temperature
- Topic: Cv vs Cp
- Replies: 17
- Views: 994
Re: Cv vs Cp
Cv is when there is no change in volume and Cp when there is no change in pressure
- Thu Feb 13, 2020 9:59 pm
- Forum: Calculating Standard Reaction Entropies (e.g. , Using Standard Molar Entropies)
- Topic: reaction entropy
- Replies: 7
- Views: 602
reaction entropy
is solving for reaction entropy like the same as solving for reaction enthalpy where its the sum of products molar entropy - sum of reactants molar entropy or is there another formula we have to use?
- Thu Feb 13, 2020 9:34 pm
- Forum: Calculating Standard Reaction Entropies (e.g. , Using Standard Molar Entropies)
- Topic: 4H.9
- Replies: 3
- Views: 340
4H.9
It says containers B and C have 1 mole of atoms bound into diatomic molecules. Does this mean that it's 1 mole of diatomics or .5 mol of diatomics bc the 1 mol of atoms would be split in half? and how would the entropies compare of 1 mol of atoms vs diatomics?
- Wed Feb 12, 2020 10:55 pm
- Forum: Entropy Changes Due to Changes in Volume and Temperature
- Topic: entropy and pressure
- Replies: 2
- Views: 202
entropy and pressure
How is entropy related to pressure? And does this change depending on what type of system is being dealt with?
- Thu Feb 06, 2020 9:57 pm
- Forum: Entropy Changes Due to Changes in Volume and Temperature
- Topic: Positive ∆S
- Replies: 5
- Views: 319
Re: Positive ∆S
Not really yet since we haven't gotten to the gibbs free energy equation deltaG=deltaH - T(deltaS), in which a reaction is spontaneous if its delta G is negative.
- Thu Feb 06, 2020 9:54 pm
- Forum: Calculating Standard Reaction Entropies (e.g. , Using Standard Molar Entropies)
- Topic: 4h.11 (d)
- Replies: 1
- Views: 173
4h.11 (d)
For the reaction 4KClO3 (s) ---> 3KClO4 (s) + KCl (s), what sign should the standard reaction enthalpy be? I got a negative number, but since it's 1 solid going into 2 wouldn't it make sense that the entropy would be increasing?
- Thu Feb 06, 2020 9:36 pm
- Forum: Calculating Standard Reaction Entropies (e.g. , Using Standard Molar Entropies)
- Topic: aq vs gas vs liquid
- Replies: 2
- Views: 128
aq vs gas vs liquid
How would you rank these in terms of relative entropy levels?
- Thu Feb 06, 2020 9:26 pm
- Forum: Concepts & Calculations Using Second Law of Thermodynamics
- Topic: Second Law
- Replies: 1
- Views: 76
Re: Second Law
I'm pretty sure the reversible part is just so we understand that the q value we're using is at its maximum, and also I don't think Lavelle said anything about having to worry about irreversible reactions
- Thu Feb 06, 2020 9:09 pm
- Forum: Entropy Changes Due to Changes in Volume and Temperature
- Topic: 4F.5
- Replies: 1
- Views: 51
4F.5
When given amount of moles and a final and initial volume, what equation and how do we use it to find change in entropy?
- Fri Jan 31, 2020 12:40 am
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: 4D.9
- Replies: 2
- Views: 117
Re: 4D.9
the kJ/mol numbers in question are probably coming from a table that's giving you each molecules deltaH of formation, which is why it's being multiplied by the number of moles of the specific molecule. O2 and N2 aren't included because they're pure molecules, and the delta H of them is 0. Everything...
- Fri Jan 31, 2020 12:36 am
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: Celcius and Kelvin
- Replies: 11
- Views: 458
Re: Celcius and Kelvin
The equation for the conversion is given to us, but when using anything with delta T, the difference in temp will be the same if you use K or C, so it doesn't really matter
- Wed Jan 29, 2020 10:56 pm
- Forum: Phase Changes & Related Calculations
- Topic: 4B.1
- Replies: 6
- Views: 242
4B.1
if work is a piston compressing the gas and the work is given as 300kJ (for example), when solving for delta U of the gas inside a close container using delta U=q+w, would w be positive or negative?
- Wed Jan 29, 2020 10:45 pm
- Forum: Phase Changes & Related Calculations
- Topic: internal energy
- Replies: 11
- Views: 627
internal energy
When calculating internal energy changes, is that the deltaU equation or q?
- Wed Jan 29, 2020 10:34 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: Standard Subtance
- Replies: 3
- Views: 135
Re: Standard Subtance
it could be solid, liquid, aqueous, or gas, it just depends what it happens to be at standard conditions: 1 atm and 25C
- Thu Jan 23, 2020 8:17 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: ice table
- Replies: 13
- Views: 567
Re: ice table
noooo only aqueous and gases are put in the ice table
- Thu Jan 23, 2020 8:15 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Q<K
- Replies: 11
- Views: 298
Re: Q<K
since there is a higher concentration of reactants, the excess of reactants will react to form product, which is why it favors the products since it goes in that direction
- Thu Jan 23, 2020 8:08 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: 6D.13
- Replies: 1
- Views: 94
Re: 6D.13
by thinking about what each molecule is, you could get a general idea, but it would probably be smart to do the ICE tables just incase the pHs are very close to one another
- Wed Jan 22, 2020 10:35 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Acidity Constant
- Replies: 4
- Views: 113
Re: Acidity Constant
Ka x Kb = Kw, so like a higher Ka for an acid (meaning it is a strong acid) would result in a lower Kb for its conjugate base (meaning it's really weak, which makes sense bc its a strong acid)
- Wed Jan 22, 2020 10:33 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: 6D.3
- Replies: 1
- Views: 62
6D.3
After finding the concentration of H3O+ from the pH, do we have to subtract this amount from the initial concentration of HClO2?
- Thu Jan 16, 2020 7:11 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Kb and pKb
- Replies: 2
- Views: 251
Re: Kb and pKb
they are logarithmically proportional. so if kb is big (strong base) then pkb will be small. If kb is small (weak base), then pkb will be larger. same goes for ka and pka
- Thu Jan 16, 2020 7:07 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Converting between K and Kc
- Replies: 2
- Views: 151
Re: Converting between K and Kc
I don't think Lavelle is expecting us to use the equation because we've pretty much moved on to acids and bases and he hasn't told us about it. I'm pretty sure the way he would want us to do it is to use PV=nRT to convert the concentration to partial pressure (or vice versa) and solve for k after
- Thu Jan 16, 2020 7:05 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Cubic functions
- Replies: 3
- Views: 129
Re: Cubic functions
like the person above said you'd have to use a graphing calculator or a program online like desmos and to do so just move all parts to one side of the equation so it equals zero, put that into the graphing program and find where the graph crosses the x axis to get the value for "x"
- Wed Jan 15, 2020 8:08 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: 5J.11
- Replies: 4
- Views: 151
5J.11
for part b what is the importance of "x" being a halogen? also how would you go about solving part b
- Wed Jan 15, 2020 8:07 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: concentration of H3O+
- Replies: 2
- Views: 84
Re: concentration of H3O+
since water has [H3O+] of 10^-7, if the acid gives a concentration of less than that, for example {H3O+] of 3x10^-9, if you add that to the [H3O+] from the autoprotolysis of water, the change in concentration of H3O+ will not have been significant enough to make a difference in the pH, then the pH w...
- Thu Jan 09, 2020 9:42 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Value of K
- Replies: 4
- Views: 236
Re: Value of K
I'm sure there is a threshold that K can reach, but it wouldn't be because of speed or anything. It'd simply have to do with the relative concentrations or pressures of the products and reactants
- Thu Jan 09, 2020 7:19 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: 5H.3
- Replies: 2
- Views: 134
Re: 5H.3
Since the K is not given for the whole equation, you have to add up two of the equations from the table to get the equation from the problem. After doing that, you take the two K values from the equations you added together and multiply the Ks to get the new K value for the overall equation.
- Thu Jan 09, 2020 7:17 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Temperature's affects on chemical equilibria
- Replies: 2
- Views: 105
Re: Temperature's affects on chemical equilibria
It depends one whether the reaction is endothermic or exothermic, and you would just treat "heat" as a reactant or product (depending on which side it's on), and use Le'Chatlier's principle to determine which side an increase or decrease in temperature would cause the reaction to proceed to.
- Wed Jan 08, 2020 10:53 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Aqueous Compounds
- Replies: 3
- Views: 140
Re: Aqueous Compounds
yes! you include aqueous solutions in both K and Q. Water is a pure liquid, so I don't know why it'd be labeled as aq, but if it's water gas then yea use it
- Wed Jan 08, 2020 10:51 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: 5I.29
- Replies: 2
- Views: 87
Re: 5I.29
Honestly, its really situational when you'd have to omit something, so if you're ever confused you could just solve for x with it being in the top and bottom, and in the end the solution (omitting the 2x^2 or not) would pretty much be the same.

