Search found 112 matches
- Thu Mar 11, 2021 6:19 pm
- Forum: Student Social/Study Group
- Topic: How are y'all doing?
- Replies: 46
- Views: 3259
Re: How are y'all doing?
i wanna cry
- Tue Mar 09, 2021 4:34 pm
- Forum: Balancing Redox Reactions
- Topic: Textbook 6K.3!
- Replies: 6
- Views: 426
Re: Textbook 6K.3!
Thanks guys, especially RDove, it slipped my mind that you can look at the charges that way!
- Tue Mar 09, 2021 4:32 pm
- Forum: Student Social/Study Group
- Topic: What organizations are you guys in?
- Replies: 53
- Views: 4129
Re: What organizations are you guys in?
I am in EARTH, super cool environment club. I've tried a handful and never really felt the spark while I was involved, hopefully whenever we return to campus I will try some more out!
- Tue Mar 09, 2021 4:27 pm
- Forum: Student Social/Study Group
- Topic: What do you miss / What are you looking forward to?
- Replies: 92
- Views: 14718
Re: What do you miss / What are you looking forward to?
Covel fries tbh :,) and the sunset when you are the top of Janns Steps, makes you feel something (I'm a sophmore)
- Tue Mar 09, 2021 4:26 pm
- Forum: Student Social/Study Group
- Topic: It is possible to study for the final in one day?
- Replies: 44
- Views: 4888
Re: It is possible to study for the final in one day?
I know several individuals who cram within 1-2 days, it's possible but if you can sacrifice a few hours of sleep each night I think that at least you will feel better going into the exam knowing that you attempted to study a few days beforehand. I wish you good luck, I hope you pass with flying colo...
- Mon Mar 08, 2021 4:11 pm
- Forum: Balancing Redox Reactions
- Topic: Textbook 6K.3!
- Replies: 6
- Views: 426
Textbook 6K.3!
Hi, I am having a bit of trouble with a) for problem 6K.3. In the solutions manual, it shows that that the half reaction for S2O3-2 is 1[S2O3-2(aq) + 5H2O(l) --> 2SO4-2(aq) + 10H+(aq) + 8e-]. However, I tried to calculate the oxidation numbers for the compounds and don't see how 8e- is added to the ...
- Tue Mar 02, 2021 3:32 pm
- Forum: Environment, Fossil Fuels, Alternative Fuels
- Topic: Energy Density
- Replies: 10
- Views: 4905
Re: Energy Density
Because if energy density is high, there is more energy in the tightly packed, small mass (in this case, gasoline) available for the car. Although, I could have worded this wrong.
- Tue Mar 02, 2021 3:19 pm
- Forum: Work, Gibbs Free Energy, Cell (Redox) Potentials
- Topic: Max Work and Delta G
- Replies: 1
- Views: 179
Max Work and Delta G
Hello,
In last week's lecture, I am still a bit confused on how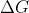 and maximum work are connected in a redox reaction (at constant temp and pressure)? I read that a decrease in Gibbs Free Energy is equal to the maximum work a system can do, but I am not sure if this is correct?
and maximum work are connected in a redox reaction (at constant temp and pressure)? I read that a decrease in Gibbs Free Energy is equal to the maximum work a system can do, but I am not sure if this is correct?
In last week's lecture, I am still a bit confused on how
- Tue Mar 02, 2021 3:01 pm
- Forum: General Science Questions
- Topic: Midterm 2 Reactions
- Replies: 79
- Views: 8172
Re: Midterm 2 Reactions
I was upset by the score on my second midterm. It just reminded me that I should not walk out even a little confident on exams lol, or it'll just hurt more. I don't have a lot of time to study for the final, but I will try to fit as much as possible.
- Tue Mar 02, 2021 2:57 pm
- Forum: Student Social/Study Group
- Topic: Moving to Westwood soon. Which restaurants should I visit?
- Replies: 45
- Views: 3612
Re: Moving to Westwood soon. Which restaurants should I visit?
Poquito Mas is a little farther away from campus, but they have good Mexican food there (especially because the tortillas are really delicious and made there in the place).
- Tue Mar 02, 2021 2:53 pm
- Forum: Student Social/Study Group
- Topic: Textbook Problem Strategies
- Replies: 17
- Views: 882
Re: Textbook Problem Strategies
When I am solving the textbook problems, I urge myself to get through every single one, and check the solution immediately after each solution. There are some problems where I have no clue how to start solving and look at the solution manual, but I highlight formulas and take small notes on what is ...
- Mon Feb 22, 2021 1:20 pm
- Forum: Student Social/Study Group
- Topic: How to Reduce Nervousness before getting results
- Replies: 63
- Views: 6664
Re: How to Reduce Nervousness before getting results
Thankfully, my parents don't tend to ask and wish me good luck instead haha! Anyhow, try to not make it a habit of checking myucla, as it is bad for your mental health. Make a plan for the week and concentrate on your work, which is what I usually do :)
- Mon Feb 22, 2021 1:16 pm
- Forum: Student Social/Study Group
- Topic: Chem 14B Final
- Replies: 86
- Views: 7428
Re: Chem 14B Final
Besides doing textbook problems and getting a feel for the equations, definitely use the outlines for each unit that are on Prof Lavelle's website! Each bullet point helps understand the equations and the material more conceptually.
- Mon Feb 22, 2021 1:14 pm
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: Thermodynamically Stable or Unstable
- Replies: 8
- Views: 870
Re: Thermodynamically Stable or Unstable
The system is thermodynamically unstable when the reaction is endothermic, meaning that Gibbs Free Energy is positive. This is because Gibbs Free Energy is basically the max amount of energy available for work in the reverse direction, thus it is not spontaneous in the forward direction.
- Mon Feb 22, 2021 12:53 pm
- Forum: Thermodynamic Systems (Open, Closed, Isolated)
- Topic: Degeneracy
- Replies: 11
- Views: 961
Re: Degeneracy
We can think of it in terms of a molecule, where its bonds are flexible and can be positioned a number of ways, increasing degeneracy; this is because it is the number of microstates, or positions, which in turn increases entropy.
- Mon Feb 22, 2021 12:46 pm
- Forum: Student Social/Study Group
- Topic: We made it through Midterm 2!
- Replies: 71
- Views: 5441
Re: We made it through Midterm 2!
AlyssaMaynard1C wrote:Since both midterms were around 15 questions, do we think the final will be 30? Or less
Last quarter I believe it was 30 questions, so it's the # of midterm questions x2 basically :)
- Tue Feb 16, 2021 10:05 am
- Forum: Concepts & Calculations Using First Law of Thermodynamics
- Topic: Values for Cv and Cp
- Replies: 3
- Views: 238
Re: Values for Cv and Cp
These values are usually given. It would explicitly ask for the heat capacity (Cv/Cp), which you can find using the equation q=m*C*DeltaT. I believe it appeared as a question, but can't remember it. There is a question on sapling where you had to modify Cv,m, so just be wary of those.
- Tue Feb 16, 2021 9:56 am
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: Spontaneity of a System
- Replies: 38
- Views: 3057
Re: Spontaneity of a System
A spontaneous reaction is a favorable reaction that requires no energy input to create products; the reaction releases more energy as heat as a result. We can usually determine a reaction is spontaneous, for example, if it is exothermic or if it has an increase in DeltaS.
- Tue Feb 16, 2021 9:45 am
- Forum: General Science Questions
- Topic: conversions [ENDORSED]
- Replies: 6
- Views: 893
Re: conversions [ENDORSED]
I know this sounds redundant haha, but the textbook questions help get used to the unit conversions. Heat (q), for example, is usually seen as kj, so it would be best to convert an answer as J to kj.
- Tue Feb 16, 2021 9:40 am
- Forum: General Science Questions
- Topic: No Lavelle Chem 14C?
- Replies: 68
- Views: 6762
Re: No Lavelle Chem 14C?
Constance Newell wrote:Does anyone know if the lectures are recorded/asynchronous or do we have to attend live?
I believe the lectures are live, otherwise the class planner would list Chem 14C as "online-recorded" :)
- Tue Feb 16, 2021 9:37 am
- Forum: Student Social/Study Group
- Topic: Midterm 2 Nerves
- Replies: 40
- Views: 2396
Re: Midterm 2 Nerves
Yes, the sapling questions really worried me, it felt as if I did not get the material at all. The textbook questions have not been too frustrating yet. Since there is so many details within thermochemistry, I feel as I will forget everything once the exam starts though. Definitely nervous.
- Sat Feb 13, 2021 7:07 pm
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: Sapling Week 5/6 #18
- Replies: 5
- Views: 325
Re: Sapling Week 5/6 #18
Abby Lam 3F wrote:nvm someone helped me figure it out! If anyone has the same issue it's because Sapling does not take 2.652x10^42 as a correct answer. You should just put 2.65x10^42
Thank you! Literally struggling on this for 20 minutes because I thought I converted the units wrong lol
- Sat Feb 13, 2021 1:31 am
- Forum: Student Social/Study Group
- Topic: Thoughts on Upcoming Midterm 2
- Replies: 41
- Views: 2120
Re: Thoughts on Upcoming Midterm 2
I'm incredibly nervous too. I have been doing the sapling homework questions, and I became so frustrated because I did not understand why particular equations were used in some questions and the terminology was a bit overwhelming. I'm going to study for the entire next week, but I don't think I'll f...
- Tue Feb 09, 2021 10:27 pm
- Forum: General Science Questions
- Topic: Recognizing Acids&Bases
- Replies: 2
- Views: 204
Re: Recognizing Acids&Bases
Try breaking the salt into the cation and the anion, and determining what effect the cation and anion have on pH. For example, for NaF, it is Na+ and F-. Na+ is from NaOH, which is a very strong base. Based on the conjugate seesaw, this means that Na+ will be a very weak acid, so that it basically ...
- Mon Feb 08, 2021 12:51 pm
- Forum: Thermodynamic Systems (Open, Closed, Isolated)
- Topic: Closed vs Isolated System
- Replies: 30
- Views: 1571
Re: Closed vs Isolated System
Closed system: Not insulated (meaning it can pass down heat to the table it is on, for example), but is completely sealed. So energy can be exchanged/transferred, but matter cannot. Isolated system: Completely sealed and insulated. Therefore, nothing is exchanged (think of a calorimeter). We usually...
- Mon Feb 08, 2021 12:43 pm
- Forum: General Science Questions
- Topic: Recognizing Acids&Bases
- Replies: 2
- Views: 204
Recognizing Acids&Bases
Hi,
So I have majority of the strong acids and bases memorized from the chempendix, but have trouble recognizing acids and bases like NH4Br or NaF(?), does anyone have any tips?
So I have majority of the strong acids and bases memorized from the chempendix, but have trouble recognizing acids and bases like NH4Br or NaF(?), does anyone have any tips?
- Mon Feb 08, 2021 12:33 pm
- Forum: Student Social/Study Group
- Topic: Best kdrama?
- Replies: 30
- Views: 2325
Re: Best kdrama?
Not necessarily a kdrama but a chinese drama, "Meteor Garden" (2018) is one of my favorite shows. The protagonist is a very tough gal who really helps the love interest change for better, I totally recommend it!
- Mon Feb 08, 2021 12:29 pm
- Forum: Student Social/Study Group
- Topic: Book Recommendations
- Replies: 135
- Views: 22342
Re: Book Recommendations
A friend of mine recommended me one called "The Knife of Never Letting Go" by Patrick Ness. In my Youtube phase I watched someone called Joey Graceffa and bought his book called "Eden" lolololol, it's not half bad tbh!
- Mon Feb 08, 2021 12:23 pm
- Forum: Student Social/Study Group
- Topic: Post Midterm 1...
- Replies: 39
- Views: 2205
Re: Post Midterm 1...
I know exactly how you are feeling! I have also done textbook problems and rewatched lectures to grasp the topics, but keep scoring below average. Everyone is saying UA workshops, but honestly they are just extra practice and personally do not work out for me. Definitely skim through the textbook to...
- Wed Feb 03, 2021 7:56 pm
- Forum: General Science Questions
- Topic: Midterm 1
- Replies: 4
- Views: 355
Re: Midterm 1
Hi guys im panicking to the point of depression. I did so so bad on the exam. When I saw that the class average was so high, I almost died because I did so bad. For those who are successful this quarter, how are you guys staying successful? I was successful in 14A by doing the textbook practice pro...
- Wed Feb 03, 2021 7:37 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Midterm
- Replies: 9
- Views: 583
Re: Midterm
Nadya Higgins 3F wrote:They're being posted today..! Pretty nervous..
Feeling the exact same way, I'm not the best at timed exams..let's hope for some good results!
- Wed Feb 03, 2021 3:23 am
- Forum: Student Social/Study Group
- Topic: Midterm Scores
- Replies: 25
- Views: 1126
Re: Midterm Scores
It appears that scores will be released tomorrow, possibly during the evening, along with our chemistry community points! :)
- Tue Feb 02, 2021 1:02 am
- Forum: Applying Le Chatelier's Principle to Changes in Chemical & Physical Conditions
- Topic: 6E.1 Textbook Question
- Replies: 3
- Views: 327
Re: 6E.1 Textbook Question
Because it is asking you for the pH rather than the pOH of the strong sulfuric acid, there are not many steps.
[H3O+] = 0.15 M
pH = -log(0.15)
= 0.8
[H3O+] = 0.15 M
pH = -log(0.15)
= 0.8
- Tue Feb 02, 2021 12:54 am
- Forum: Administrative Questions and Class Announcements
- Topic: Reflecting on the midterm
- Replies: 17
- Views: 916
Re: Reflecting on the midterm
yes! I had to guess on a couple of problems too. I tend to not think through questions because of the pressure of finishing on time, as well. I used the "skip problems you don't know the answer to" trick, and it helped me get to problems I could solve. However, it could only help so much.
- Mon Feb 01, 2021 5:51 pm
- Forum: Student Social/Study Group
- Topic: Congrats!!
- Replies: 21
- Views: 1344
Re: Congrats!!
I just realized we are in week 5, good job to everyone for finishing the midterm :). For those who had a hard time and are finding it bittersweet, you are not alone in feeling that way! But pat yourself on the back, you finished the first round!
- Mon Jan 25, 2021 6:48 pm
- Forum: Student Social/Study Group
- Topic: CHEM 14B Midterm 1 Notes
- Replies: 23
- Views: 1344
Re: CHEM 14B Midterm 1 Notes
Your notes are so neat and organized, thank you! Good luck with the midterm!
- Mon Jan 25, 2021 6:43 pm
- Forum: Student Social/Study Group
- Topic: Questions about Friday/Midterm
- Replies: 7
- Views: 320
Re: Questions about Friday/Midterm
Hi! So, this is my first time taking Lavelle, and I wanted to ask how people think his exams are? Would you say they're typically easier or harder compared to the questions assigned in homework and in the textbook? Also, are the exams on a time crunch, meaning you're rushing to answer all the quest...
- Mon Jan 25, 2021 5:58 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: Difference between standard reaction enthalpy, enthalpy of formation, and enthalpy of combustion
- Replies: 2
- Views: 250
Re: Difference between standard reaction enthalpy, enthalpy of formation, and enthalpy of combustion
Standard reaction enthalpy: All reactants and products are in their standard states (their stable forms). Enthalpy of formation: Reactants must be in their standard state (their stable forms) and there must be only one product present with a coefficient of 1. Enthalpy of combustion: Usually, there m...
- Mon Jan 25, 2021 5:44 pm
- Forum: Student Social/Study Group
- Topic: Favorite Music
- Replies: 113
- Views: 17104
Re: Favorite Music
Jazz is really nice for studying, especially the Bossa Nova songs by Joao Gilberto because they have a nice beat and are "not boring". Still Woozy, Sissyfuss, and Stella Brown by Jelani Aryeh are really mellow and non-distracting indie music, totally recommend.
- Mon Jan 25, 2021 5:11 pm
- Forum: Student Social/Study Group
- Topic: Questions about Friday/Midterm
- Replies: 7
- Views: 320
Questions about Friday/Midterm
Hii, When it was mentioned that our midterm would be during our lecture time, does it mean those who have a 10am lecture will take it at 10am, those who have a 1pm lecture will take it at 1pm, etc.? And does anyone know if there will be a lecture recording on Friday? Just want to plan ahead for the ...
- Thu Jan 21, 2021 1:47 am
- Forum: Applying Le Chatelier's Principle to Changes in Chemical & Physical Conditions
- Topic: Sapling Week 2, Question 3
- Replies: 4
- Views: 228
Re: Sapling Week 2, Question 3
I got the same message and when it tells us to "use the quadratic formula", it means that we cannot assume x is an extremely small number and omit it out of our calculations. We will need to use the quadratic formula to solve for x since we now have to do a more complex calculation, with ...
- Thu Jan 21, 2021 1:12 am
- Forum: Applying Le Chatelier's Principle to Changes in Chemical & Physical Conditions
- Topic: Sapling Week 2, Question 3
- Replies: 4
- Views: 228
Sapling Week 2, Question 3
"Muscles produce lactic acid, CH3CH(OH)COOH(aq), during exercise. Calculate the percent ionization (deprotonation), pH, and pOH of a 0.1013 M solution of lactic acid. The acid‑dissociation (or ionization) constant, Ka, of this acid is 8.40×10^−4." I was able to solve the pH and pOH of this...
- Tue Jan 19, 2021 11:41 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: help with textbook 5I.15
- Replies: 4
- Views: 181
Re: help with textbook 5I.15
Is that really the reason? I've seen problems where the products have -x change and the reactants have +x change, so I'm a bit confused.
- Tue Jan 19, 2021 9:36 pm
- Forum: General Science Questions
- Topic: Textbook Problem: A little off with algebra
- Replies: 2
- Views: 196
Textbook Problem: A little off with algebra
I was looking over my answer for 5I.5 and realized that the ratio for the reactants and products Kc=\frac{[PCl_{3}][Cl_{2}]}{[PCl_{5}]} went from 25=\frac{[PCl_{3}][5.43]}{[1.18]} to PCl_{3}=\frac{[25][1.18]}{[5.43]} . I know it's a little silly, but could someone explain the algebra behind this? I ...
- Tue Jan 19, 2021 9:25 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: help with textbook 5I.15
- Replies: 4
- Views: 181
help with textbook 5I.15
"When solid NH4HS and 0.400 mol NH3(g) were placed in a vessel of volume 2.0 L at 24°C, the equilibrium NH4HS(s)⇌NH3(g)+H2S(g), for which Kc=1.6×10−4, was reached. What are the equilibrium concentrations of NH3 and H2S?" Hii, so as I was completing the calculations for this problem, I chec...
- Tue Jan 19, 2021 12:13 am
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Moles vs Concentrations in ICE Box
- Replies: 21
- Views: 1059
Re: Moles vs Concentrations in ICE Box
It really depends on the type of problem. If you are given the moles and volume of an reactant or products, I use the concentration since its usually the initial concentration needed for ICE Box calculations. However, if I am already given at least one concentration at equilibrium for a reactant or ...
- Tue Jan 19, 2021 12:01 am
- Forum: Student Social/Study Group
- Topic: New Joke Chain
- Replies: 46
- Views: 3633
Re: New Joke Chain
Saw this one pretty recently:
What should you do if no one laughs at your chemistry jokes?
Keep telling them until you get a reaction.
What should you do if no one laughs at your chemistry jokes?
Keep telling them until you get a reaction.
- Thu Jan 14, 2021 7:14 pm
- Forum: General Science Questions
- Topic: Charge sign in front of molecular formula meaning?
- Replies: 4
- Views: 328
Re: Charge sign in front of molecular formula meaning?
I believe the positive charge is in the front because it is the charge of N, as it is the central atom all 3 H and 1 O seem to bond with.
- Thu Jan 14, 2021 2:35 am
- Forum: Student Social/Study Group
- Topic: Topics we need to know from chem 14a for chem 14b
- Replies: 6
- Views: 393
Re: Topics we need to know from chem 14a for chem 14b
Two simpler things you should know from chem14a is definitely unit conversions and balancing chemical equations! I think that applies to nearly every chemistry course you'll take.
- Wed Jan 13, 2021 8:30 pm
- Forum: Student Social/Study Group
- Topic: Comfort Movies
- Replies: 168
- Views: 44714
Re: Comfort Movies
I have more comfort shows than I do movies, but my comfort movies are usually by DreamWorks, like How to Train Your Dragon and Puss in Boots hahahaha
- Wed Jan 13, 2021 2:10 am
- Forum: Student Social/Study Group
- Topic: Advice for study
- Replies: 31
- Views: 1732
Re: Advice for study
Definitely do the practice problems from the textbook, especially on your own completely as a trial run, and then look through the answer key provided to see what are your common mistakes. Skim through the textbook too, as there are a few key concepts and vocab not always mentioned in lecture. Revie...
- Mon Jan 11, 2021 10:27 pm
- Forum: General Science Questions
- Topic: Acid & Base Equilibria Lecture Question
- Replies: 7
- Views: 387
Acid & Base Equilibria Lecture Question
Hiya, I am slightly confused on the autoprotolysis reaction's equilibrium constant from lecture. Why is [H20] removed from Kc = \frac{[H_{3}O^{+}][OH^{+}]}{[H_{2}O]^{2}} ? Professor Lavelle mentioned that the concentration of [H2O] is the same on the left and the right of the reaction, which is why ...
- Sat Jan 09, 2021 7:41 pm
- Forum: Non-Equilibrium Conditions & The Reaction Quotient
- Topic: Confusion with terms
- Replies: 9
- Views: 421
Re: Confusion with terms
Yes, when the reaction favors the products it means that the forward reaction is favored; it's noticeable when the equilibrium constant K is large, due to the large amount of concentration from the products.
- Sat Jan 09, 2021 7:26 pm
- Forum: Non-Equilibrium Conditions & The Reaction Quotient
- Topic: Changes in Q vs K
- Replies: 6
- Views: 590
Changes in Q vs K
Hi all, I just had a question after going to a step up session and seeing all the Q&K questions on here. Why does a change in pressure or volume not change the equilibrium constant K, but changes Q? Why does a change in temperature affect K?
- Fri Jan 08, 2021 3:12 am
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Sapling HW Week 1 #5
- Replies: 2
- Views: 170
Re: Sapling HW Week 1 #5
Basically, you would have to see which of the following equations would be able to match the first one when added together. This can be done by either multiplying the equations by a constant or reversing their reaction. For example, one of the equations that would be used in the combination is the f...
- Wed Jan 06, 2021 9:24 pm
- Forum: Student Social/Study Group
- Topic: Groupme for 14B
- Replies: 5
- Views: 299
Re: Groupme for 14B
Hi there, Sorry if this question has already been asked, but is there a Groupme for Lecture 2 of 14B with Lavelle? If so, I'd be really grateful if you could send the link. Thanks! There was a doc linked on here with all course groupme chats, but there is no groupme made specifically for Lecture 2 ...
- Wed Jan 06, 2021 9:20 pm
- Forum: Student Social/Study Group
- Topic: Learning Sessions
- Replies: 24
- Views: 1424
Re: Learning Sessions
I think this varies for most students! I personally like to go to at least 2 UA sessions/week (I usually go to 1 step-up and 1-2 workshops). Sometimes I would go to more before exams because the UA's oftentimes provide review questions. During the week before finals, I went to about one per day bec...
- Fri Dec 11, 2020 10:05 pm
- Forum: Lewis Acids & Bases
- Topic: Textbook Problem J.7
- Replies: 2
- Views: 203
Textbook Problem J.7
In this question, it was asking to select an acid or base for a neutralization reaction that results in a) potassium bromide b) zinc nitrate etc. and to write its chemical formula. However, I am not sure how to go about this question, does anyone have any pointers?
- Fri Dec 11, 2020 3:17 pm
- Forum: Hybridization
- Topic: Textbook 2.45!
- Replies: 2
- Views: 170
Re: Textbook 2.45!
Hybridization isn't determined by the number of valence electrons that each atom has but by the number of regions of electron density. So for example, a double bond, like the one in C=C, would be considered one region of electron density. If the atom has two single bonds and one double bond, then i...
- Fri Dec 11, 2020 1:36 am
- Forum: Hybridization
- Topic: Textbook 2.45!
- Replies: 2
- Views: 170
Textbook 2.45!
So I was looking at the answer key for part b) of this question, and was wondering why the hybridization of the orbitals for the C=C bond was sigma(C 2sp^2, C 2sp^2) instead of sigma(C sp^3, C sp^3)? In a previous problem, the hybridization of Si (same period as C) was sp^3. Why are they different i...
- Tue Dec 08, 2020 5:12 pm
- Forum: Student Social/Study Group
- Topic: lavelle study review time??
- Replies: 3
- Views: 271
Re: lavelle study review time??
Hi! I hope i don't seem dumb but what time is prof. lavelle's review session tomorrow (Wednesday)? I swear I can't find it on any of the announcements or emails. I might just be overwhelmed with studying lol He mentioned in his email that it is his regular Wednesday discussion time, which is from 5...
- Tue Dec 08, 2020 1:41 am
- Forum: Student Social/Study Group
- Topic: Week 10 Review Sessions
- Replies: 7
- Views: 439
Re: Week 10 Review Sessions
Definitely going to the Lewis Structure review sessions, they seem to throw me off the most. Funny enough I find some of the fundamental topics a bit more difficult than quantum, so hopefully I can attend that one too!
Re: en
I don't believe there are other bidentate ligands we have been too familiarized with like 'en', but it might be useful to review other bidentate ligands and their structure :)
- Sun Dec 06, 2020 12:59 pm
- Forum: Naming
- Topic: Helpful Videos for Naming
- Replies: 22
- Views: 1358
Re: Helpful Videos for Naming
I love organic chemistry tutor as much as the next chem student, thanks for sharing these videos!
- Sun Dec 06, 2020 12:54 pm
- Forum: Student Social/Study Group
- Topic: Point distribution
- Replies: 11
- Views: 779
Re: Point distribution
I totally understand your point of view, but he is allowing many people to pass with a C- by just earning half the points in the course. I know several of us really want to pass with an A, including myself, but the pandemic sadly puts everyone in a difficult position. Do not let earning a grade lowe...
- Mon Nov 30, 2020 11:26 pm
- Forum: General Science Questions
- Topic: study methods/recs
- Replies: 37
- Views: 2669
Re: study methods/recs
The textbook! Could not emphasize this enough. Read through the topics that we have covered in lectures, taking extra notes that seem important, and solve the problems assigned from the outline in the syllabus! I found that focusing on these is helpful.
- Mon Nov 30, 2020 11:21 pm
- Forum: Student Social/Study Group
- Topic: Week 8/9 Thoughts/Worries
- Replies: 66
- Views: 5088
Re: Week 8/9 Thoughts/Worries
This quarter has been a rollercoaster, especially this class, but I still enjoy the material quite a lot. I definitely felt like I could have done better on the midterms, all the little mistakes I made on them cost me a higher grade in the class and it's an icky feeling. Mentally preparing myself fo...
- Thu Nov 26, 2020 10:22 pm
- Forum: Student Social/Study Group
- Topic: How have your study habits changed?
- Replies: 45
- Views: 2403
Re: How have your study habits changed?
Yes they have changed, I start studying for an exam about 2 weeks before to avoid cramming. I am still not getting the grades I want though. I relate to this a lot actually, I developed better study habits but never seemed to get the grades I want on the exams. I guess it's a matter of constantly c...
- Thu Nov 26, 2020 1:48 am
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: Lecture Example Chelating Ligand
- Replies: 2
- Views: 175
Lecture Example Chelating Ligand
For the chelating ligand example in lecture #24 with the cation Co2+, why is it considered a bidentate (unless I misheard Prof Lavelle)? And what is meant by rotation? Does it mean that NH2 can become H2N to bond with Co2+? I cannot seem to grasp the chelating concept well, sorry if this question se...
- Wed Nov 25, 2020 12:49 am
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Sapling wk 8 #17
- Replies: 2
- Views: 142
Re: Sapling wk 8 #17
Your last structure should look like something like this: H H \ / C / \ C = C / \ H H Sorry if it seems out of proportion, but I am assuming this is what they mean by cyclic because of the ring of Carbons.
- Tue Nov 24, 2020 10:19 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: sapling HW question three
- Replies: 5
- Views: 379
Re: sapling HW question three
I think what it's really asking is the smallest bond angle possible, and because this is a linear shape, it would be 180 degrees. These measurements were talked about in the lectures, so you don't have to necessarily do any calculations :)
- Mon Nov 23, 2020 6:58 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: VSEPR T-Shaped Molecular Geometry
- Replies: 7
- Views: 443
Re: VSEPR T-Shaped Molecular Geometry
Samantha Pedersen 2D wrote:Hello, will we be expected to recognize T-shaped molecular geometry as one of the VSEPR shapes? Thank you!
If it helps, you can recognize whether a VSEPR shape is T-shaped by seeing if it has 3 outer atoms connected to the central atom and 2 lone pairs on the central atom!
- Wed Nov 18, 2020 7:10 pm
- Forum: Student Social/Study Group
- Topic: Big Sad: Midterm 2
- Replies: 86
- Views: 9450
Big Sad: Midterm 2
After taking the second midterm, I just realized how many mistakes I made and how probable it is that I am not going to pass it haha. While there are students worried about getting an A-, I am worried about getting a passing grade haha. For those of us who have taken it, how are you feeling?
- Wed Nov 18, 2020 12:01 am
- Forum: Dipole Moments
- Topic: Dipole representation on midterm 2
- Replies: 9
- Views: 495
Re: Dipole representation on midterm 2
Oh, correct me if I'm wrong, but I always thought the direction of the arrow would be in the positive direction because the partial positive is attracting the negatively charged electrons. In other words, the electrons would be the ones to move closer to the positive source I think this is true for...
- Tue Nov 17, 2020 11:29 pm
- Forum: Resonance Structures
- Topic: Resonance
- Replies: 4
- Views: 184
Re: Resonance
Resonance does not necessarily mean the lowest energy structure. Resonance is all possible Lewis structures of a molecule, in which you can find the most stable structure (because it will have the lowest energy).
- Mon Nov 16, 2020 3:26 pm
- Forum: Ionic & Covalent Bonds
- Topic: Textbook 2A.5
- Replies: 4
- Views: 217
Re: Textbook 2A.5
Usually for cations and anions, I like to find the configuration of the atom before it loses or gains electrons as a basis. For Bi, the ground state configuration would be [Xe]4f^14 5d^10 6s^2 6p^3 (we did not learn about how to write electron configurations with f orbitals, but the f orbitals shou...
- Mon Nov 16, 2020 3:15 pm
- Forum: Ionic & Covalent Bonds
- Topic: Textbook Problem 2A.5
- Replies: 3
- Views: 253
Re: Textbook Problem 2A.5
Faith Veenker 3H wrote:I think you are looking at 2A.6, which is not an assigned problem. For 2A.5, [Ar]3d^10 is the configuration for Cu+.
I just noticed that haha, my mistake! Thank for answering though!
- Mon Nov 16, 2020 3:09 pm
- Forum: Ionic & Covalent Bonds
- Topic: Textbook Problem 2A.5
- Replies: 3
- Views: 253
Textbook Problem 2A.5
For (a), the electron configuration for Zr^4+ is [Ar]3d^10. Why can it not be [Kr], just as how we used [Ar] for S^2-? Is there a clear reason for this?
- Wed Nov 11, 2020 2:49 am
- Forum: General Science Questions
- Topic: Which would have lower ionization energy?
- Replies: 13
- Views: 839
Which would have lower ionization energy?
Out of curiosity, which would have lower ionization energy, Cl or O? If it were to ever be needed in making a Lewis structure.
- Wed Nov 11, 2020 1:36 am
- Forum: Ionic & Covalent Bonds
- Topic: Increasing Covalent Character
- Replies: 3
- Views: 243
Re: Increasing Covalent Character
Much of it has to do with electron density. As the anion in an ionic silver halide becomes bigger, meaning it has more electrons that are very loosely held, the cation Ag will distort the electrons even more than if it were a smaller anion. This causes the electrons to be shared more covalently betw...
- Wed Nov 11, 2020 12:10 am
- Forum: Student Social/Study Group
- Topic: Lecture on Veteran's Day
- Replies: 10
- Views: 440
Re: Lecture on Veteran's Day
Will there be a lecture posted this Wednesday? Just checking to see as it's Veteran's Day. In addition to that, what happens if we have a lab on that day? I assume that we might have to join another lab session instead but I am not sure. You do not show up to lab if it it's on that day, and you don...
- Tue Nov 10, 2020 1:26 am
- Forum: Student Social/Study Group
- Topic: grade worries
- Replies: 119
- Views: 26689
Re: grade worries
After deciding to apply to grad school in the near future, yes, grades are a huge worry for me. I think for the next two exams, aim for no more than 30 points incorrect each. It really depends on what you got on your first midterm and what you want as a final grade, but any more than 30 points incor...
- Tue Nov 10, 2020 1:22 am
- Forum: Student Social/Study Group
- Topic: Mid-quarter Check in
- Replies: 67
- Views: 4999
Re: Mid-quarter Check in
Thanks for asking this question, I am honestly bummed out because there is so many assignments that I can't even leave my room, and it's become so repetitive. College is supposed to be a balance of social and academic, but it's purely academic for me. Last year this time around was not super great f...
- Wed Nov 04, 2020 5:15 pm
- Forum: Student Social/Study Group
- Topic: Post Midterm Blues
- Replies: 71
- Views: 4844
Re: Post Midterm Blues
Same here, I was just in my discussion and my TA mentioned that our average was high compared to other sections, and all the other students were happy about it. But I know I did not really contribute to that average, and it upsets me. Let's hope the next midterm is better! :')
- Wed Nov 04, 2020 5:11 pm
- Forum: Student Social/Study Group
- Topic: Favorite TV shows
- Replies: 277
- Views: 57322
Re: Favorite TV shows
S*x Education is so so so good! If you don't mind something a little explicit, hence the name, it's hilarious and interesting! The Good Place never gets old either, they just added a new season on Netflix!
- Wed Nov 04, 2020 4:50 pm
- Forum: Lewis Structures
- Topic: Valence Electrons
- Replies: 10
- Views: 474
Re: Valence Electrons
Here's an extra tip! For Lewis structures, remember that the first shell can hold 2 electrons, the second and third shell can hold 8, and the fourth and fifth shell can hold 18. Once you count the electrons (atomic number), you can draw it out and the amount left over on the last shell are the valen...
- Wed Nov 04, 2020 4:43 pm
- Forum: Ionic & Covalent Bonds
- Topic: 2 paired electrons
- Replies: 3
- Views: 142
Re: 2 paired electrons
I agree with what Liam said, and when you see a singular electron ( . ) forming a bond with another singular electron from another atom, it preferably becomes a dash rather then the regular electron pair ( : )
- Mon Nov 02, 2020 8:51 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Midterm Scores
- Replies: 28
- Views: 1443
Re: Midterm Scores
Update: someone went to Prof Lavelle's OH and he mentioned that the scores would be up Thursday!
- Thu Oct 29, 2020 12:43 am
- Forum: Einstein Equation
- Topic: E=pc vs E=hv
- Replies: 15
- Views: 940
Re: E=pc vs E=hv
I believe E=pc is usually used in cases like DeBroglie problems, where p=mass x velocity. E=hv is a more common formula we have been using because we are often given problems involving frequency.
- Thu Oct 29, 2020 12:39 am
- Forum: Empirical & Molecular Formulas
- Topic: Review for Midterm
- Replies: 11
- Views: 822
Re: Review for Midterm
I have heard that the formulas will be provided so I do not think you need to know them based on names. However, it is good practice to know because many of the practice problems in the outlines tend to use names a lot.
- Thu Oct 29, 2020 12:34 am
- Forum: General Science Questions
- Topic: Understanding Velocity
- Replies: 3
- Views: 217
Understanding Velocity
Sorry for the silly question! I have seen some practice problems regarding finding the velocity of an electron when given wavelength, work function etc. Is there particular formulas we can use for velocity of an electron?
- Thu Oct 29, 2020 12:20 am
- Forum: Student Social/Study Group
- Topic: Lavelle's Jokes
- Replies: 26
- Views: 2497
Re: Lavelle's Jokes
An electron walks into a bar. He asks the bartender, "How much for a beer?" The bartender frowns and tells him, "No can do, you got a negative on your tab."
I really liked the neutron bartender joke and modified it to be an electron joke :)
I really liked the neutron bartender joke and modified it to be an electron joke :)
- Mon Oct 26, 2020 5:32 pm
- Forum: Einstein Equation
- Topic: Sapling Homework - electron affinity
- Replies: 5
- Views: 313
Re: Sapling Homework - electron affinity
Yeah I couldn't get the right answer for the second part either. I wonder if there is anything wrong with my conversions. @Samantha, could you explain your steps for the first part and how you got 1.003? I used E=hc/lambda and converted nm to m as well as J to eV, but I still didn't get to 1.003. T...
- Sun Oct 25, 2020 5:24 pm
- Forum: DeBroglie Equation
- Topic: Help on DeBroglie Post-Module #34?
- Replies: 2
- Views: 97
Re: Help on DeBroglie Post-Module #34?
Hi! I solved the problem the exact same way and got the same answer. But when you choose an answer, you have to remember to use the correct number of sig figs. So while 99908.32439 is the exact answer, 1.00 x 10^5 is the correct answer when you use the right number of sig figs. Another way to look ...
- Sun Oct 25, 2020 2:57 pm
- Forum: DeBroglie Equation
- Topic: Help on DeBroglie Post-Module #34?
- Replies: 2
- Views: 97
Help on DeBroglie Post-Module #34?
"If an electron (mass 9.11 x 10^-31 kg) has an associated wavelength of 7.28 x 10^-9 m, what is its speed? Is your answer reasonable, why?" I keep using the formula v=h/(wavelength)(mass), but I get a long decimal number (99908.32439) and not the one stated in the multiple choice options (...
- Tue Oct 20, 2020 9:24 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Video Learning Modules?
- Replies: 5
- Views: 178
Re: Video Learning Modules?
I think the post-module assessment in the modules are especially useful, it lets us practice a lot of what is taught in the videos. I hope we get to see more soon, especially if future topics are more difficult to understand.
- Tue Oct 20, 2020 9:11 pm
- Forum: Student Social/Study Group
- Topic: How to memorize formulas?
- Replies: 7
- Views: 434
How to memorize formulas?
Hello! I was wondering if anyone had any tips for knowing which formulas to use for certain problems to prep for the midterm? For example, if it asks for maximum kinetic energy, or the frequency of an incident light? Or would just memorizing example problems and the formulas used to solve them be en...
- Tue Oct 20, 2020 9:01 pm
- Forum: Student Social/Study Group
- Topic: Midterm Fundamentals Review Sheet
- Replies: 7
- Views: 455
Re: Midterm Fundamentals Review Sheet
You are amazing!! Thanks so much! This is actually so good for memorizing :)
- Tue Oct 20, 2020 8:56 pm
- Forum: Photoelectric Effect
- Topic: Photoelectric Effect Module Question
- Replies: 3
- Views: 80
Re: Photoelectric Effect Module Question
For this problem, you need to use the formula E(kinetic energy)=1/2(MV^2). Thus, when you plug in the values the problem provides you, it is 1/2(9.10938 x 10^-31)(6.61 x 10^5)^2. All you have to do is solve :)
- Mon Oct 19, 2020 5:21 pm
- Forum: Student Social/Study Group
- Topic: How are you studying?
- Replies: 204
- Views: 28429
Re: How are you studying?
Brian_Wu_3G wrote:I see everyone mentioning step up sessions. What are those again? I might want to attend them if they help.
Step up sessions basically go through prepared worksheets and practice problems based on the topic the week focuses on. They do help if you do not understand a certain topic.

