Search found 149 matches
- Fri Mar 12, 2021 6:51 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Resources for Final?
- Replies: 5
- Views: 351
Re: Resources for Final?
I think I'll just skim over textbook chapters which is what I've done for the midterms and it seems to have worked so far (in addition to practice problems)
- Fri Mar 12, 2021 6:50 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Intermediates in overall rate law
- Replies: 3
- Views: 372
Re: Intermediates in overall rate law
I don't think you're supposed to ever have intermediates in the overall rate law. If the slow step ends up having one we usually do the pre-equilibrium method to get rid of it. I don't know what the case would be if we explicitly know the previous steps isn't fast at equilibrium. Also, the UAs have ...
- Fri Mar 12, 2021 6:47 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Sapling Week 9/10 #13
- Replies: 6
- Views: 388
Re: Sapling Week 9/10 #13
others have explained it in detail but yeah you need to do the pre-equilibrium method
- Fri Mar 12, 2021 3:15 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Calculating standard cell potentials
- Replies: 4
- Views: 437
Re: Calculating standard cell potentials
They both give the same answer!
- Fri Mar 12, 2021 3:13 pm
- Forum: Arrhenius Equation, Activation Energies, Catalysts
- Topic: Textbook 7.1
- Replies: 2
- Views: 195
Re: Textbook 7.1
I don't think temperature changes activation energy, it just increases the rate.
- Fri Mar 12, 2021 3:08 pm
- Forum: Arrhenius Equation, Activation Energies, Catalysts
- Topic: Sapling HW Week 9/10 #17
- Replies: 9
- Views: 535
Re: Sapling HW Week 9/10 #17
It helps to draw the diagram out. This is an exothermic reaction which you know from the enthalpy so you'll draw the diagram going from high to low. The difference between the products and reactants is a magnitude of 49. So this is from one end to another. However, the middle has a little hump you n...
- Fri Mar 12, 2021 3:04 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: textbook 4E. 5
- Replies: 2
- Views: 333
Re: textbook 4E. 5
Yes this structure has resonance (I think it is benzene). So instead of forming 3 double and 3 single bonds you're actually forming 6 mixed bonds. This is assuming you're breaking all the bonds first and then reforming everything again (which is the method I prefer)
- Sun Mar 07, 2021 6:15 pm
- Forum: Balancing Redox Reactions
- Topic: Resources for Redox Reactions
- Replies: 6
- Views: 467
Re: Resources for Redox Reactions
Yeah I've been struggling with them too. I think I'll just try to watch some videos and do practice problems to get the hang of it.
- Sun Mar 07, 2021 6:13 pm
- Forum: General Rate Laws
- Topic: Deriving Equations
- Replies: 6
- Views: 442
Re: Deriving Equations
I think it was just to understand since we spend most of our time doing calculations but it might be helpful for conceptual type questions
- Sun Mar 07, 2021 6:11 pm
- Forum: Student Social/Study Group
- Topic: Anxiety
- Replies: 109
- Views: 9955
Re: Anxiety
Just realizing that I'm trying my best and that one grade during such a difficult doesn't define me! We'll get through this, we're almost done! Take some breaks, remember to eat and sleep, take some walks and watch tv when it gets too overwhelming. You got this :))
- Sun Mar 07, 2021 5:56 pm
- Forum: Administrative Questions and Class Announcements
- Topic: [CHEM 14B KARAOKE]
- Replies: 68
- Views: 7447
Re: [CHEM 14B KARAOKE]
glad you guys were able to put this event on!
- Sun Mar 07, 2021 5:53 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Constants and Equations Sheet
- Replies: 6
- Views: 412
Re: Constants and Equations Sheet
no new ones have been added as far as I know
- Sun Mar 07, 2021 5:52 pm
- Forum: Student Social/Study Group
- Topic: How do you deal with burnout?
- Replies: 144
- Views: 22125
Re: How do you deal with burnout?
I am currently not dealing with it LMAO
- Sun Feb 28, 2021 9:08 pm
- Forum: Work, Gibbs Free Energy, Cell (Redox) Potentials
- Topic: Sapling Week 7/8 question 15
- Replies: 6
- Views: 441
Re: Sapling Week 7/8 question 15
I think you use Nernst equation. So you need to find the Enaught and then plug everything in to the equation. For Q you would do .761 on top and then .01 in the denominator.
- Sun Feb 28, 2021 8:31 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Sapling week 7/8 number 15
- Replies: 3
- Views: 281
Re: Sapling week 7/8 number 15
You use the Nernst Equation so you are given products and reactants molarity for Q. T= 298 and R=8.314. n is the mol of electrons transferred and then F is a constant. Pretty much all you need to calculate is E naught from the table and plug everything into the equation. To calculate E naught do the...
- Sun Feb 28, 2021 7:43 pm
- Forum: Work, Gibbs Free Energy, Cell (Redox) Potentials
- Topic: Sapling Week 7/8 #7
- Replies: 10
- Views: 667
Re: Sapling Week 7/8 #7
does the order of the shorthand notation matter?
- Sun Feb 28, 2021 6:09 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Sapling #12
- Replies: 3
- Views: 276
Re: Sapling #12
you might be forgetting to convert to kJ. That's what was happening to me and it took so many tries for me to figure that out.
- Sun Feb 28, 2021 5:31 pm
- Forum: Work, Gibbs Free Energy, Cell (Redox) Potentials
- Topic: Sapling #12
- Replies: 4
- Views: 293
Re: Sapling #12
I was stuck on this one too so thanks!
- Sun Feb 28, 2021 11:11 am
- Forum: Calculating Work of Expansion
- Topic: Finding Reversibility
- Replies: 8
- Views: 560
Re: Finding Reversibility
doesn't isothermal mean irreversible or is that not always the case?
- Sun Feb 21, 2021 3:33 pm
- Forum: Student Social/Study Group
- Topic: points needed for c instead of c minus
- Replies: 7
- Views: 869
Re: points needed for c instead of c minus
I think you can just divide the number of points earned by the total points possible to find out right? Since it is a straight scale
- Sun Feb 21, 2021 3:31 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: Calibrating Calorimeter
- Replies: 6
- Views: 2886
Re: Calibrating Calorimeter
I think it is to find the heat capacity of the calorimeter so that you can use it to find the heat released in another reaction
- Sun Feb 21, 2021 3:23 pm
- Forum: Concepts & Calculations Using First Law of Thermodynamics
- Topic: Average Kinetic Energy
- Replies: 5
- Views: 512
Re: Average Kinetic Energy
I didn't understand this problem either so I just skipped it
- Sun Feb 21, 2021 3:22 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Tips for Concentrating during a test
- Replies: 31
- Views: 1523
Re: Tips for Concentrating during a test
Try studying in a similar environment as your exams and build up your concentration by using timers
- Sun Feb 21, 2021 3:07 pm
- Forum: Student Social/Study Group
- Topic: Post Midterm 2 De-stressing
- Replies: 92
- Views: 9100
Re: Post Midterm 2 De-stressing
I have to cram for my 7B midterm on Monday but I have been binging episodes of Lucifer in between study sessions
- Sun Feb 14, 2021 7:19 pm
- Forum: Entropy Changes Due to Changes in Volume and Temperature
- Topic: Sapling Learning Week 5 and 6 Homework Question 7
- Replies: 4
- Views: 1472
Re: Sapling Learning Week 5 and 6 Homework Question 7
I was also stuck on this so thank you!
- Sun Feb 14, 2021 7:11 pm
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: Units
- Replies: 20
- Views: 828
Re: Units
Yeah it's a bit confusing with all the different units going on. I just write out all the units when doing calculations so I can check if they are crossing out.
- Sun Feb 14, 2021 3:08 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Midterm Material
- Replies: 10
- Views: 585
Re: Midterm Material
Jalyn Davis 3D wrote:Sana Nagori 2H wrote:Was friday's lecture part of outline 4 stuff?
Based on Lavelle's email from yesterday, it seems like Friday's lecture was the end of outline 4, so it is the last lecture that includes midterm content.
ah ok thank you! Guess I actually have to watch it now ://
- Sun Feb 14, 2021 3:07 pm
- Forum: Entropy Changes Due to Changes in Volume and Temperature
- Topic: Entropy Equations
- Replies: 3
- Views: 140
Re: Entropy Equations
Same, it seems like there are so many equations to keep track of
- Sun Feb 14, 2021 3:06 pm
- Forum: Thermodynamic Systems (Open, Closed, Isolated)
- Topic: Temperature and K
- Replies: 21
- Views: 1258
Re: Temperature and K
K as in Kelvin?
- Sun Feb 14, 2021 3:05 pm
- Forum: Student Social/Study Group
- Topic: Fav Shows of the Moment
- Replies: 115
- Views: 57844
Re: Fav Shows of the Moment
I'm currently watching Lucifer and started Supernatural! I'm also looking forward to watching World War 2 in Color once midterms end!
- Sun Feb 14, 2021 3:00 pm
- Forum: Student Social/Study Group
- Topic: Fav Shows of the Moment
- Replies: 115
- Views: 57844
Re: Fav Shows of the Moment
Namratha Gujje wrote:I have been watching Teen Wolf which honestly is a really good show. I wasn't expecting it to be this good when I first started it. 10/10 would recommend.
I was obsessed with teen wolf in middle school. I miss those days
- Sun Feb 14, 2021 2:58 pm
- Forum: Student Social/Study Group
- Topic: Classes for next quarter?
- Replies: 165
- Views: 21137
Re: Classes for next quarter?
Ls7c, maybe chem 14c and maybe ls23l. I don't feel like taking any classes tbh
- Sun Feb 14, 2021 2:57 pm
- Forum: Student Social/Study Group
- Topic: Balance / Self Care Tips
- Replies: 62
- Views: 4185
Re: Balance / Self Care Tips
Personally doing an absolutely poor job of it. It was much easier to do with in-person classes because there was a clear distinction between classes and your free time. I just try to do my relaxing during the day when I'm most unproductive anyway and then study at night when I feel more awake.
- Sun Feb 14, 2021 2:55 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Midterm Material
- Replies: 10
- Views: 585
Re: Midterm Material
Was friday's lecture part of outline 4 stuff?
- Sun Feb 14, 2021 2:54 pm
- Forum: Administrative Questions and Class Announcements
- Topic: struggling
- Replies: 73
- Views: 4707
Re: struggling
Maybe you need to spend some time focusing on the concepts and understanding what is truly happening. I think it makes approaching problems much easier and is especially helpful if you're stuck on a problem.
- Sun Feb 07, 2021 8:18 pm
- Forum: Student Social/Study Group
- Topic: Midterm 2 study tips
- Replies: 26
- Views: 1545
Re: Midterm 2 study tips
Janna Shakiba wrote:
When you say outline questions, are you referring to the problems listed in the syllabus?
Yes! They are on the syllabus and also at the top of the outlines that the professor has on his website. It's just the relevant questions in the textbook.
- Sun Feb 07, 2021 8:16 pm
- Forum: Student Social/Study Group
- Topic: Studying When You Don't Want to
- Replies: 48
- Views: 3651
Re: Studying When You Don't Want to
This may seem counterintuitive, but sometimes just taking a day off is the best thing you can do for yourself. There are days where I know even if I force myself to study, I won't actually retain anything. I take those days to recharge, do something to de-stress and hopefully bounce back the next da...
- Sun Feb 07, 2021 7:51 pm
- Forum: Student Social/Study Group
- Topic: Midterm 2 study tips
- Replies: 26
- Views: 1545
Re: Midterm 2 study tips
As most people will say, the textbook and the outline questions are your best friend. I haven't had time to go to any UA sessions this quarter, which are good extra practice, but even doing the textbook problems made me feel better about the material!
- Sun Feb 07, 2021 7:49 pm
- Forum: Student Social/Study Group
- Topic: Book Recommendations
- Replies: 135
- Views: 20550
Re: Book Recommendations
A Thousand Splendid Suns is one of my favorites but maybe not the happiest read. Station Eleven is also a cool book, especially since it's set in a post-pandemic world.
- Sun Feb 07, 2021 7:44 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: Sapling Week 3/4 #13
- Replies: 8
- Views: 524
Re: Sapling Week 3/4 #13
One mistake I was making was that I didn't realize there were solids as the reactants. There will be work done if the products have more moles of gas and since the reaction goes from 0 to 1 mole of gas then it is doing work.
- Sun Feb 07, 2021 7:43 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Midterm 2
- Replies: 33
- Views: 2515
Re: Midterm 2
I'm still mentally on midterm 1
- Sun Jan 31, 2021 10:22 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Reflecting on the midterm
- Replies: 17
- Views: 880
Re: Reflecting on the midterm
This was my exact same problem. Last quarter I finished the exams at least 10 minutes early but I guess the content this quarter is a little more calculation heavy. I guess we just need to get more practice!
- Sun Jan 31, 2021 6:56 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: Figuring out bond enthalpy
- Replies: 11
- Views: 543
Re: Figuring out bond enthalpy
pretty sure you can yup
- Sun Jan 31, 2021 6:55 pm
- Forum: Applying Le Chatelier's Principle to Changes in Chemical & Physical Conditions
- Topic: Adding a gas
- Replies: 26
- Views: 1267
Re: Adding a gas
It's an inert gas so it won't be taken into account for Le Chateliers principle
- Sun Jan 31, 2021 6:53 pm
- Forum: Thermodynamic Systems (Open, Closed, Isolated)
- Topic: R Constant
- Replies: 91
- Views: 8286
Re: R Constant
writing out your units is always helpful in calculations so you can keep track of what cancels out and whether you end up with the correct units needed for you answer.
- Sun Jan 31, 2021 6:52 pm
- Forum: Student Social/Study Group
- Topic: Spring 2021
- Replies: 106
- Views: 22351
Re: Spring 2021
I'm probably taking it in fall or even later
- Sun Jan 31, 2021 6:50 pm
- Forum: Student Social/Study Group
- Topic: Midterm Scores
- Replies: 25
- Views: 1075
Re: Midterm Scores
It takes about a week to two to get them out usually.
- Sun Jan 24, 2021 5:21 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: [B]formal
- Replies: 5
- Views: 378
Re: [B]formal
I was confused by this too! I just used the Kb equation and ended up solving for [B]
- Sun Jan 24, 2021 5:19 pm
- Forum: Student Social/Study Group
- Topic: CHEM 14B Midterm 1 Notes
- Replies: 23
- Views: 1306
Re: CHEM 14B Midterm 1 Notes
That's so nice thanks!
- Sun Jan 24, 2021 5:18 pm
- Forum: Administrative Questions and Class Announcements
- Topic: midterm
- Replies: 11
- Views: 688
Re: midterm
Janna Shakiba wrote:So we will need 2 devices to take the midterm like last quarter?
yes pretty much. Most people take the exam on their laptop and then use their phones for the zoom
- Sun Jan 24, 2021 5:16 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Textbook Problems Not Listed in Syllabus
- Replies: 3
- Views: 300
Re: Textbook Problems Not Listed in Syllabus
No I don't look at the unassigned ones. The ones on the outline seemed to be good enough for 14A
- Sun Jan 24, 2021 5:15 pm
- Forum: General Science Questions
- Topic: Studying
- Replies: 67
- Views: 4396
Re: Studying
As most people said, I found doing the textbook/outline questions at least twice helped me the most and then looking up random practice problems online for extra practice
- Sun Jan 17, 2021 8:42 pm
- Forum: Student Social/Study Group
- Topic: Exercising Our Minds and Bodies
- Replies: 120
- Views: 30159
Re: Exercising Our Minds and Bodies
I've been doing such a poor job of exercising my body but these are all great ideas!
- Sun Jan 17, 2021 8:41 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Sapling Homework #5
- Replies: 9
- Views: 407
Re: Sapling Homework #5
Hi! For this one, I would first figure out which equations that were given will give us the reaction at the top. I would focus on getting all of the species on the correct side of the equation to match the one at the top. For example, I would flip N2 + 3H2 <-> 2NH3 so that the species are on the sa...
- Sun Jan 17, 2021 8:22 pm
- Forum: Non-Equilibrium Conditions & The Reaction Quotient
- Topic: Endothermic vs Exothermic
- Replies: 7
- Views: 418
Re: Endothermic vs Exothermic
I think delta H is what we look at because it tells us whether the heat of the system increased or decreased with the reaction
- Sun Jan 17, 2021 7:38 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Sapling #4
- Replies: 6
- Views: 266
Re: Sapling #4
For this problem, I set up an ICE box and used it to find x. The initial pressure of PCl5 would be 0.0370 bar and the change would be -x, while the other initial pressures would be 0, and the change would be +x. After using Kp and finding x, you would calculate the pressures at equilibrium and add ...
- Sun Jan 17, 2021 7:37 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Sapling Homework #5
- Replies: 9
- Views: 407
Re: Sapling Homework #5
I'm also kind of stuck on this one. Should we be adding/subtracting these equations and using the inverse K value when possible?
- Sun Jan 17, 2021 7:35 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Sapling Week 1 #3
- Replies: 9
- Views: 387
Re: Sapling Week 1 #3
Kaiya_PT_1H wrote:Not sure if this helps, but I spent a long time on this trying to do the quadratic formula when you can just square root the Kc formula and the value for Kc to solve for x.
I wish I saw this beforehand. I also did the whole quadratic formula out haha
- Sun Jan 17, 2021 7:32 pm
- Forum: Student Social/Study Group
- Topic: Study Strategies
- Replies: 31
- Views: 1519
Re: Study Strategies
I find textbook problems and youtube videos help me the most
- Sun Jan 10, 2021 9:28 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Week 1 Assignment
- Replies: 17
- Views: 853
Re: Week 1 Assignment
Sapling is our graded homework but it's only graded on completion! The textbook problems given in the outlines are the ones for extra practice and are optional
- Sun Jan 10, 2021 9:26 pm
- Forum: Ideal Gases
- Topic: n/V = concentration
- Replies: 19
- Views: 2588
Re: n/V = concentration
It's just rearranging the equation and realizing that molarity=concentration= mol/L
- Sun Jan 10, 2021 9:24 pm
- Forum: Ideal Gases
- Topic: Reviewing PV=NRT
- Replies: 7
- Views: 409
Re: Reviewing PV=NRT
605291562 wrote:On sapling you can find a pdf version you can download which is probably easiest, they also have all the odd answers in the back of the book.
Definitely suggest downloading the pdf if you plan on doing a lot of practice problems.
- Sun Jan 10, 2021 9:22 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Substances for Calculating K
- Replies: 4
- Views: 164
Re: Substances for Calculating K
Also he showed that in aqueous solutions even if you included H20 in both the reactants and products it would cancel out.
- Sun Jan 10, 2021 2:15 am
- Forum: Student Social/Study Group
- Topic: Study Habits
- Replies: 96
- Views: 8476
Re: Study Habits
Practice problems are the best preparation! Also on days you're lacking motivation I highly suggest going to UA sessions. You end up getting some extra practice and it isn't as painful as forcing yourself to stare at textbook problems for hours.
- Sun Jan 10, 2021 2:11 am
- Forum: Administrative Questions and Class Announcements
- Topic: Chemical Principles through Bookshelf
- Replies: 3
- Views: 164
Re: Chemical Principles through Bookshelf
There is also a free online pdf that you can download. I find it easier to navigate.
- Tue Dec 15, 2020 3:57 pm
- Forum: Amphoteric Compounds
- Topic: Is Silicon an amphoteric element?
- Replies: 5
- Views: 661
Re: Is Silicon an amphoteric element?
I don't think it is!
- Tue Dec 15, 2020 3:55 pm
- Forum: Lewis Acids & Bases
- Topic: Classifying Other
- Replies: 4
- Views: 249
Re: Classifying Other
I think like NaCl would be other since it is not an acid or base
- Tue Dec 15, 2020 3:52 pm
- Forum: Lewis Acids & Bases
- Topic: Strong Lewis Bases?
- Replies: 3
- Views: 387
Re: Strong Lewis Bases?
Oxides and Hydroxides of group 1 and 2 elements!
- Tue Dec 15, 2020 3:51 pm
- Forum: Amphoteric Compounds
- Topic: Glycine Amphoteric
- Replies: 3
- Views: 322
Re: Glycine Amphoteric
Racquel Fox 3L wrote:
Why would N want a +1 charge thought?
It's not that N would WANT a +1 charge but more that oxygen really doesn't want one.
- Tue Dec 15, 2020 3:50 pm
- Forum: Student Social/Study Group
- Topic: How are you?
- Replies: 154
- Views: 18410
Re: How are you?
Just finished my last exam so that's a big relief! Now I only have an essay left.
- Sat Dec 12, 2020 8:13 pm
- Forum: Properties & Structures of Inorganic & Organic Acids
- Topic: Stronger acids and stability?
- Replies: 5
- Views: 2328
Re: Stronger acids and stability?
stronger acids are more stable after they have lost their H+. If a molecule loses a proton and is stable it is less likely to want to pick one up again and therefore will stay completely deprotenated so that's why a stronger acid will have a more stable conjugate base!
- Sat Dec 12, 2020 8:10 pm
- Forum: Student Social/Study Group
- Topic: Study music
- Replies: 59
- Views: 7523
Re: Study music
this might just be me but I actually like listening to songs with words in them. Usually I turn them up loud enough where it just becomes background noise and I don't get distracted but it sounds counterintuitive.
- Sat Dec 12, 2020 8:09 pm
- Forum: Acidity & Basicity Constants and The Conjugate Seesaw
- Topic: 6C. 17
- Replies: 3
- Views: 193
Re: 6C. 17
I think it's because bases with Nitrogen are weak bases generally. Also the minus charge on BrO- would really want to form a bond with a hydrogen because it's an ion and wants to get rid of that charge and that is why it's the stronger base
- Sat Dec 12, 2020 8:07 pm
- Forum: Bond Lengths & Energies
- Topic: hydrogen bonding vs ion-dipole
- Replies: 5
- Views: 2787
Re: hydrogen bonding vs ion-dipole
I think ion-dipole is stronger than H-bonds
- Sat Dec 12, 2020 8:05 pm
- Forum: Identifying Acidic & Basic Salts
- Topic: 12.65 (e) and (f)
- Replies: 5
- Views: 1832
Re: 12.65 (e) and (f)
I don't know about the hydrates part but for determining the pH, the positive charge on the TM cations would want to bond with the partially negative oxygen atom of water thus weakening a O-H bond and releasing a H+ so that is why the pH<7
- Sat Dec 12, 2020 3:04 pm
- Forum: Amphoteric Compounds
- Topic: Glycine Amphoteric
- Replies: 3
- Views: 322
Re: Glycine Amphoteric
I think because if oxygen donated a pair it would end up with a positive formal charge and oxygens do not want that.
- Sat Dec 12, 2020 3:02 pm
- Forum: Calculating pH or pOH for Strong & Weak Acids & Bases
- Topic: Sapling HW 10 Question#5
- Replies: 3
- Views: 227
Re: Sapling HW 10 Question#5
do we have to know Kw?
- Sat Dec 12, 2020 3:01 pm
- Forum: Lewis Acids & Bases
- Topic: Textbook Question 6.5
- Replies: 2
- Views: 118
Re: Textbook Question 6.5
I kinda just thought about it where usually nonmetal oxides are acids and that's why SO3 is the acid but I'm not sure if that's correct
- Thu Dec 10, 2020 8:32 am
- Forum: Administrative Questions and Class Announcements
- Topic: Matthew's Workshop - Week 10
- Replies: 2
- Views: 977
Re: Matthew's Workshop - Week 10
Thank you! The workshop was really helpful!
- Sun Dec 06, 2020 6:54 pm
- Forum: *Molecular Orbital Theory (Bond Order, Diamagnetism, Paramagnetism)
- Topic: Sigma and Pi bond rotation
- Replies: 5
- Views: 1200
Re: Sigma and Pi bond rotation
single bonds are always sigma bonds so they can rotate. However, a double bond has an addition pi bond which limits the rotation and essentially locks the sigma bond in place because they are at 90 degrees to each other (I think). This is why a double and triple bond would not be able to rotate.
Re: Potassium
when naming, the cations outside the bracket like Na or K wouldn't have prefixes and the same with anions like Cl on the outside of brackets. They would just be potassium or chloride.
Re: Ferrate
yes! Copper, silver, and gold off the top of my head also have exceptions
- Sun Dec 06, 2020 6:41 pm
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: Polydentate Ligands We Have to Know
- Replies: 3
- Views: 210
Re: Polydentate Ligands We Have to Know
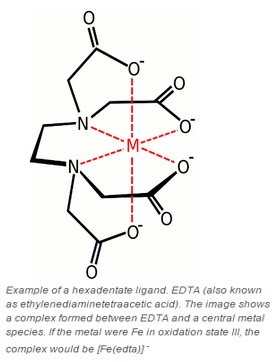
There is a list on the professors website that has the names of the ligands. However, it doesn't specify the number of bonding sites for each. From what I've heard, the main polydentate ions to know are oxalato (2), carbonato (1 or 2), en (2), dien (3), and edta (6). Someone else can let me know if ...
Re: -ate
yes you have to check if the TM compound has an overall negative charge. It's easy in these questions because it is just the anion but sometimes when there is a cation (like Na or K) present the negative charge isn't written and you have to look at the two components separately to realize the TM com...
- Sun Dec 06, 2020 6:38 pm
- Forum: Naming
- Topic: Naming [Co(CN)5(OH2)]2-
- Replies: 5
- Views: 522
Re: Naming [Co(CN)5(OH2)]2-
some ligands have special names when naming coordination compounds. Water is aqua, not hydro. Aqua comes first alphabetically which is why aqua comes first and then pentacyano. Also, since the compound has an overall negative charge, the TM is named with a "ate" ending- cobaltate- to signi...
- Fri Nov 27, 2020 9:29 pm
- Forum: Lewis Structures
- Topic: Ring structures
- Replies: 4
- Views: 173
Re: Ring structures
CesarLec1 wrote:I believe that for our purposes, when we see a molecule with 6 carbon atoms, it will most likely be in a ring structure
Yeah usually when there aren't enough Hydrogens to fill out a zigzag structure I try to fit it into a ring structure.
- Fri Nov 27, 2020 9:27 pm
- Forum: Sigma & Pi Bonds
- Topic: Question on Sigma Bonds and Polarity
- Replies: 5
- Views: 365
Re: Question on Sigma Bonds and Polarity
Maybe someone out there has a more definite answer
- Fri Nov 27, 2020 9:27 pm
- Forum: Sigma & Pi Bonds
- Topic: Question on Sigma Bonds and Polarity
- Replies: 5
- Views: 365
Re: Question on Sigma Bonds and Polarity
This is actually an interesting thought. Maybe they can briefly cancel but overall it will remain what it dominantly is.
- Fri Nov 27, 2020 9:13 pm
- Forum: Sigma & Pi Bonds
- Topic: Delocalized Pi Bond
- Replies: 5
- Views: 979
Re: Delocalized Pi Bond
When a molecule has resonance where a double/triple bond can be found in multiple locations, the electrons in those bonds are delocalized. If a molecule has resonance, the pi bonds making up those double/triple bonds are delocalized.
- Fri Nov 27, 2020 9:09 pm
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: Chelating Ligands
- Replies: 4
- Views: 300
Re: Chelating Ligands
Marcus Lagman 1B wrote:Hello!
I do think you are correct! Here is an image of where the bonding sites would occur:
this is a really helpful image!
- Fri Nov 27, 2020 9:05 pm
- Forum: Hybridization
- Topic: Bound Atoms
- Replies: 5
- Views: 165
Re: Bound Atoms
If two atoms are "bound", they have a bond between them. If this bond is a sigma bond (a single bond), then the atom can rotate freely. Once the bonds become double or triple, there are pi bonds that inhibit this freedom and the atoms can no longer rotate from the plane they are in.
- Sat Nov 21, 2020 9:55 pm
- Forum: Ionic & Covalent Bonds
- Topic: Polar/ Nonpolar
- Replies: 25
- Views: 1406
Re: Polar/ Nonpolar
Yes but it still needs to have dipole moments. Molecules with only H-C bonds will never have a significant difference in electronegativity so no matter the shape they will never be polar.
- Sat Nov 21, 2020 9:52 pm
- Forum: Sigma & Pi Bonds
- Topic: 2F 19
- Replies: 3
- Views: 286
Re: 2F 19
I haven't caught up on all the lectures so I don't know what it means in this context but orthogonal just means perpendicular.
- Sat Nov 21, 2020 9:46 pm
- Forum: Student Social/Study Group
- Topic: Study Tips for Final Exam
- Replies: 57
- Views: 3446
Re: Study Tips for Final Exam
I think I will probably watch some youtube videos on stuff I'm still confused on and just try to do as many practice problems as I can because I learn better by actually doing problems and not just watching lectures.
- Sat Nov 21, 2020 9:45 pm
- Forum: Ionic & Covalent Bonds
- Topic: Radicals
- Replies: 10
- Views: 478
Re: Radicals
Yeah you're probably right because an odd number electrons means it is a radical
- Sat Nov 21, 2020 9:41 pm
- Forum: Electron Configurations for Multi-Electron Atoms
- Topic: Ground State vs. Excited State
- Replies: 6
- Views: 2629
Re: Ground State vs. Excited State
Ground state configuration would be the configuration that you expect to see then following the rules that we learned about in class like Pauli's Exclusion Principle and Hunds rule. In an excited state, you'll see in electron in a higher energy level than expected. For example, Helium's ground state...
- Fri Nov 20, 2020 10:19 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: LDF
- Replies: 5
- Views: 423
Re: LDF
There's a bit in the textbook that talks about this regarding boiling points where it said that LDFs became more important than polarity but honestly it confused me.
- Fri Nov 20, 2020 9:44 pm
- Forum: Student Social/Study Group
- Topic: post midterm 2
- Replies: 27
- Views: 1408
Re: post midterm 2
I just had mine today and I'm just glad it's over tbh. I've been watching Netflix all day since I took it.
- Fri Nov 20, 2020 9:36 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Week 8 Chem 14A Friday Lecture
- Replies: 7
- Views: 491
Re: Week 8 Chem 14A Friday Lecture
I don't think we're supposed to have lecture because on CCLE it says that there's no school.
- Tue Nov 17, 2020 10:09 am
- Forum: Lewis Structures
- Topic: Lewis structures for Ionic bonds
- Replies: 1
- Views: 101
Lewis structures for Ionic bonds
How do we know when to do the lewis structures with the brackets and no lines vs the lewis structures with the lines (for covalent molecules). Would we have to be given the electronegativity and calculate whether the difference is larger than 2 to determine its an ionic bond? Or at least told that i...