Search found 107 matches
- Sun Mar 07, 2021 8:38 pm
- Forum: Student Social/Study Group
- Topic: Almost through!
- Replies: 8
- Views: 893
Almost through!
Hey everyone, we're heading into our last week of the quarter and I want to remind you that you are amazing and should be proud of yourself. Hopefully this quarter went well for yall. What is the one thing you are most proud of that you did/persevered through/completed this past quarter?
- Sun Mar 07, 2021 8:34 pm
- Forum: Student Social/Study Group
- Topic: Note Taking
- Replies: 145
- Views: 16703
Re: Note Taking
I have always done my notes handwritten because I can format them the way I want and I feel like I remember things better that way.
- Sun Mar 07, 2021 8:32 pm
- Forum: General Science Questions
- Topic: Final Exam
- Replies: 39
- Views: 2135
Re: Final Exam
The final review sessions are really helpful; I definitely recommend going!
- Sun Mar 07, 2021 5:25 pm
- Forum: Student Social/Study Group
- Topic: How do you deal with burnout?
- Replies: 144
- Views: 15596
Re: How do you deal with burnout?
Wow, I love everyone's responses. This is something I struggle with a lot, especially this quarter for some reason. I find myself procrastinating on simple tasks, so I try to get rid of the pressure to do those tasks by giving me a time limit that is easily attainable. For example, if I have two ass...
- Fri Feb 26, 2021 8:41 pm
- Forum: Balancing Redox Reactions
- Topic: Sapling week 7/8 #5
- Replies: 2
- Views: 240
Sapling week 7/8 #5
Identify the oxidizing and reducing agents in the skeletal (unbalanced) reaction. Then, balance the reaction, including the phase (solid, liquid, etc.) of each species. The reaction takes place in basic aqueous solution. Cl2O7(g)+H2O2(aq)⟶ClO2^-(aq)+O2(g) Can someone explain how to come up with the ...
- Fri Feb 26, 2021 3:09 pm
- Forum: Student Social/Study Group
- Topic: Book Recommendations
- Replies: 135
- Views: 14333
Re: Book Recommendations
Love seeing all of your recommendations; I will be sure to look into them. My favorite book is Jurassic Park by Michael Crichton, but more recently I have read An Absolutely Remarkable Thing by Hank Green (and am currently reading the sequel), and The Kite Runner by Khaled Hosseini.
- Thu Feb 25, 2021 8:17 am
- Forum: Student Social/Study Group
- Topic: Chem 14B Final
- Replies: 86
- Views: 5685
Re: Chem 14B Final
I would say the final is similar to the midterms. My biggest issue was not having enough time, so you can practice going quicker or try doing the easier questions first so that you don't lose those points.
- Wed Feb 24, 2021 10:36 am
- Forum: Balancing Redox Reactions
- Topic: Sapling #3 for Wk 7 and 8
- Replies: 3
- Views: 253
Re: Sapling #3 for Wk 7 and 8
Mine was a different reaction, but similar steps. You have to split into half-reactions, balance out the oxygens with water, then finish balancing out the hydrogens with H+, and finally add as much OH- as you need on each side to change the H+ to water until everything is balanced. Use the feedback ...
- Wed Feb 24, 2021 8:26 am
- Forum: Balancing Redox Reactions
- Topic: Sapling week 7/8 #8
- Replies: 2
- Views: 461
Sapling week 7/8 #8
Separate this redox reaction into its balanced component half‑reactions. Use the symbol e− for an electron. Cl2+2Cu⟶2CuCl I am having trouble determining the two half-reactions of this. So I think chlorine is the one being oxidized because it is losing electrons because it normally has a -1 charge, ...
- Wed Feb 24, 2021 8:18 am
- Forum: Student Social/Study Group
- Topic: Work and school
- Replies: 16
- Views: 888
Re: Work and school
I struggle with procrastination and thinking I have more time than I do, so I also have planners to help me set tasks for the day and visualize what the days ahead of me will look like. I have a school planner and a general planner to include everything that I might forget about. I also have trouble...
- Fri Feb 19, 2021 10:34 pm
- Forum: Student Social/Study Group
- Topic: Post Midterm 2 De-stressing
- Replies: 92
- Views: 7261
Re: Post Midterm 2 De-stressing
Good luck to all of you that still have midterms! Make sure to relax afterward and do something fun. I will be watching YouTube or baking cookies.
- Wed Feb 17, 2021 9:16 pm
- Forum: Student Social/Study Group
- Topic: points needed for c instead of c minus
- Replies: 7
- Views: 787
Re: points needed for c instead of c minus
I think the grading scale is a standard letter scale up until then. However, last quarter I ended up with a grade higher than I expected based on percentage.
- Wed Feb 17, 2021 9:13 pm
- Forum: Calculating Standard Reaction Entropies (e.g. , Using Standard Molar Entropies)
- Topic: 4I.7 & Sapling #9
- Replies: 2
- Views: 233
Re: 4I.7 & Sapling #9
Entropy (S) is different than change in entropy ( S), which is the change in entropy of the system or surroundings or universe. So use the formulas when finding that change.
S), which is the change in entropy of the system or surroundings or universe. So use the formulas when finding that change.
- Wed Feb 17, 2021 8:59 pm
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: sapling #18 week 5/6
- Replies: 3
- Views: 276
sapling #18 week 5/6
This was the problem given formulas, delta g values in a table, and was asking to find the K value. I was able to solve it using delta Gr= -RTln(K), but I was wondering if anyone knew why for the second reaction, the K value was so large. I thought I had to be doing something wrong, but I checked my...
- Tue Feb 16, 2021 9:46 pm
- Forum: Student Social/Study Group
- Topic: Winter GEs?
- Replies: 33
- Views: 2762
Winter GEs?
So I think I will be taking 14BL in the winter if it's available, but if it's not I want to take a GE. Does anyone have some recommendations for interesting classes? I feel like there are fewer available than I thought. Anyway, I'd love to hear about your experiences!
- Tue Feb 16, 2021 9:43 pm
- Forum: Thermodynamic Systems (Open, Closed, Isolated)
- Topic: Which R value to choose
- Replies: 13
- Views: 1565
Re: Which R value to choose
They each have their own units being used so they are each applicable to their own problems. I had a hard time differentiating the two for a bit until I paid attention to the units.
- Sun Feb 14, 2021 8:39 am
- Forum: Student Social/Study Group
- Topic: Valentine's Day Long Weekend!
- Replies: 30
- Views: 2048
Re: Valentine's Day Long Weekend!
Happy Valentine's Day to everyone! Any plans?
- Sat Feb 13, 2021 10:22 am
- Forum: Entropy Changes Due to Changes in Volume and Temperature
- Topic: Sapling #8 week 5/6
- Replies: 4
- Views: 287
Sapling #8 week 5/6
Calculate the standard molar entropy of vaporization of water at 29.0 °C , given that its standard molar entropy of vaporization at 100.0 °C is 109.0 J⋅K−1⋅mol−1 and the molar heat capacities at constant pressure for liquid water and water vapor are 75.3 J⋅K−1⋅mol−1 and 33.6 J⋅K−1⋅mol−1 , respective...
- Fri Feb 12, 2021 10:04 pm
- Forum: Thermodynamic Definitions (isochoric/isometric, isothermal, isobaric)
- Topic: Sapling Number 5
- Replies: 8
- Views: 479
Re: Sapling Number 5
Thank you all for the help. I was able to solve it using your directions, but why does the feedback say convert Cv to C? Is there another way to solve or am I misinterpreting it?
- Fri Feb 12, 2021 9:57 pm
- Forum: Student Social/Study Group
- Topic: Classes for next quarter?
- Replies: 165
- Views: 16141
Re: Classes for next quarter?
I'm planning on chem 14BL, math 3c, and ls 7b. But if anyone knows of some interesting ge's I'd love to hear them!
- Sun Feb 07, 2021 11:12 am
- Forum: Student Social/Study Group
- Topic: Superbowl
- Replies: 10
- Views: 602
Superbowl
Who do you think will win the Superbowl?
- Sun Feb 07, 2021 10:40 am
- Forum: Ideal Gases
- Topic: Sapling week 3/4 #15
- Replies: 7
- Views: 562
Sapling week 3/4 #15
I was having trouble with this problem:
2NaN3(s)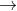 2Na(s) + 3N2(g)
2Na(s) + 3N2(g)
Calculate the value of work for the system if 21.1 g NaN3 reacts completely at 1.00 atm and 22 ∘ C.
I think I'm supposed to use PV=nRT, however I'm not sure how to find the number of moles for N2 to plug in for n.
2NaN3(s)
Calculate the value of work for the system if 21.1 g NaN3 reacts completely at 1.00 atm and 22 ∘ C.
I think I'm supposed to use PV=nRT, however I'm not sure how to find the number of moles for N2 to plug in for n.
- Sun Feb 07, 2021 10:26 am
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: Sapling #8 Week3/4
- Replies: 6
- Views: 349
Re: Sapling #8 Week3/4
You divide the original delta H value by number of moles, so 358.8/4. Then you divide the given kJ value by that value to get the number of moles of CS2 there are. Finally, multiply the number of moles by the molar mass of CS2.
- Sun Jan 31, 2021 7:02 pm
- Forum: Student Social/Study Group
- Topic: Exercising Our Minds and Bodies
- Replies: 120
- Views: 19337
Re: Exercising Our Minds and Bodies
Love this question! I have been swimming almost every day!
- Sun Jan 31, 2021 7:00 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Midterm #1 Results
- Replies: 18
- Views: 914
Re: Midterm #1 Results
I don't think he's said anything yet. I would wait for an email or for our TAs to say anything.
- Wed Jan 27, 2021 8:28 pm
- Forum: General Science Questions
- Topic: Careless Mistakes
- Replies: 54
- Views: 3770
Re: Careless Mistakes
Sometimes it's not really possible, but I try to ask myself if my answer makes sense. If my answer seems too high or low, I double-check the little things like decimal places.
- Wed Jan 27, 2021 8:27 pm
- Forum: Student Social/Study Group
- Topic: Spring 2021
- Replies: 106
- Views: 15019
Re: Spring 2021
Hello! I'm currently taking Chem 14BL right now, and I just want to advise that this class, while not the most difficult material, is a lot of work. So if you think you are already going to have a busy schedule next quarter, you might want to consider whether you can handle this extra workload (wee...
- Wed Jan 27, 2021 11:43 am
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: Using Bond enthalpies
- Replies: 9
- Views: 438
Using Bond enthalpies
For problems where you need to calculate reaction enthalpies using bond enthalpies, is there a standard table of bond enthalpies? When I look them up, there is a wide range of answers, which would have a big effect on the final answer. Thanks.
- Wed Jan 27, 2021 11:39 am
- Forum: Calculating Work of Expansion
- Topic: Difference between reversible and irreversible expansion
- Replies: 4
- Views: 210
Re: Difference between reversible and irreversible expansion
Reversible reactions can go both ways while irreversible ones cannot (once reactants form products it is difficult/impossible to reverse). I think you would only know experimentally, for example wood burning would be irreversible, while melting ice is reversible because you can freeze it again.
- Wed Jan 27, 2021 11:34 am
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: Sapling #5
- Replies: 9
- Views: 323
Re: Sapling #5
Thank you everyone for the help. Why is it that you kept the first equation the same? Is it because it was the longest and could be canceled out by the others easiest?
- Sun Jan 24, 2021 11:04 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Midterm 1 Zoom
- Replies: 8
- Views: 443
Re: Midterm 1 Zoom
Also, do you think we will all be on one large zoom, or further separated like last quarter in our discussion groups?
- Sun Jan 24, 2021 11:02 pm
- Forum: Student Social/Study Group
- Topic: Favorite Music
- Replies: 113
- Views: 11689
Re: Favorite Music
I mostly listen to alternative or indie rock music, but recently I have enjoyed punk rock or 2000's pop-rock. If anyone has suggestions I'd love to hear them.
- Sun Jan 24, 2021 10:58 pm
- Forum: Ideal Gases
- Topic: Acids and Bases
- Replies: 7
- Views: 343
Re: Acids and Bases
Do you mean on the midterm? If so, there is a section on acid and base equilibria, and the corresponding textbook questions are in the syllabus.
- Sun Jan 24, 2021 10:57 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: When to use ICE box
- Replies: 23
- Views: 909
Re: When to use ICE box
You use the ICE box when being asked to find the equilibrium concentration. I think a good way to approach a problem is to see what you are given and what you already know or can determine, and then see what would apply to solve the question.
- Sat Jan 23, 2021 7:02 pm
- Forum: Student Social/Study Group
- Topic: Post All Chemistry Jokes Here
- Replies: 9651
- Views: 3608314
Re: Post All Chemistry Jokes Here
Man: Is it a crime to throw sodium chloride in your enemy's eyes?
Judge: Yes, that's assault.
Man: I know it's a salt, but is it a crime?
Judge: Yes, that's assault.
Man: I know it's a salt, but is it a crime?
- Sat Jan 23, 2021 6:55 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: ICE Tables
- Replies: 36
- Views: 1866
Re: ICE Tables
It only has to do with the coefficients.
- Sat Jan 16, 2021 9:43 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Sapling #5
- Replies: 5
- Views: 270
Re: Sapling #5
Thank you everyone for the discussion on this problem! It helped me a lot. Are there any problems like this in the textbook we can practice?
- Sat Jan 16, 2021 9:25 pm
- Forum: Student Social/Study Group
- Topic: Comfort Movies
- Replies: 168
- Views: 27319
Re: Comfort Movies
I love the nostalgia of watching Jurassic Park! However I mostly watch YouTube or shows on Netflix like New Girl.
- Sat Jan 16, 2021 9:23 pm
- Forum: Calculating the pH of Salt Solutions
- Topic: Textbook Problem 6D.13
- Replies: 2
- Views: 188
Re: Textbook Problem 6D.13
Make sure you calculated the pH, not pOH.
- Sat Jan 16, 2021 9:17 pm
- Forum: Student Social/Study Group
- Topic: New Joke Chain
- Replies: 46
- Views: 3117
Re: New Joke Chain
What do you call an acid with an attitude?
A-mean-oh acid
A-mean-oh acid
- Wed Jan 13, 2021 7:49 pm
- Forum: Administrative Questions and Class Announcements
- Topic: What units will be in Midterm 1?
- Replies: 6
- Views: 293
Re: What units will be in Midterm 1?
I'm guessing we will have time to get to unit 3, and I'm also guessing our TAs will send an email about it too.
- Wed Jan 13, 2021 7:47 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Sapling #9
- Replies: 3
- Views: 286
Re: Sapling #9
Also remember to pay attention to the coefficients and use the right amount of significant figures.
- Wed Jan 13, 2021 7:45 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: pH and pOH conceptually
- Replies: 3
- Views: 136
Re: pH and pOH conceptually
Yes and yes, I believe. Water is necessary for the H3O+ and OH- to form, but water is not included when solving the equation because it is in large excess.
- Sun Jan 10, 2021 3:21 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Sapling #4
- Replies: 4
- Views: 167
Re: Sapling #4
I had different initial values, but that answer sounds too high. If I recall it is asking for the total pressure, so you plug your x value into the equilibrium row of the ice table, and add all three values together. (My answer was 0.0910 bars for reference.)
- Sun Jan 10, 2021 3:18 pm
- Forum: Ideal Gases
- Topic: Advice on how to navigate Chem Community
- Replies: 17
- Views: 921
Re: Advice on how to navigate Chem Community
Good question! I normally use the Active topics link under Quick links in the upper lefthand corner to read and respond to more recent posts. To post a question, you create a post and put it within a category. To see how many posts you have per week, you go to Your posts under Quick links, and I cha...
- Fri Jan 08, 2021 8:53 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Question #2 Sapling
- Replies: 2
- Views: 130
Re: Question #2 Sapling
You will need to set up an ice table. For initial SO3, find concentration by doing moles divided by liters, which were given. The .120 moles O2 at equilibrium is in the O2 column and row 'E'. Use the exponents to fill in the 'C' row with x's. Now you can solve for x given the O2 value. Figure out th...
- Thu Jan 07, 2021 7:41 pm
- Forum: Ideal Gases
- Topic: Bars to atm [ENDORSED]
- Replies: 41
- Views: 1900
Re: Bars to atm [ENDORSED]
My TA said that we will be approximating 1 bar = 1 atmosphere because they are similar enough when converting between them.
- Thu Jan 07, 2021 7:38 pm
- Forum: Administrative Questions and Class Announcements
- Topic: Sapling Assignments
- Replies: 30
- Views: 1927
Re: Sapling Assignments
Those additional ones are optional but were helpful to me when I didn't know how to draw molecules, so they can help prepare you for future assigned Sapling problems.
- Thu Jan 07, 2021 1:53 pm
- Forum: Student Social/Study Group
- Topic: Learning Sessions
- Replies: 24
- Views: 1183
Re: Learning Sessions
I suggest at least a few times per week, but more if you have time. I guess it just depends on if they work for you too. If you can learn independently, then great. But it's helpful to have an experienced student and others to work with. And before midterms and the final is especially helpful. Also,...
- Tue Jan 05, 2021 10:33 am
- Forum: Student Social/Study Group
- Topic: Advice for someone who didn't take 14A with professor Lavelle
- Replies: 61
- Views: 2998
Re: Advice for someone who didn't take 14A with professor Lavelle
There a lot of resources available for extra help. UA sessions are where you can be given practice problems, whereas office hours are where you would ask for specific problem help. Of course, make sure you are doing the sapling, chemistry community, and textbook problems. To get used to the general ...
- Sun Dec 13, 2020 9:44 am
- Forum: Calculating pH or pOH for Strong & Weak Acids & Bases
- Topic: Textbook Question 6B.3
- Replies: 2
- Views: 137
Re: Textbook Question 6B.3
pH is determined by -log([H+]), so for part a you just plug in the .025 M for H+ concentration. For part b you take into account the wrong volume flask. You take the -log of 200/250 times the concentration (.025 M). The actual pH should be slightly less acidic.
- Sat Dec 12, 2020 10:35 am
- Forum: Student Social/Study Group
- Topic: Plans for Relaxing After Finals
- Replies: 98
- Views: 15172
Re: Plans for Relaxing After Finals
I love reading all your responses; they gave me some ideas on what to do. I plan on painting and drawing because it was hard to make time for that during school. Anyone have movie recommendations from Hulu or Netflix?
- Sat Dec 12, 2020 10:32 am
- Forum: Student Social/Study Group
- Topic: Winter Break
- Replies: 44
- Views: 2315
Re: Winter Break
I think I'm going to make flashcards on the main terms and topics because writing them down helps me retain things better. I feel like that requires the least brainpower during a break, because I don't think I will have the motivation to do practice problems.
- Thu Dec 10, 2020 8:04 pm
- Forum: Identifying Acidic & Basic Salts
- Topic: Sapling HW 10 Question 11
- Replies: 6
- Views: 486
Re: Sapling HW 10 Question 11
Based on periodic table trends, an acid is weaker when their bond to H is stronger, because it is harder to give up an H+.
- Thu Dec 10, 2020 8:01 pm
- Forum: Bronsted Acids & Bases
- Topic: Sapling HW Week 10 #1
- Replies: 3
- Views: 311
Re: Sapling HW Week 10 #1
Vivian that is a great suggestion! Yeah I think you have to memorize the strong acids and bases, and the rest will all be weak.
- Sun Dec 06, 2020 6:56 pm
- Forum: *Molecular Orbital Theory (Bond Order, Diamagnetism, Paramagnetism)
- Topic: Sigma and Pi bond rotation
- Replies: 5
- Views: 1153
Re: Sigma and Pi bond rotation
Yes I think so because I remember him saying that the pi bonds don't allow rotation because it connects at the two ends, and rotation would break the bond, so for any double or triple bonds that would happen.
- Sun Dec 06, 2020 6:54 pm
- Forum: Student Social/Study Group
- Topic: Point distribution
- Replies: 11
- Views: 679
Re: Point distribution
Do not let earning a grade lower than an A discourage you! These tests do not define how much you have learned or are capable of! :) I totally agree! It's too late for me to get an A now considering my past exams, but I've accepted it. I'm still working hard to get a B, even though I wanted to just...
- Sun Dec 06, 2020 6:50 pm
- Forum: Student Social/Study Group
- Topic: How are you?
- Replies: 154
- Views: 14233
Re: How are you?
Surprisingly okay. Just focusing on studying for finals and staying optimistic for the time I can finally go to campus.
- Sun Nov 29, 2020 9:50 am
- Forum: Determining Molecular Shape (VSEPR)
- Topic: 120 degree bond angles
- Replies: 3
- Views: 189
Re: 120 degree bond angles
I found this table to be really useful for studying shapes and angles. https://8dd969e7-a-ff053077-s-sites.googlegroups.com/a/coe.edu/principles-of-structural-chemistry/chemistry-and-climate-change/molecular-shapes/CD%20Spectrum.png?attachauth=ANoY7crF6LZuLn5XS4eaA7LRqj1A37qe5pqikDjjrIUlsarDWzmRkpKP...
- Sun Nov 29, 2020 9:46 am
- Forum: General Science Questions
- Topic: Electron Affinity
- Replies: 4
- Views: 174
Re: Electron Affinity
Electron affinity is the change in energy when an electron is added and defines how likely that atom is to gain an electron, so the noble gases have full valence shells and don't want any more electrons so they have an affinity of 0. It is true that they can gain electrons in the same shell, but I t...
- Sun Nov 29, 2020 9:38 am
- Forum: Hybridization
- Topic: Confusion on certain carbon hybridizations
- Replies: 5
- Views: 389
Re: Confusion on certain carbon hybridizations
Each carbon atom would be sp2 because there are three regions of electron density (the two single bonds and the one double bond). You have to count that double bond for the other carbon too. Having a visual makes it much easier to see the regions of electron density for any atom. And this works for ...
- Sun Nov 29, 2020 9:35 am
- Forum: Student Social/Study Group
- Topic: How to study for class
- Replies: 30
- Views: 1346
Re: How to study for class
Hello, I only use the textbook for practice problems, but those are very helpful. To help understand concepts, I like to watch Youtube videos to hear other people teaching me. I really like The Organic Chemistry Tutor, Professor Dave Explains, and CrashCourse.
- Sun Nov 22, 2020 1:08 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: London Dispersion
- Replies: 33
- Views: 1887
Re: London Dispersion
Yes, london dispersion forces are always present, but are very weak until they add up when many molecules are present.
- Sun Nov 22, 2020 1:05 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Molecular Shape Polarity
- Replies: 3
- Views: 110
Re: Molecular Shape Polarity
The same shape molecule can be polar or nonpolar, so a certain shape is not always one or the other.
Table of molecular shapes being polar or nonpolar:
[url]
https://www.pinterest.com/pin/215117319680930383/[/url]
Table of molecular shapes being polar or nonpolar:
[url]
https://www.pinterest.com/pin/215117319680930383/[/url]
- Sun Nov 22, 2020 1:01 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Visualizing VSEPR Shapes
- Replies: 5
- Views: 193
Re: Visualizing VSEPR Shapes
I would practice drawing them the geometric way with the bolded wedge and dashed wedge, instead of just lines, so that you can keep visualizing each molecule.
- Fri Nov 20, 2020 7:54 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Bond Angle
- Replies: 9
- Views: 487
Re: Bond Angle
Yes, I think all molecular shape bond angles can differ slightly from what is expected based on what atoms are in it. For example, SF4 has a see-saw geometry but has bond angles of 173 degrees and 102 degrees.
- Fri Nov 20, 2020 2:37 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: VSEPR notation
- Replies: 9
- Views: 366
Re: VSEPR notation
I think it can give us a general idea of the shape, but is not always quantitatively accurate, because if the surrounding atoms are not all the same, the bond angles will differ slightly than 'expected'.
- Mon Nov 16, 2020 7:22 pm
- Forum: Electron Configurations for Multi-Electron Atoms
- Topic: Problem 2A.17
- Replies: 2
- Views: 278
Re: Problem 2A.17
Well remember a full valence shell would be 8, so the answer is going to be 8 or less. The way I thought of it was the 3d^3 is the description of the last electrons of the electron configuration, so there must be 3 valence electrons. Another way to think of it is that Mn would normally have 7 valenc...
- Sun Nov 15, 2020 8:11 pm
- Forum: Ionic & Covalent Bonds
- Topic: D orbitals
- Replies: 3
- Views: 181
Re: D orbitals
The ground state electron configuration of Ag is [Kr] 5s^{2}4d^{9} , however its actual configuration based on experimentation is [Kr] 5s^{1}4d^{10} so Ag(+1) would be [Kr] 4d^{10} because you take away one electron to make it a positive charge, which you take away from the s subshell because it is ...
- Fri Nov 13, 2020 11:18 am
- Forum: Dipole Moments
- Topic: Boiling point of molecules
- Replies: 5
- Views: 326
Re: Boiling point of molecules
The way I think of it is that the stronger the intermolecular bonds, the more energy is required to break those bonds. Heat is a form of energy that will break those bonds and cause it to change states (liquid to gas phase change). This is why hydrogen bonds between water are so important, because i...
- Mon Nov 09, 2020 8:57 pm
- Forum: Student Social/Study Group
- Topic: Mid-quarter Check in
- Replies: 67
- Views: 3570
Re: Mid-quarter Check in
Hey! I'm honestly really struggling, especially with this class. I only took the basic high school chemistry and took calculus instead of physics, so I find this content very confusing and difficult. I have never struggled with a class like this and I feel like it's even worse through online. I fee...
- Mon Nov 09, 2020 8:52 pm
- Forum: Student Social/Study Group
- Topic: Step-Up vs. Drop-In Sessions
- Replies: 7
- Views: 431
Re: Step-Up vs. Drop-In Sessions
I like step-up sessions because they have prepared problems from the topic you are currently on. Drop-in is more for if you have a specific question like a textbook problem you wanted help on. Each has its advantages depending on what you need.
- Mon Nov 09, 2020 8:50 pm
- Forum: Bond Lengths & Energies
- Topic: Electron Pairs Interacting
- Replies: 3
- Views: 205
Re: Electron Pairs Interacting
Because electrons all have a negative charge, they will repel each other. This is also why positively charged protons attract electrons.
- Sun Nov 08, 2020 9:46 pm
- Forum: Student Social/Study Group
- Topic: Studying From Home
- Replies: 91
- Views: 6821
Re: Studying From Home
I also find it hard to study because of distractions at home, such as my family members, noise, my phone, etc. I try to isolate myself from distractions as best as possible and PLAN what I am going to do and by when. Making a plan is like creating short term goals for the day, and will motivate you ...
- Fri Nov 06, 2020 9:41 pm
- Forum: Student Social/Study Group
- Topic: Making it through Midterm Results
- Replies: 13
- Views: 765
Making it through Midterm Results
So I'll admit I did not do very well on the midterm. I missed some conceptual problems and also made errors in my calculations that led to wrong answers. Congrats to everyone who did do well though! If you also didn't do well, how do you motivate yourself or make yourself feel better? If you did do ...
- Fri Nov 06, 2020 9:31 pm
- Forum: Lewis Structures
- Topic: Lecture #16 Question
- Replies: 2
- Views: 106
Re: Lecture #16 Question
Silver halides are just an exception to the rules about covalent bonds, that you couldn't figure out on your own without experimentation. So you are correct if you refer to other bonds and their characteristics.
- Thu Nov 05, 2020 10:01 pm
- Forum: Lewis Structures
- Topic: How to assign formal charge
- Replies: 3
- Views: 249
Re: How to assign formal charge
For finding formal charge in general, I found this video really helpful. It has multiple examples and is easy to understand.
https://youtu.be/C2l-76VP8s0
https://youtu.be/C2l-76VP8s0
- Wed Nov 04, 2020 4:59 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: Orbital vs Subshell
- Replies: 7
- Views: 532
Re: Orbital vs Subshell
The orbital is the magnetic quantum number ( m_{l} ) whereas the subshell is the angular momentum quantum number (l). Orbital is given in terms from -l...0...l, and describes the pathway of an electron in a certain shell, while subshell is the l value, and is a math function that describes the wave-...
- Tue Nov 03, 2020 7:11 pm
- Forum: Quantum Numbers and The H-Atom
- Topic: Are there subshells past f?
- Replies: 28
- Views: 1158
Re: Are there subshells past f?
I recall Professor Lavelle saying that there are also g, h, i (and so on) subshells, but we won't need to worry about that, especially in this class.
- Tue Nov 03, 2020 7:08 pm
- Forum: Electron Configurations for Multi-Electron Atoms
- Topic: valence electrons in d block
- Replies: 3
- Views: 132
Re: valence electrons in d block
Remember that when you start writing the electron configurations for d-block metals that the d-block goes before the s-block. So d-block metals will usually have 2 valence electrons because of this. There are some exceptions where there is only 1 valence electron, for example, copper, which is why i...
- Tue Nov 03, 2020 7:01 pm
- Forum: Electron Configurations for Multi-Electron Atoms
- Topic: Formation of Ions
- Replies: 5
- Views: 315
Re: Formation of Ions
Yes, you are correct. You fill up the 4s orbital first because it has lower energy and orbitals are filled from lowest to highest energy. Once the d orbital is full, it has more energy now and 4s is no longer the lowest energy, so 3d electrons must be removed first. Essentially 4s is only lower when...
- Mon Nov 02, 2020 7:36 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: Lecture Concept Question
- Replies: 4
- Views: 107
Re: Lecture Concept Question
Orbitals are like a math function because it is like a prediction and description for its location in relation to the nucleus. I think of it as a coordinate system where the nucleus is the origin. He doesn't want us to think of it like hotel rooms because the three quantum numbers are not the litera...
- Sun Nov 01, 2020 9:53 am
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: Ground State Electron Configuration
- Replies: 4
- Views: 179
Re: Ground State Electron Configuration
You can tell it is excited if the electron configuration has a section that is not 'full'. For example, if you were given 1s^{2}2s^{2}2p^{6}3s^{1}3p^{2} , you may realize that the 3s^{1} is not full, and should be 3s^{2} if it were in its ground state, yet the electron configuration keeps going. Thi...
- Sun Nov 01, 2020 9:39 am
- Forum: Student Social/Study Group
- Topic: Favorite TV shows
- Replies: 277
- Views: 40058
Re: Favorite TV shows
I loved Gravity Falls as a kid and still do now. Some great shows I've watched recently are New Girl, Sherlock, and Bojack Horseman. I also plan on watching Brooklyn 99.
- Wed Oct 28, 2020 8:17 pm
- Forum: Photoelectric Effect
- Topic: Photoelectric Effect post module
- Replies: 4
- Views: 220
Re: Photoelectric Effect post module
This looks like the same example Professor Lavelle did in his lecture on October 14. You rearrange E=hv and c=lambda*v to get lambda=(hc)/E. E is given and h and c are constants.
- Wed Oct 28, 2020 8:06 pm
- Forum: Properties of Electrons
- Topic: Sapling hw #13
- Replies: 3
- Views: 144
Re: Sapling hw #13
I was also having trouble with this problem but I figured it out. After rearranging the de Broglie equation and E=1/2mv^2, you should get ^{2}) to solve for the energy of the electron.
to solve for the energy of the electron.
- Wed Oct 28, 2020 6:07 pm
- Forum: DeBroglie Equation
- Topic: sapling hw problem
- Replies: 5
- Views: 499
sapling hw problem
The E. coli bacterium is about 1.9 μm long. Suppose you want to study it using photons of that wavelength or electrons having that de Broglie wavelength. What is the energy of the electron? I figured out the answer to the first part of the problem, but I'm having trouble with this part. The feedback...
- Sat Oct 24, 2020 12:47 am
- Forum: Heisenberg Indeterminacy (Uncertainty) Equation
- Topic: 1B.25
- Replies: 3
- Views: 173
1B.25
What is the minimum uncertainty in the speed of an electron confined within a lead atom of diameter 350. pm? Model the atom as a one-dimensional box with a length equal to the diameter of the actual atom. I was trying to solve a problem using the equation \bigtriangleup p\bigtriangleup x\geq \frac{h...
- Wed Oct 21, 2020 6:09 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: Homework up to Q19 and Midterm 1 [ENDORSED]
- Replies: 7
- Views: 270
Re: Homework up to Q19 and Midterm 1 [ENDORSED]
I am wondering if sapling is not in the same order for everyone because some of my questions 1-19 do cover material we have not yet learned such as quantum numbers and some questions past 19 cover material that we have learned such as DeBroglie. Is there anyway that a list of the specific questions...
- Wed Oct 21, 2020 4:27 pm
- Forum: Properties of Electrons
- Topic: Properties of Electrons and Matter as a Whole
- Replies: 5
- Views: 272
Re: Properties of Electrons and Matter as a Whole
Yes that is correct. Electrons have wavelike properties because they can be diffracted by light, and particle-like properties because they have a mass that can collide with other masses (other particles/electrons). The reason larger objects in classical mechanics do not possess wavelike properties i...
- Wed Oct 21, 2020 9:39 am
- Forum: Administrative Questions and Class Announcements
- Topic: Respondus Setup factored in Midterm time?
- Replies: 2
- Views: 121
Re: Respondus Setup factored in Midterm time?
I highly doubt they would ever take away time from the midterm from us. I took a Respondus test for another class and the timer at the top didn't start until I finished taking the pictures and setting up. Also, my TA said if we want we should show up early to the discussion so that we can set up, an...
- Wed Oct 21, 2020 9:33 am
- Forum: DeBroglie Equation
- Topic: Sapling Question #10
- Replies: 8
- Views: 611
Re: Sapling Question #10
Use lambda=h/mv. h is a constant and velocity is given to us. To find m, divide the mass of two fluorine molecules by Avogadro's number to get the mass of one particle, then convert that value to kg because h, Planck's constant, is in units of Js, or kg*(m^2)*(s^-2)*s. Lastly, remember to use the co...
- Wed Oct 21, 2020 9:20 am
- Forum: Empirical & Molecular Formulas
- Topic: Diatomic Elements
- Replies: 3
- Views: 182
Re: Diatomic Elements
I believe the problem will state it is diatomic.
- Mon Oct 19, 2020 11:25 pm
- Forum: Einstein Equation
- Topic: Sapling homework Question 9
- Replies: 3
- Views: 382
Re: Sapling homework Question 9
Whenever I am given a problem I always see what is given and what I am being asked to find. We are given the value for energy and we are asked to find the wavelength. We can use E=hc/lambda, rewrite that as lambda=hc/E, then solve for lambda because h and c are constants. Just make sure to convert y...
- Sat Oct 17, 2020 8:12 pm
- Forum: Properties of Light
- Topic: Frequency vs Wavelength
- Replies: 22
- Views: 1306
Frequency vs Wavelength
Hey everyone I have a clarification question. I know that we organize and separate waves by their wavelength, but does the frequency have any effect on what type of wave it is? I understand what each term means and its significance to solving equations, but I was just wondering if frequency has a di...
- Sat Oct 17, 2020 8:04 pm
- Forum: Student Social/Study Group
- Topic: How are you studying?
- Replies: 204
- Views: 20442
Re: How are you studying?
From what I've noticed Professor Lavelle stresses doing the homework problems which are on the syllabus. When I do the problems, I make sure not to check the textbook answer guide until I am done so that I'm not relying on it subconsciously. Also of course going to the drop-in sessions, office hours...
- Sat Oct 17, 2020 8:00 pm
- Forum: Administrative Questions and Class Announcements
- Topic: STEP UP SESSIONS
- Replies: 3
- Views: 89
Re: STEP UP SESSIONS
I have not attended a full one, but instead I like to pop in and listen. They were doing practice problems one at a time, chosen by the UA.
- Sat Oct 17, 2020 7:55 pm
- Forum: Heisenberg Indeterminacy (Uncertainty) Equation
- Topic: Midterm #1
- Replies: 3
- Views: 152
Re: Midterm #1
It's a bit late, but someone posted these videos about the Heisenberg Uncertainty Principle. by Mackenzie Van Valkenburgh 3G » Sat Oct 17, 2020 7:25 pm These videos give some background information and explanations of the Heisenberg Uncertainty Principle. I found them helpful, so hopefully someone e...
- Sat Oct 17, 2020 7:49 pm
- Forum: Properties of Light
- Topic: Conversions of Units and Equations
- Replies: 3
- Views: 115
Re: Conversions of Units and Equations
I think we will be allowed to use the one on his website, but I'm not sure about the conversions. I would just know m to nm because it is common for the quantum questions.
- Tue Oct 06, 2020 4:21 pm
- Forum: Limiting Reactant Calculations
- Topic: Sapling HW 10
- Replies: 3
- Views: 141
Re: Sapling HW 10
Thank you Mary, I finally got it!

