Search found 100 matches
- Sat Mar 14, 2020 8:58 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: HCl as anode and cathode in cell diagram
- Replies: 3
- Views: 261
Re: HCl as anode and cathode in cell diagram
You would take the half reactions: 2H+ (aq,1.0M) + 2e- --> H2 (g,1atm) // E cathode = 0.00V H2 (g,1atm) --> 2H+ (aq,0.075M) + 2e- // E anode = 0.00V Add them together: 2H+ (aq, 1.0M) + H2 (g,1atm) --> 2H+ (aq,0.075M) + H2 (g,1atm) and get Ecell = 0.00V Then use the nernst equation to get: E=E^\circ...
- Sat Mar 14, 2020 5:52 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: HCl as anode and cathode in cell diagram
- Replies: 3
- Views: 261
HCl as anode and cathode in cell diagram
How does this cell diagram work Pt(s)|H 2 (g, 1.0 bar)|HCl(aq, 0.075 mol/L )||HCl(aq, 1.0 mol/L)|H 2 (g, 1.0 bar)|Pt(s)
and how would I balance it to solve for its cell potential?
and how would I balance it to solve for its cell potential?
- Sat Mar 14, 2020 4:47 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Redox from cell diagram
- Replies: 6
- Views: 490
Re: Redox from cell diagram
Similarly with |Cu^{2+}(aq)||Ce^{4+}(aq),Ce^{3+}(aq)|Pt(s)) , how do I view the commas in the diagram to write out a half redox reaction?
, how do I view the commas in the diagram to write out a half redox reaction?
- Sat Mar 14, 2020 4:23 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Redox from cell diagram
- Replies: 6
- Views: 490
Redox from cell diagram
In the cell diagram:
|H_{2}(g)|H^{+}(aq)||Cl^{-}(aq)|Cl_{2}(g)|Pt(s))
H and Cl look like they are both oxidized, how would I write the half redox reactions?
H and Cl look like they are both oxidized, how would I write the half redox reactions?
- Sat Mar 14, 2020 3:12 pm
- Forum: Method of Initial Rates (To Determine n and k)
- Topic: Finding k 7B.3
- Replies: 1
- Views: 231
Finding k 7B.3
How would you go about finding k in part c? 2A 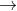 B + C, given that [A]0 = 0.153 mol/L and that after 115 s the concentration of B rises to 0.034 mol/L. Would I use 1/2 A= change in concentration of B?
B + C, given that [A]0 = 0.153 mol/L and that after 115 s the concentration of B rises to 0.034 mol/L. Would I use 1/2 A= change in concentration of B?
- Fri Mar 13, 2020 8:51 pm
- Forum: First Order Reactions
- Topic: 7B.1 mass of drug left in body
- Replies: 2
- Views: 273
7B.1 mass of drug left in body
Does the initial mass of the drug replace the concentration in the rate law equation for solving for the left over mass? 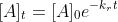
Would I use this equation in 7B.1 to solve for how much drug is left?
Would I use this equation in 7B.1 to solve for how much drug is left?
- Fri Mar 13, 2020 8:34 pm
- Forum: First Order Reactions
- Topic: Rate of reaction and temperature
- Replies: 2
- Views: 241
Rate of reaction and temperature
If the rate constant and initial molarity are given for a first order reaction and so is a temperature at which the reaction is happening, does the temperature factor in to the rate of the reaction?
- Fri Mar 13, 2020 8:05 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Galvanic vs Electrolytic cells
- Replies: 6
- Views: 567
Re: Galvanic vs Electrolytic cells
What exactly would an electrolytic cell do? Is it not generating a voltage charge like a galvanic cell since the energy is turned into chemical?Kimberly Koo 2I wrote:An electrolytic cell converts electrical energy into chemical energy, while a galvanic cell converts chemical energy into electrical energy.
- Fri Mar 13, 2020 7:18 pm
- Forum: Method of Initial Rates (To Determine n and k)
- Topic: Order reaction
- Replies: 4
- Views: 415
Order reaction
How do you tell which order the reaction rate is at? Is it always given? What is it dependent on? I don't very much understand this concept at all, please explain! Thank you!
- Fri Mar 13, 2020 7:08 pm
- Forum: Method of Initial Rates (To Determine n and k)
- Topic: rate constant
- Replies: 5
- Views: 447
rate constant
What is the kr constant? The book just explains it as a proportion of the initial rate of consumption? I am having a hard time understanding this concept.
- Fri Mar 13, 2020 2:37 pm
- Forum: Balancing Redox Reactions
- Topic: redox of chlorine in water 6K.3 d
- Replies: 2
- Views: 233
redox of chlorine in water 6K.3 d
Cl 2 (g) 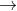 HClO(aq) + Cl 2 (g). I don't know how to find the reduction half of this problem, it shows Cl going to Cl which isn't a loss or gain of electrons.
HClO(aq) + Cl 2 (g). I don't know how to find the reduction half of this problem, it shows Cl going to Cl which isn't a loss or gain of electrons.
- Fri Mar 13, 2020 1:52 pm
- Forum: Balancing Redox Reactions
- Topic: Oxidation number
- Replies: 12
- Views: 1249
Oxidation number
How does oxidation number of an element relate to how many electrons it will lose or gain? I am confused on how to calculate when in acidic or basic solutions it changes the net charges.
- Thu Mar 12, 2020 11:14 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Galvanic vs Electrolytic cells
- Replies: 6
- Views: 567
Galvanic vs Electrolytic cells
Can someone explain the difference between the two? I don't understand what an electrolytic cell is and how it could be different than a galvanic cell.
- Thu Mar 12, 2020 7:27 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Chemistry community posts
- Replies: 3
- Views: 329
Chemistry community posts
Hi, is there a way to separate and count posts made in 14A from 14B as to see how many you've done for each class rather than in total?
- Thu Mar 05, 2020 2:36 am
- Forum: Work, Gibbs Free Energy, Cell (Redox) Potentials
- Topic: finding n in G=-nFE
- Replies: 15
- Views: 1053
finding n in G=-nFE
How do you find n?
- Thu Mar 05, 2020 2:32 am
- Forum: Work, Gibbs Free Energy, Cell (Redox) Potentials
- Topic: Gibbs free energy and balancing redox reactions
- Replies: 2
- Views: 267
Gibbs free energy and balancing redox reactions
Should we check and balance each redox half reaction before trying to calculate gibbs free energy? For example 6L.1: 2 Ce4+ (aq) + 3 I- (aq) 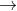 2Ce3+ (aq) + I3- (aq), E cell ° = +1.08 V. Before calculating gibbs, would I need to balance the reaction?
2Ce3+ (aq) + I3- (aq), E cell ° = +1.08 V. Before calculating gibbs, would I need to balance the reaction?
- Thu Mar 05, 2020 2:19 am
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: Delta G and Delta G naught
- Replies: 2
- Views: 248
Delta G and Delta G naught
What is the difference between 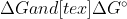 ? Why can
? Why can 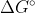 +RTlnQ = [tex]\Delta G?
+RTlnQ = [tex]\Delta G?
- Thu Mar 05, 2020 12:02 am
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: 11.15
- Replies: 12
- Views: 3193
Re: 11.15
How do we know this reaction is at equilibrium? Shouldn't only Q be used?
- Tue Feb 11, 2020 10:24 pm
- Forum: Phase Changes & Related Calculations
- Topic: Heat required for sublimation
- Replies: 2
- Views: 353
Heat required for sublimation
When calculating the heat required for ice to change into water vapor, do the steps need to be broken down into ice to liquid and liquid to water vapor and taking the sum of that since the heat capacities are different at different phases?
- Tue Feb 11, 2020 9:30 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: 4D.15 Sum of enthalpy in equation is positive, but answer is negative
- Replies: 1
- Views: 169
4D.15 Sum of enthalpy in equation is positive, but answer is negative
Subtracting the enthalpy of formation of the product from the reactants generates a positive number, but the answer is negative. Can someone please explain why this is the case?
- Mon Feb 10, 2020 10:11 pm
- Forum: Phase Changes & Related Calculations
- Topic: 4C.7 vaporization enthalpy
- Replies: 2
- Views: 253
4C.7 vaporization enthalpy
How do you go about solving this? The moles of liquid methane are given and the heat required. The only equation I know for enthalpy of vaporization is  Hvap= Hm(vapor) - Hm(liquid).
Hvap= Hm(vapor) - Hm(liquid).
- Mon Feb 10, 2020 10:08 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: 4C.3 finding temperature of ideal gas
- Replies: 1
- Views: 162
Re: 4C.3 finding temperature of ideal gas
The book only has partial answer to the question, the temperature it gives is for constant volume not the pressure. I think I did it correctly.
- Mon Feb 10, 2020 9:49 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: 4C.3 finding temperature of ideal gas
- Replies: 1
- Views: 162
4C.3 finding temperature of ideal gas
I tried to solve the first part of this problem using Cp,m= 5/2 R and q= 765J and finding the change in temperature with q/(moles)(Cp,m) but I keep getting the final temperature as 343K when the book says 373 K.
- Mon Feb 10, 2020 8:24 pm
- Forum: Calculating Work of Expansion
- Topic: 4B.13 B finding moles
- Replies: 1
- Views: 156
4B.13 B finding moles
How do you find moles for the equation when temperature, pressure, and volume are given?
- Mon Feb 10, 2020 8:13 pm
- Forum: Calculating Work of Expansion
- Topic: Initial pressure of gas [ENDORSED]
- Replies: 1
- Views: 201
Initial pressure of gas [ENDORSED]
If the system has a given pressure and another pressure is acting on it doing work, do you ignore the systems initial pressure to solve for work? For reference, 4B.13 says calculate the work for a gas sample in a piston assembly with T=305 K, P=1.79 atm, and V=4.29 L: (a) irreversible expansion agai...
- Mon Feb 10, 2020 1:48 am
- Forum: Polyprotic Acids & Bases
- Topic: 6E.3 Ignoring second deprotonation
- Replies: 1
- Views: 347
6E.3 Ignoring second deprotonation
The question tells to calculate the pH of the diprotic acids and to ignore the second deprotonation if approximation is justified, how would you know to ignore or not?
- Sun Feb 09, 2020 7:49 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Finding concentration of H30 and OH from Kw
- Replies: 2
- Views: 321
Finding concentration of H30 and OH from Kw
Are Ka and Kb always equal when Kw is given? I don't know how else to solve for the concentration of H30+ and OH- when only the value of Kw was given. For reference, homework problem 6A.21
- Sun Feb 09, 2020 7:44 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: finding pH from concentration of H30+
- Replies: 2
- Views: 236
finding pH from concentration of H30+
If the log[H30+], without the negative sign, is already positive, is it wrong? For example, a concentration of 3.1 M results in a pH= -.49, using -log(3.1).
- Sun Feb 09, 2020 7:32 pm
- Forum: Applying Le Chatelier's Principle to Changes in Chemical & Physical Conditions
- Topic: Volume decrease
- Replies: 7
- Views: 289
Volume decrease
I thought the rule for volume decrease is that the side with less moles will be favored; however, in 5.39 the volume was halved and the reactant which has more moles in equation, I calculated it to have increased in concentration.
- Sun Feb 09, 2020 7:30 pm
- Forum: Ideal Gases
- Topic: Partial pressure plot
- Replies: 2
- Views: 180
Partial pressure plot
In 5.35 theres a partial pressure/ Kpa vs time graph and you have to figure out how to balance the equation as well as calculate K from it. How are we supposed to read this?
- Sun Feb 09, 2020 7:28 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Halogen equilibrium reaction
- Replies: 1
- Views: 106
Halogen equilibrium reaction
How is X2(g)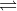 2X(g), where X is a halogen, affected by temperature? Does it favor reactant or product?
2X(g), where X is a halogen, affected by temperature? Does it favor reactant or product?
- Sun Feb 09, 2020 7:23 pm
- Forum: Ideal Gases
- Topic: Change in pressure
- Replies: 7
- Views: 525
Change in pressure
What is the rule for increasing or decreasing pressure and its affect on shifting the reaction?
- Sun Feb 09, 2020 7:22 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Change in concentration
- Replies: 2
- Views: 126
Change in concentration
Does increasing or decreasing the concentration of a product affect the concentration of another product? For example, for the reaction 4NH3(g) + 5O2(g) 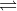 4NO(g) + 6H20(g), does increasing the concentration of NO affect the concentration of H20?
4NO(g) + 6H20(g), does increasing the concentration of NO affect the concentration of H20?
- Sun Feb 09, 2020 7:19 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Pressure change
- Replies: 3
- Views: 183
Pressure change
How does pressure increasing or decreasing change the concentration of products or reactants in equilibrium? Does a change in pressure for a reactant affect another reactant in the reaction?
- Sun Feb 09, 2020 7:18 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Hw 5I.23
- Replies: 1
- Views: 176
Hw 5I.23
Not sure what I am doing wrong, but in solving the concentrations using an ice table I am getting a larger change in concentration than what was initially there.
- Tue Feb 04, 2020 7:54 pm
- Forum: Calculating Work of Expansion
- Topic: Reversible and irreversible process
- Replies: 2
- Views: 132
Reversible and irreversible process
Can someone explain what this means in terms of expansion? How can work be irreversible?
- Wed Jan 29, 2020 11:11 pm
- Forum: Calculating Work of Expansion
- Topic: 4A.3
- Replies: 2
- Views: 144
4A.3
I cannot get the correct answer. I am using the area of a cylinder with .15 m as radius and .2 m as height times the height to get the volume times 2 atm as the pressure.
- Wed Jan 29, 2020 11:06 pm
- Forum: Concepts & Calculations Using First Law of Thermodynamics
- Topic: 4a.9
- Replies: 3
- Views: 285
4a.9
What formula is used to calculate the change in temperature from copper at 100 degrees C placed in water at 22 degrees C? I don't know how to begin this problem
- Wed Jan 22, 2020 9:59 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Hw I.15 ICE table
- Replies: 1
- Views: 144
Hw I.15 ICE table
How do you calculate the change in concentration when only the products are used in the reaction and the equilibrium concentration is given? Initial concentration of .2 M of NH3 with H2S and K= 1.6 x 10-4
- Wed Jan 22, 2020 6:16 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Homework 5H.1
- Replies: 1
- Views: 94
Homework 5H.1
How does K= 41 go to K= 1.7 x103 when the reaction has twice as many moles in reactants and products? For reference the original reaction is N2 + 3H2 = 2NH3 and K= 41 and the reaction is changed to 2N2 + 6H2 = 4NH3.
- Tue Jan 14, 2020 5:42 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: equilibrium concentration with gases
- Replies: 7
- Views: 248
equilibrium concentration with gases
If the reaction is homogeneous with gas phase but the question is in terms of concentration and gives the moles of the reactants and products, is pressure ignored?
- Sat Dec 07, 2019 9:03 pm
- Forum: Lewis Acids & Bases
- Topic: Lewis structure of acids and bases
- Replies: 2
- Views: 237
Re: Lewis structure of acids and bases
The filling of the octets is the most important aspect - double and triple bonds come in later. Double bonds should be used when both the central and attached atom has an extra lone pair each, and if the formal charge conditions are most favorable. I am not sure what you are talking about regarding...
- Sat Dec 07, 2019 8:42 pm
- Forum: Lewis Acids & Bases
- Topic: Lewis structure of acids and bases
- Replies: 2
- Views: 237
Lewis structure of acids and bases
When figuring out the lewis structure of an acid or base, should you stay away from double or triple bonds even if it stabilizes the molecule? Do the bonds make it have less acid/base characteristic. In all of the examples I've seen of finding if a molecule is an acid or base from the lewis structur...
- Sat Dec 07, 2019 8:27 pm
- Forum: Lewis Acids & Bases
- Topic: SO3 molecule as lewis acid
- Replies: 1
- Views: 210
SO3 molecule as lewis acid
The book draws SO3 (hw 6.5) with one double bond and two single bonds, but it is more unstable than SO3 having three double bonds. Does it have to do with SO3 acting as an acid or base?
- Sat Dec 07, 2019 5:01 pm
- Forum: Conjugate Acids & Bases
- Topic: Reaction arrows
- Replies: 3
- Views: 298
Reaction arrows
Do we need a reverse arrow in all acid and base chemical equations? 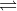
- Sat Dec 07, 2019 4:07 pm
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: CO3 2-
- Replies: 1
- Views: 90
CO3 2-
CO3 with a -2 charge is either a mono or bidentate ligand, according to the book. Is it possibly monodentate because the molecule is small?
- Sat Dec 07, 2019 3:56 pm
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: Ligand polydentate
- Replies: 4
- Views: 428
Ligand polydentate
How do you figure out how many sites a ligand can bind to on a metal?
- Sat Dec 07, 2019 2:31 pm
- Forum: Dipole Moments
- Topic: Lone pairs
- Replies: 6
- Views: 369
Re: Lone pairs
If the lone pairs are on opposite sides then they cancel out. An example of this would be XeF4 since it is square planar and all the dipole movements from the Fs and electron pairs are negated. So you need to know the molecular shape before figuring out if the lone pairs align to create a dipole mo...
- Sat Dec 07, 2019 2:29 pm
- Forum: Conjugate Acids & Bases
- Topic: Strength
- Replies: 2
- Views: 256
Strength
Will a strong acid have a strong conjugate base and the same for the opposite?
- Fri Dec 06, 2019 10:19 pm
- Forum: Bronsted Acids & Bases
- Topic: Strong vs. Weak
- Replies: 2
- Views: 260
Strong vs. Weak
If the product of an acidic reaction still has hydrogens in its molecule is it commonly a weak acid? Like acetic acid, CH3COOH after dissolving in water its left as CH3CO2-, whereas HCl is left as Cl-.
- Fri Dec 06, 2019 8:41 pm
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: what is a ligand?
- Replies: 7
- Views: 542
Re: what is a ligand?
In lecture, I'm pretty sure Lavelle said that CO was a ligand and I'm confused as to how that is so.
- Fri Dec 06, 2019 12:10 am
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: what is a ligand?
- Replies: 7
- Views: 542
what is a ligand?
Can someone explain what a ligand is and what it does?
- Fri Dec 06, 2019 12:08 am
- Forum: Resonance Structures
- Topic: localization
- Replies: 1
- Views: 237
localization
are all resonance structures delocalized electrons?
- Fri Dec 06, 2019 12:08 am
- Forum: Bond Lengths & Energies
- Topic: single bonds and s bonds [ENDORSED]
- Replies: 1
- Views: 218
single bonds and s bonds [ENDORSED]
How do you total in the strength in a bond when factoring how many bonds and the s- orbital characteristic of it? If I'm not mistaken having a hybridized orbital sp is stronger than sp2 but is a double bond stronger than a single bond of 2p-2p?
- Fri Dec 06, 2019 12:05 am
- Forum: Dipole Moments
- Topic: Lone pairs
- Replies: 6
- Views: 369
Lone pairs
How do you figure out if lone pairs align to create a dipole moment?
- Fri Dec 06, 2019 12:03 am
- Forum: Determining Molecular Shape (VSEPR)
- Topic: lone pairs
- Replies: 2
- Views: 136
lone pairs
How would you determine where the lone pairs on a central atom would be in a molecular shape? How would you know if they align to create a dipole moment or cancel eachother out?
- Thu Dec 05, 2019 11:59 pm
- Forum: Hybridization
- Topic: Determining Hybridization
- Replies: 4
- Views: 242
Re: Determining Hybridization
If the electrons are localized, then it's how many electron density regions it has. So like CH4, the carbon has 4 electron density regions, so it has sp3.
- Thu Dec 05, 2019 11:57 pm
- Forum: Hybridization
- Topic: Hybridization & Sigma and Pi Bonds
- Replies: 4
- Views: 315
Re: Hybridization & Sigma and Pi Bonds
Unhybridized orbitals are used to make pi bonds
- Thu Dec 05, 2019 11:56 pm
- Forum: Hybridization
- Topic: resonance structures
- Replies: 1
- Views: 144
resonance structures
Is it correct that resonance structures have unhybridized orbitals since the electrons are delocalized?
- Thu Dec 05, 2019 11:54 pm
- Forum: Hybridization
- Topic: s orbital [ENDORSED]
- Replies: 1
- Views: 171
s orbital [ENDORSED]
why is having a sp hybridization stronger than sp2?
- Thu Dec 05, 2019 11:53 pm
- Forum: Hybridization
- Topic: unhybridized orbitals
- Replies: 4
- Views: 174
unhybridized orbitals
Is it possible for a sigma bond to be unhybridized?
- Thu Dec 05, 2019 11:16 pm
- Forum: Hybridization
- Topic: Simple explanation
- Replies: 1
- Views: 202
Simple explanation
Can someone simply explain hybridization for me? Can't understand what it means and the 4 different hybrid orbitals.
- Wed Dec 04, 2019 9:58 pm
- Forum: Lewis Acids & Bases
- Topic: Ionic and compounds
- Replies: 1
- Views: 637
Ionic and compounds
How do you know when an acid and base reaction forms ions or compounds of eachother? Like Ca(OH)2 + 2HCN forms Ca(CN)2 + 2H2O but 2Na + 2OH- + H2S forms 2H2O + 2NA+ + S2-
- Wed Nov 20, 2019 11:44 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Name confusion
- Replies: 4
- Views: 303
Name confusion
How do you know when the shape is being asked for, for instance, seesaw or its asking for tetrahedral? What are the differences between those two different categories of naming the molecule shape? Sorry if this isn't clear, I just don't know if I should be predicting the molecule as t-shaped or trig...
- Wed Nov 20, 2019 10:53 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: Dipole-Dipole and induced dipole
- Replies: 4
- Views: 268
Re: Dipole-Dipole and induced dipole
You should look at the OVERALL characteristic. This molecule is polar overall because of the electronegativity difference between the C and Cl. Assuming that this molecule is interacting with only itself, dipole induced-dipole is not possible because it requires a non polar molecule which ClCH3 is ...
- Wed Nov 20, 2019 10:46 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: Strengths of forces
- Replies: 4
- Views: 305
Re: Strengths of forces
If you are referring to diethyl ether to having dipole-dipole intermolecular forces and water to have hydrogen bonding, then water (hydrogen bonding) would have a stronger intermolecular force because hydrogen bonds are stronger than dipole-dipole bonds. Thank you this is what I was asking! Do you ...
- Wed Nov 20, 2019 10:42 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: tetrahedral vs. trigonal pyramidal
- Replies: 4
- Views: 407
tetrahedral vs. trigonal pyramidal
Are tetrahedral and trigonal pyramidal the same?
- Wed Nov 20, 2019 10:29 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Bond Angles
- Replies: 6
- Views: 406
Bond Angles
Lone pairs on central atom cause the expected bond angles to be slightly less?
- Wed Nov 20, 2019 10:10 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Lone pairs
- Replies: 13
- Views: 766
Lone pairs
Do lone pairs not on the center atom account for shape? For example HCN
- Wed Nov 20, 2019 9:47 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: Strengths of forces
- Replies: 4
- Views: 305
Strengths of forces
Would hydrogen bonding (in water) be stronger than one dipole moment (diethyl ether)?
- Wed Nov 20, 2019 9:30 pm
- Forum: Dipole Moments
- Topic: Melting points
- Replies: 15
- Views: 1360
Melting points
Would a double bond create a higher melting point than a dipole moment?
- Wed Nov 20, 2019 9:10 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: 3F.5
- Replies: 4
- Views: 310
3F.5
How does butanol have a higher melting point than diethyl ether when they have the same atoms?
- Wed Nov 20, 2019 9:09 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: Hydrogen Bonding
- Replies: 2
- Views: 170
Re: Hydrogen Bonding
But how would you count the amount of hydrogen bonding sites?
- Wed Nov 20, 2019 8:43 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: Hydrogen Bonding
- Replies: 2
- Views: 170
Hydrogen Bonding
Are possible hydrogen bonding sites on every H and O,N,F electron lone pairs in a molecule? And the amount of lone pairs also tells how many hydrogen bonds could happen on an atom in a molecule?
- Tue Nov 19, 2019 8:02 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: Dipole-Dipole and induced dipole
- Replies: 4
- Views: 268
Dipole-Dipole and induced dipole
How do you separate the two different forces? How does ClCH3 have dipole-dipole interactions rather than dipole-induced dipole?
- Tue Nov 19, 2019 7:52 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: Hydrogen bonding
- Replies: 3
- Views: 172
Hydrogen bonding
Is hydrogen bonding only possible in a molecule with hydrogen already in it? Is it impossible for that type of interaction to occur on molecules containing O,N, or F with lone pairs that aren't already bonding to hydrogen?
- Tue Nov 05, 2019 6:50 pm
- Forum: Administrative Questions and Class Announcements
- Topic: DINO NUGGETS Review Session! Download Problems HERE [ENDORSED]
- Replies: 52
- Views: 6869
Re: DINO NUGGETS Review Session! Download Problems HERE [ENDORSED]
In the answer key for the mini dino nuggets, the dipole moments in question 2a are going towards the negative partial charge when in the textbook it says to go towards the positive charge. What would be correct?
- Sun Nov 03, 2019 6:40 pm
- Forum: Photoelectric Effect
- Topic: Photons emitted in a time interval
- Replies: 2
- Views: 172
Re: Photons emitted in a time interval
I forgot to times it by Avogadro's constant to find amount of photons. But this number should be multiplied by two?
- Sun Nov 03, 2019 6:38 pm
- Forum: Photoelectric Effect
- Topic: Photons emitted in a time interval
- Replies: 2
- Views: 172
Photons emitted in a time interval
For reference, I am doing 1B.9, which gives the wavelength of the light and watts of the light and I am stumped on how many photons are being emitted in 2 seconds. I used E=hv to find the energy of a singular photon and then I divided that by the watts which gave me the answer in the back of the boo...
- Sun Nov 03, 2019 6:13 pm
- Forum: Photoelectric Effect
- Topic: Energy per Photon
- Replies: 4
- Views: 264
Re: Energy per Photon
If the question is asking this but in a 2 second interval would you do the same but times it by 2?
- Sun Nov 03, 2019 5:42 pm
- Forum: Photoelectric Effect
- Topic: Energy of electron
- Replies: 2
- Views: 204
Energy of electron
1B.7 is asking for the energy emitted by a sodium atom when it generates a photon and for the energy of 5.00 mg of sodium emitting light. Do I think of the excited sodium atom as a photon in this problem? As in the formula E=hv applies to this question, not the kinetic energy of an electron. Then, I...
- Sun Nov 03, 2019 5:17 pm
- Forum: Photoelectric Effect
- Topic: Work function of an atom
- Replies: 4
- Views: 253
Re: Work function of an atom
What is the question asking for?Brittney Hun 1K wrote:So I used two different equations to derive the energy because it's not asking for the energy of an electron, which is what the kinetic energy formula finds. I used E=hv, but found v first using v=c/lamda for part a.
- Sun Nov 03, 2019 5:16 pm
- Forum: Photoelectric Effect
- Topic: Work function of an atom
- Replies: 4
- Views: 253
Re: Work function of an atom
Never mind I know that doesn't work because the v in that formula is for the speed of the electron. Is the work function of the atom supposed to be given to find this if only wavelength of light is provided? I should be using (hc)/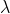 -
- 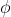 = Ek
= Ek
- Sun Nov 03, 2019 5:02 pm
- Forum: Photoelectric Effect
- Topic: Work function of an atom
- Replies: 4
- Views: 253
Work function of an atom
If a problem (1B.7) is asking for the energy emitted by an atom when it generates a photon and the wavelength of the photon ejected is given, would I use the Ek= 1/2 me x v2 formula?
- Sat Nov 02, 2019 9:31 pm
- Forum: Empirical & Molecular Formulas
- Topic: Formula Units
- Replies: 2
- Views: 356
Formula Units
What is being asked by finding the formula units and number of molecules from moles of a molecule? For reference Fundamentals E.21 asks to calculate the amount of moles, amount of molecules and formula units from 10 g of Al2O3.
- Sat Nov 02, 2019 8:45 pm
- Forum: Accuracy, Precision, Mole, Other Definitions
- Topic: Atoms to Moles
- Replies: 7
- Views: 452
Atoms to Moles
Is it correct to divide the amount of atoms in a sample by the molar constant to find the amount of moles?
- Tue Oct 29, 2019 2:59 pm
- Forum: Ionic & Covalent Bonds
- Topic: Homework 2A.9
- Replies: 2
- Views: 104
Re: Homework 2A.9
Thank you! So this question is asking for what possible ions follow those configurations and have a 2+ charge?
- Mon Oct 28, 2019 7:28 pm
- Forum: Ionic & Covalent Bonds
- Topic: Homework 2A.9
- Replies: 2
- Views: 104
Homework 2A.9
The question asks which M2+ ions are expected to have the following ground-state electron configurations: a) [Ar]3d7 b) [Ar]3d6
I'm confused by what the question is asking and how the electron configuration can change for this ion?
I'm confused by what the question is asking and how the electron configuration can change for this ion?
- Mon Oct 21, 2019 10:16 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Week 4 homework problems
- Replies: 5
- Views: 372
Re: Week 4 homework problems
205296774 wrote:Our homework and units aren't broken up by week. The quantum unit is multiple weeks long so the page with the key concepts and hw problems apply to this week, too.
Hope this helps!
Did Lavelle say that we can use chemical bond problems for this weeks homework?
- Mon Oct 21, 2019 10:11 pm
- Forum: Lewis Structures
- Topic: Octet
- Replies: 2
- Views: 935
Re: Octet
The octet rule is violated in compounds with more than eight electrons assigned to their valence shell. These are called expanded valence shell molecules. Such compounds are formed only by central atoms in the third row of the periodic table and beyond that have empty d orbitals in their valence sh...
- Mon Oct 21, 2019 10:09 pm
- Forum: Lewis Structures
- Topic: Homework 2B.3 d
- Replies: 2
- Views: 122
Homework 2B.3 d
How do you figure out the lewis structure of BrF3 when they share 24 valence electrons?
- Mon Oct 21, 2019 9:54 pm
- Forum: Electron Configurations for Multi-Electron Atoms
- Topic: Ground state electron configuration of ions
- Replies: 4
- Views: 271
Ground state electron configuration of ions
In the homework it asks for the ground state electron configuration of ions, does ground state mean that the ions need to be changed to a neutral atom?
- Wed Oct 09, 2019 9:23 pm
- Forum: Molarity, Solutions, Dilutions
- Topic: Fundamentals G
- Replies: 4
- Views: 595
Fundamentals G
In the second part of G.17 it asks for the mass of CuSO4 5H20 that must be used to prepare 250 mL of .2 M CuSO4(aq). I am very confused by what to do with the water molecule in this problem, I don't know how to start solving this.
- Wed Oct 09, 2019 9:14 pm
- Forum: Molarity, Solutions, Dilutions
- Topic: Fundamentals G13
- Replies: 6
- Views: 2794
Re: Fundamentals G13
I am having trouble with this concept as well, the amount of individual moles in a molecule has the same ratio as moles of the molecule? So 2 nitrogen moles are always in 1 mole of NH4NO3?
- Wed Oct 09, 2019 7:38 pm
- Forum: Molarity, Solutions, Dilutions
- Topic: Mole ratios/ G.5 hw
- Replies: 2
- Views: 301
Mole ratios/ G.5 hw
In homework problem G.5 the question says there is 2.111 g of sodium carbonate (Na 2 CO 3 ) and you have to find the molarity in a solution, is it possible to find the mole ratio of the molecule and apply it to the elements? The molar mass of sodium carbonate is 105.81 gmol -1 , so in a sample of 2....
- Wed Oct 02, 2019 9:45 pm
- Forum: SI Units, Unit Conversions
- Topic: Sig figs
- Replies: 18
- Views: 693
Re: Sig figs
No, the number of sig figs can vary because it is based on the least amount of sig figs present in the problem. However, any conversion factors do not count when determining the number of sig figs. Thank you this helped a lot. I was looking for the homework question to mention how many sig figs to ...
- Wed Oct 02, 2019 9:40 pm
- Forum: Limiting Reactant Calculations
- Topic: L1 homework
- Replies: 2
- Views: 63
L1 homework
The question asks how much can Br2 can be produced from .30 moles of ClO2 when the balanced reaction is given. Is it simple enough to assume that 6 moles of ClO2 produces one mole of Br2 so .30/6 would be the amount of Br2 in moles produced?
- Wed Oct 02, 2019 8:20 pm
- Forum: SI Units, Unit Conversions
- Topic: Sig figs
- Replies: 18
- Views: 693
Re: Sig figs
If the question doesn't mention sig figs, should I not worry about the precision of the answer then?
- Wed Oct 02, 2019 7:58 pm
- Forum: Accuracy, Precision, Mole, Other Definitions
- Topic: E.7 calculate atoms in moles
- Replies: 4
- Views: 207
E.7 calculate atoms in moles
The question gives how many carbon atoms are in a molecule of DNA and to calculate in moles how many atoms of carbon there are. Is this asking to divide the number of atoms by moles to be in terms of mole or to multiple the amount of carbon by Avogadro's constant to find the true amount of atoms? So...
- Wed Oct 02, 2019 7:47 pm
- Forum: SI Units, Unit Conversions
- Topic: Sig figs
- Replies: 18
- Views: 693
Sig figs
For the homework are we supposed to be using a set significant figure number?

