Search found 100 matches
- Thu Mar 11, 2021 10:27 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Anode<Cathode
- Replies: 1
- Views: 175
Re: Anode<Cathode
The E should be positive whenever it is considered a galvanic cell. I don't think the E changes with the concentration of the anode/cathode. Since the batteries undergo one reaction at a time, we can determine whether E is positive or negative when we calculate their individual E's.
- Thu Mar 11, 2021 10:10 pm
- Forum: Gibbs Free Energy Concepts and Calculations
- Topic: Textbook Problem 4.43
- Replies: 3
- Views: 399
Re: Textbook Problem 4.43
Yes, it is an error. You can probably just think of it as STP so you have the same calculations as the manual.
- Thu Mar 11, 2021 9:46 pm
- Forum: Calculating Work of Expansion
- Topic: -PDeltaV
- Replies: 2
- Views: 296
Re: -PDeltaV
Remember that the P in w=-PDeltaV signifies external pressure. When something undergoes a reversible expansion, they change the external pressure and internal pressure with infinitely small intervals. This is why reversible expansions take more work.
- Thu Mar 11, 2021 9:33 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: 4E #7b
- Replies: 3
- Views: 386
Re: 4E #7b
Remember that when you break bonds you have a positive enthalpy while when you form bonds you get a negative enthalpy. So the bonds formed should subtract from the bonds broken.
- Thu Mar 11, 2021 9:19 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Ignoring second deprotonations
- Replies: 5
- Views: 698
Re: Ignoring second deprotonations
I think you can ignore the 2nd deprotonation when the 2nd deprotonation is less than a factor of 10^3 from the first deprotonation. It says this in 6E.1 if you want to learn more. for example: tartaric acid (weak acid), C2H4O2(COOH)2 ka1= 6.0×10^−4 ka2 = 1.5×10^−5 in this case, you will do the 2nd d...
- Sun Mar 07, 2021 3:38 pm
- Forum: General Rate Laws
- Topic: half life
- Replies: 2
- Views: 209
Re: half life
You can determine it based on the time and concentration it took to do a half-life. Notice that the time increased as the concentration decreased. With this, you can use the half-life equations to figure out which order has this trend.
- Sun Mar 07, 2021 3:21 pm
- Forum: Second Order Reactions
- Topic: Textbook Question 7B 13
- Replies: 2
- Views: 344
Re: Textbook Question 7B 13
I think it has to do with the rate flatting out as the reaction goes on in a 2nd order reaction. The 2nd order graph has a decaying exponential line. Correct me if I'm wrong but this is my guess.
- Sun Mar 07, 2021 2:59 pm
- Forum: General Rate Laws
- Topic: 7B9
- Replies: 2
- Views: 213
Re: 7B9
We would subtract .006 mols L-1 from 0.015 mols L-1 because that is how many A's are needed to create 0.018 mols L-1 of B. We want to know the [A]t so we would subtract the amount of A needed to create .018 mols L-1 of B from the initial A condition. Note that A is still present in the solution at 3...
- Sun Mar 07, 2021 2:49 pm
- Forum: General Rate Laws
- Topic: State
- Replies: 1
- Views: 159
Re: State
I think that for the first, second, and zero-order equations it doesn't matter since we are only considering the reactants. It will likely matter when we consider the whole equation.
- Sun Mar 07, 2021 2:35 pm
- Forum: Experimental Details
- Topic: Determining Rate Law of a Reaction With Multiple Reactants
- Replies: 2
- Views: 375
Re: Determining Rate Law of a Reaction With Multiple Reactants
Yes. I think it is also due to the fact that the other elements will be so large that their changes will be negligible. If the other two elements are negligible, we can determine that the change in its rate is only based on the element with the small concentration.
- Sat Feb 27, 2021 6:06 pm
- Forum: Appications of the Nernst Equation (e.g., Concentration Cells, Non-Standard Cell Potentials, Calculating Equilibrium Constants and pH)
- Topic: week 8 sapling #17
- Replies: 2
- Views: 223
Re: week 8 sapling #17
I think the section with the closest problems is in 6N but it might be confusing. For this problem, you can start by looking at the info given to you. S.H.E.'s reduction eq= 2H+(aq)+2e−⇀ H2(g) E(nought)= 0 With [H+]=0.46 [MPH2]=4.9 atm, you can find Q (remember [P]^coefficent/[R]^c) With this, you h...
- Sat Feb 27, 2021 5:46 pm
- Forum: Work, Gibbs Free Energy, Cell (Redox) Potentials
- Topic: Sapling Week 7-8 #12
- Replies: 3
- Views: 241
Re: Sapling Week 7-8 #12
You don't need to multiply the E(c & a) by the coefficients. You should get 3.878 V. Apparently, a standard reduction potential is an intensive property and it does not depend on how many times the reaction occurs. My Work Au3+(aq) + 3e– → Au(s) E=+1.498 (cathode) Mg2+(aq) + 2e– → Mg(s) E=–2.38 ...
- Sat Feb 27, 2021 5:37 pm
- Forum: Work, Gibbs Free Energy, Cell (Redox) Potentials
- Topic: Sapling #9
- Replies: 3
- Views: 262
Re: Sapling #9
The information you should look at is "galvanic cell." Galvanic cells undergo a spontaneous redox reaction so that means that the E should be positive (remember that Delta G = - n*F*E). With this, you can find which combination gives you a positive E. Useful info Au3+(aq) + 3e– → Au(s) +1....
- Sat Feb 27, 2021 5:25 pm
- Forum: Interesting Applications: Rechargeable Batteries (Cell Phones, Notebooks, Cars), Fuel Cells (Space Shuttle), Photovoltaic Cells (Solar Panels), Electrolysis, Rust
- Topic: Cathodes and Anodes
- Replies: 4
- Views: 303
Re: Cathodes and Anodes
You can also tell by looking at the equations themselves. The anodes will undergo oxidation while the cathodes undergo reduction.
- Sat Feb 27, 2021 5:15 pm
- Forum: Kinetics vs. Thermodynamics Controlling a Reaction
- Topic: Diamond vs. Graphite
- Replies: 23
- Views: 1164
Re: Diamond vs. Graphite
Yeah, it will take billions of years because the energy absorbed from STP is way too low.
According to a study done by a physicist in India, the graphite needed to experience over 100 kbars of pressure and over 2000 C of temperature for it to turn into a diamond.
According to a study done by a physicist in India, the graphite needed to experience over 100 kbars of pressure and over 2000 C of temperature for it to turn into a diamond.
- Sun Feb 21, 2021 3:45 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Salt Bridge
- Replies: 6
- Views: 387
Re: Salt Bridge
I think the salt bridge has three main functions: - preventing the accumulation of positive and negative charges around the respective electrodes. in other words, keeping the reaction neutral - allowing a continual flow of electrons between the two sides. - limits/prevents the liquids of each side f...
- Sun Feb 21, 2021 3:36 pm
- Forum: Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams
- Topic: Salt Bridge
- Replies: 5
- Views: 316
Re: Salt Bridge
A salt bridge is used to maintain the charge balance of the reduction and oxidation of the battery. If it is removed, the reaction will likely continue in their respective halves of the cell and will become more negatively/positively charged. This leads the battery to eventually come to equilibrium ...
- Sun Feb 21, 2021 3:26 pm
- Forum: Balancing Redox Reactions
- Topic: Sapling Week 7/8 #2
- Replies: 6
- Views: 2276
Re: Sapling Week 7/8 #2
Just like Anna said, you would be adding the two half-reactions. When deciding on how many moles of an ion there are, I like looking at the number of electrons being exchanged and adjust the moles based on that. In this reaction, since Sn loses 2 electrons, you will need 2 moles of Ags to take up th...
- Sun Feb 21, 2021 3:22 pm
- Forum: Balancing Redox Reactions
- Topic: Electrolyte Used [ENDORSED]
- Replies: 4
- Views: 305
Re: Electrolyte Used [ENDORSED]
I think as of now, the most important thing to focus on is the number of electrons being exchanged and how to manipulate the electrolytes to be able to do that.
- Sun Feb 21, 2021 3:03 pm
- Forum: Balancing Redox Reactions
- Topic: Sapling HW 7/8 #1
- Replies: 8
- Views: 1422
Re: Sapling HW 7/8 #1
Oxidation numbers are used to find how many electrons are removed/gained in an atom in a given molecule. I wanted to share this website since it really helped me understand how to find these values. https://chem.libretexts.org/Bookshelves/Analytical_Chemistry/Supplemental_Modules_(Analytical_Chemist...
- Sat Feb 13, 2021 10:34 pm
- Forum: Concepts & Calculations Using Second Law of Thermodynamics
- Topic: kB
- Replies: 3
- Views: 277
Re: kB
k = 1.381 x 10^-23 J/K
You can find the Boltzmann's constant on the constants and formulas sheet on the top right (it says k but kB and k are the same thing).
You can find the Boltzmann's constant on the constants and formulas sheet on the top right (it says k but kB and k are the same thing).
- Sat Feb 13, 2021 10:28 pm
- Forum: Calculating Work of Expansion
- Topic: q and w
- Replies: 3
- Views: 240
Re: q and w
ΔU=q+w which comes from the first law of thermodynamics. q = the transfer of the thermal energy. The energy usually goes from an object of high temperature to an object of low temperature. q is negative when the heat is released by the system. (exothermic) q is positive when the heat is absorbed int...
- Sat Feb 13, 2021 10:10 pm
- Forum: Calculating Work of Expansion
- Topic: calculating irreversible work of any gas vs ideal isothermal gas
- Replies: 1
- Views: 255
Re: calculating irreversible work of any gas vs ideal isothermal gas
Yes, you would have to use different equations to figure the amount of work done. For irreversible rxns, you would use dw= -p*dV or w =- Sp dV (S = integral). For reversible isothermal gas rxns, you would use w= -nRTln(V1/V2). The difference in these equations arises from the differences in p in the...
- Sat Feb 13, 2021 9:56 pm
- Forum: Thermodynamic Definitions (isochoric/isometric, isothermal, isobaric)
- Topic: Reversible Pathway
- Replies: 2
- Views: 213
Re: Reversible Pathway
Adiabatic compression/expansion of gases is also a reversible pathway. It transfers energy only through work.
- Sat Feb 13, 2021 9:41 pm
- Forum: Concepts & Calculations Using Second Law of Thermodynamics
- Topic: Textbook Question 4H.11 part d
- Replies: 1
- Views: 111
Re: Textbook Question 4H.11 part d
They say that because the entropy of the system decreased. Entropy is referred to as the measure of the disorder of a system, so if it is decreased there is more stability in the system. This difference in stability largely arises from the KCl's(s) lower entropy.
- Sat Feb 06, 2021 4:25 pm
- Forum: Concepts & Calculations Using First Law of Thermodynamics
- Topic: Work
- Replies: 4
- Views: 357
Re: Work
Lorena_Morales_1K wrote:What exactly does it mean when work is positive? Thank you in advance for your help :)
Work is positive when done on a system.
Work is negative when done by the system.
I recommend this video for more info.
https://youtu.be/NyOYW07-L5g
- Sat Feb 06, 2021 4:16 pm
- Forum: Thermodynamic Systems (Open, Closed, Isolated)
- Topic: Sapling 14
- Replies: 2
- Views: 271
Re: Sapling 14
It has to do with the pressure value. The isothermal equation is derived from the original w=-PΔV equation. The reason w = -PΔV works is because the P (a constant) can be taken out of the integral in −∫pdV. However, in an isothermal reaction, the external pressure isn't constant so we can't take it ...
- Sat Feb 06, 2021 3:33 pm
- Forum: Thermodynamic Definitions (isochoric/isometric, isothermal, isobaric)
- Topic: isothermal reactions
- Replies: 9
- Views: 496
Re: isothermal reactions
To add on, I like to use the "thermal" in isothermal to help myself remember that it is related to temperature. In an isothermal reaction, the temperature is constant. I hope this helps!
- Fri Feb 05, 2021 9:42 pm
- Forum: Concepts & Calculations Using First Law of Thermodynamics
- Topic: Relationship between cv and cp
- Replies: 2
- Views: 124
Re: Relationship between cv and cp
Cv = ΔU/ΔT is the heat capacity at a constant volume Cp = ΔH/ΔT is the heat capacity at a constant pressure In order to find the relationship between these heat capacities, we need to dissect the ΔH. ΔH = ΔU + PV PV can be changed to nRΔT bc PV = nRΔT so, ΔH = ΔU + nRΔT is formed. Cp = ΔH/ΔT = (nRΔT...
- Fri Feb 05, 2021 9:16 pm
- Forum: Concepts & Calculations Using First Law of Thermodynamics
- Topic: 4B.3 HW Problem
- Replies: 4
- Views: 219
Re: 4B.3 HW Problem
I think the textbook is wrong. When we went over this in my discussion section, we found that work was 490J.
w= change of U - q = 982J - 492J = 490J.
w= change of U - q = 982J - 492J = 490J.
- Sat Jan 30, 2021 3:55 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: Sapling Question 14
- Replies: 2
- Views: 236
Re: Sapling Question 14
I think that it has to do with the changing pressure of the external pressure. If we were dealing with one pressure, we can take it out of the integral (since it's a constant) and solve it with the equation given to us in class. That is why we do w= p *change of volume. But now since P is changing, ...
- Sat Jan 30, 2021 3:32 pm
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: Question 10 Sapling
- Replies: 2
- Views: 103
Re: Question 10 Sapling
In order to solve this problem, you will need to use this equation. heat required to melt ice + heat required to bring the grams of melted ice to the final temperature=(negative) heat released that cools grams of water to the final temperature For heat required to melt ice - use the standard enthalp...
- Sat Jan 30, 2021 3:19 pm
- Forum: Concepts & Calculations Using First Law of Thermodynamics
- Topic: Finding the value of q
- Replies: 2
- Views: 234
Re: Finding the value of q
Like the post above me, you should use the equation ΔU = Q + W. However, I think that the signs of each value (Q, W and U) depend on the problem. For work: w = + when work is done "on the system" w= - when work is done "by the system" for q: when heat is absorbed by the system it...
- Sat Jan 30, 2021 3:02 pm
- Forum: Thermodynamic Systems (Open, Closed, Isolated)
- Topic: Ways of Changing the Energy in the System
- Replies: 5
- Views: 225
Re: Ways of Changing the Energy in the System
I think you should watch this video. It has really helped me get a grasp of this concept.
https://youtu.be/LsqKL3pBVMA
https://youtu.be/LsqKL3pBVMA
- Sat Jan 30, 2021 2:56 pm
- Forum: Thermodynamic Systems (Open, Closed, Isolated)
- Topic: Sapling HW Week 3 & 4 #10
- Replies: 3
- Views: 159
Re: Sapling HW Week 3 & 4 #10
You should set up the problem with the equation below to solve for Tf.
heat required to melt ice (by doing what Amanda said) + heat required to bring the 53.6 g of melted ice to the final temperature = (negative) heat released that cools 364 g of water to the final temperature
heat required to melt ice (by doing what Amanda said) + heat required to bring the 53.6 g of melted ice to the final temperature = (negative) heat released that cools 364 g of water to the final temperature
- Sun Jan 24, 2021 12:22 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: Standard State
- Replies: 3
- Views: 349
Re: Standard State
I think that the standard state for complex molecules will be given to us. If you want to learn about the standard state of the pure elements, I recommend looking at this site and changing the temperature to 25C.
https://ptable.com/#Properties
https://ptable.com/#Properties
- Sun Jan 24, 2021 12:14 pm
- Forum: Phase Changes & Related Calculations
- Topic: Leidenfrost Effect
- Replies: 3
- Views: 264
Re: Leidenfrost Effect
I think that the Leidenfrost effect happens when an object of extremely hot temperature (the lava in this case) touches a liquid. The extreme temperature difference between the lava and the sweat or water on the skin causes the water to form a vapor layer that shields the skin from direct contact wi...
- Sun Jan 24, 2021 11:59 am
- Forum: Heat Capacities, Calorimeters & Calorimetry Calculations
- Topic: converting L*atm to joules
- Replies: 2
- Views: 81
Re: converting L*atm to joules
We'll have access to all of these conversions on the constants and formulas sheet.
Here's a link
https://lavelle.chem.ucla.edu/wp-content/supporting-files/Chem14B/Constants_Equations.pdf
Here's a link
https://lavelle.chem.ucla.edu/wp-content/supporting-files/Chem14B/Constants_Equations.pdf
- Sun Jan 24, 2021 11:55 am
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: Direction of arrows
- Replies: 11
- Views: 568
Re: Direction of arrows
I think that the one-sided arrow just tells us that the reaction is heavily favored to that side. For these questions, I think we can assume that the reactants are completely used. I think that it was done to make the calculations easier.
- Sun Jan 24, 2021 11:46 am
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: Best Method for Finding Enthalpy Changes
- Replies: 6
- Views: 313
Re: Best Method for Finding Enthalpy Changes
I think the only distinction we need to make is that bond enthalpy can be inaccurate (except for diatomic elements) because it is the average. The other two seem to be as accurate as each other so the method that you use will largely depend on the information given.
- Sun Jan 17, 2021 1:51 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: 6E.3 Question
- Replies: 2
- Views: 211
Re: 6E.3 Question
The 2nd deprotonation is when the acid loses another H after the first reaction.
For example, when H2SO4 reacts with water, the 2nd deprotonation will be when HSO4- turns into SO4 2-
Sometimes we are able to ignore the 2nd ka because the change to the pH is so small.
For example, when H2SO4 reacts with water, the 2nd deprotonation will be when HSO4- turns into SO4 2-
Sometimes we are able to ignore the 2nd ka because the change to the pH is so small.
- Sun Jan 17, 2021 1:38 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Diprotic Acids
- Replies: 2
- Views: 121
Re: Diprotic Acids
To add on, I think you have to consider the 2nd ka value when doing strong acids like Sulfuric acid H2SO4.
- Sun Jan 17, 2021 1:24 pm
- Forum: Ideal Gases
- Topic: Inverse Kc [ENDORSED]
- Replies: 41
- Views: 2476
Re: Inverse Kc [ENDORSED]
You will typically use the inverse Kc when solving for the reverse equation.
For example in the reaction 2SO3 = 2SO2 +O2.
kc will equal ([SO2]^2[02])/[SO3]^2
The inverse kc will equal [SO3]^2/([SO2]^2[02])
For example in the reaction 2SO3 = 2SO2 +O2.
kc will equal ([SO2]^2[02])/[SO3]^2
The inverse kc will equal [SO3]^2/([SO2]^2[02])
- Sun Jan 17, 2021 1:17 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: sapling week 1/2 #2
- Replies: 3
- Views: 161
Re: sapling week 1/2 #2
A couple of places you should check:
- whether you changed moles to molar concentration
- whether your kc formula is correct = (SO2)^2*(O2)/SO3^2
- whether the concentrations of the final values are correct
- remember that the change for the ice table will be -2x = 2x x
- whether you changed moles to molar concentration
- whether your kc formula is correct = (SO2)^2*(O2)/SO3^2
- whether the concentrations of the final values are correct
- remember that the change for the ice table will be -2x = 2x x
- Sat Jan 16, 2021 9:34 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Textbook Question 6E.1
- Replies: 2
- Views: 136
Re: Textbook Question 6E.1
Since H2SO4 can deprotonate 2 times you will have to make an ice table for the weak acid HSO4-. The ice table should start with .15M of HSO4- and .15M of H3O+.
- Sun Jan 10, 2021 2:56 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: What are the small elements over the equilibrium symbols?
- Replies: 5
- Views: 298
Re: What are the small elements over the equilibrium symbols?
Yes, I would also assume that the elements above are catalysts of the reaction. They likely affect the reactants in a way that allows the chemical reaction to occur while not being consumed by the reaction.
- Sun Jan 10, 2021 2:47 pm
- Forum: Non-Equilibrium Conditions & The Reaction Quotient
- Topic: Are there reactions that just do not/ can not ever reach equilibrium?
- Replies: 18
- Views: 1892
Re: Are there reactions that just do not/ can not ever reach equilibrium?
Every chemical reaction should have the potential to reach equilibrium given that it has the right conditions to reach the reaction's equilibrium constant. This could depend on things like time, since reactions need time to occur and reach that equilibrium constant. I think that there are reactions...
- Sun Jan 10, 2021 1:50 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: I in ICE Chart
- Replies: 11
- Views: 515
Re: I in ICE Chart
The question will likely tell you when the product is not zero. The examples provided above will also imply that the product isn't zero.
- Sun Jan 10, 2021 1:47 pm
- Forum: Ideal Gases
- Topic: inert gas and pressure changes
- Replies: 9
- Views: 411
Re: inert gas and pressure changes
It might be good to consider these inert gases like the solvents in the normal solution. Since the inert gases don't react to anything, when solving for the K constant, we put the inert gas both on the reaction and product part of the equation, thus causing them to cancel each other out.
- Sun Jan 10, 2021 1:23 pm
- Forum: Equilibrium Constants & Calculating Concentrations
- Topic: Calculating Equilibrium Concentrations
- Replies: 10
- Views: 442
Re: Calculating Equilibrium Concentrations
You should be careful with the liquids. While solvents aren't taken into account for the K value, liquids that are reacted together can be involved in the K concentration.
- Fri Dec 11, 2020 10:16 pm
- Forum: Calculating pH or pOH for Strong & Weak Acids & Bases
- Topic: Example Problem
- Replies: 2
- Views: 317
Re: Example Problem
You could also use -log (3.50 x 10-3 M) to find the pOH and subtract the result from 14 (because pH+pOH = 14). You should get 11.54 pH
- Fri Dec 11, 2020 10:11 pm
- Forum: Lewis Acids & Bases
- Topic: Aluminum Chloride naming in today's review
- Replies: 4
- Views: 253
Re: Aluminum Chloride naming in today's review
Since it is an ionic compound, we don't use the prefixes because we can find out the number by checking their respective charges. We use prefixes in covalent bonds because the number of atoms in the molecule is more ambiguous.
- Fri Dec 11, 2020 9:18 pm
- Forum: Calculating the pH of Salt Solutions
- Topic: Na and Cl's influence on pH
- Replies: 11
- Views: 893
Re: Na and Cl's influence on pH
Rather than not interacting, Na+ isn't strong enough to make the water molecules lose a hydrogen like Fe, Cr, Al, Cu, and Ni. A general rule to follow is that the cations in groups 1 and 2 aren't strong enough to affect the pH. For anions, the really strong anions create a more basic solution becaus...
- Fri Dec 11, 2020 9:03 pm
- Forum: Amphoteric Compounds
- Topic: memorizing amphoteric oxides
- Replies: 3
- Views: 701
Re: memorizing amphoteric oxides
Here is a picture of the amphoteric oxides for convenience.


- Fri Dec 11, 2020 8:26 pm
- Forum: Properties & Structures of Inorganic & Organic Acids
- Topic: Textbook 6D.11
- Replies: 3
- Views: 144
Re: Textbook 6D.11
How do we know that Al 3+ forms a complex with water and that it'll react with more water to form hydronium ions? In one of his classes (I think it's the 12/7 one), our professor went over the small cations that acted as lewis acids when alone in an aqueous solution. (this is why the problem is Ph<...
- Sun Dec 06, 2020 2:46 pm
- Forum: Bronsted Acids & Bases
- Topic: Strong acids
- Replies: 7
- Views: 494
Re: Strong acids
I actually think the atom with the larger radius is the "stronger" acid because the bonds are more easily broken (longer bonds = weaker bonds). This is why HF is considered a weaker acid than HI. If we look at the group 17 acids, HF<HCl<HBr<HI in terms of acidity. Remember that the weak/st...
- Sun Dec 06, 2020 2:11 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: Memorizing shapes based on general formula?
- Replies: 5
- Views: 505
Re: Memorizing shapes based on general formula?
My TA gave us this resource to memorize the vespr model. It was a very helpful resource and wanted to share.
http://chem.illinois.edu/chemdoodleweb/table.html
http://chem.illinois.edu/chemdoodleweb/table.html
- Sun Dec 06, 2020 2:00 pm
- Forum: Lewis Acids & Bases
- Topic: Equilibrium
- Replies: 3
- Views: 220
Re: Equilibrium
^ Adding onto this, the equation that he has in his lecture Ka = [A-][H+]/[AH] is just the concentrations of the products divided by the concentration of the reactants, but I am not 100% sure if we would have to know this/calculate the equilibrium of the weak acid so if someone could answer that'd ...
- Sun Dec 06, 2020 1:53 pm
- Forum: Lewis Acids & Bases
- Topic: Example Problem
- Replies: 5
- Views: 362
Re: Example Problem
For future reference, I think it is also good to memorize the specific charge for the ligands so you can easily find the charge (or oxidation) of the transition metal.
- Sun Dec 06, 2020 1:44 pm
- Forum: Calculating pH or pOH for Strong & Weak Acids & Bases
- Topic: pH and pOH
- Replies: 6
- Views: 439
Re: pH and pOH
The difference between pH and pOH is that they both measure different things. pH measures the concentration of H3O+ while pOH measures the concentration of OH-. They both add up to 14 so if they give you one of them, you can find the other one easily. However, we usually use pH so be careful not to ...
- Fri Nov 27, 2020 4:33 pm
- Forum: Sigma & Pi Bonds
- Topic: Carbon bonds
- Replies: 4
- Views: 275
Re: Carbon bonds
The carbon usually forms 4 bonds because it wants to fill up it's 4 orbitals (2sp3) to be stable. There are many reasons why carbon can be called "special" - it can form huge chains of carbon that can form macromolecules which are big enough for larger organism to use. - it is very stable ...
- Fri Nov 27, 2020 4:14 pm
- Forum: Sigma & Pi Bonds
- Topic: Delocalized Pi Bonds
- Replies: 4
- Views: 626
Re: Delocalized Pi Bonds
I think you can spot delocalized bonds by assessing their resonance structures. For example, in benzene since the combination of the resonance structures will result in single/double bond characteristics in all the bonds. With this, we can tell that there is one sigma bond between each of them and a...
- Fri Nov 27, 2020 3:52 pm
- Forum: Sigma & Pi Bonds
- Topic: Delocalized Pi Bond
- Replies: 5
- Views: 977
Re: Delocalized Pi Bond
Hello. I think this image does a good job describing how a delocalized pi bond is formed in benzene. It is because the resonance structures all combine to form a molecule with bonds with both double and single bond characteristics. During this process, the pi bonds forms a large e- cloud (which is w...
- Fri Nov 27, 2020 3:40 pm
- Forum: Sigma & Pi Bonds
- Topic: Main Difference between Sigma & Pi Bonds Q
- Replies: 4
- Views: 821
Re: Main Difference between Sigma & Pi Bonds Q
As Tamara said above, the major difference that you should look out for is that sigma bonds can rotate and the pi bonds are rigid. I also wanted to add a visual to help. https://cdn.shortpixel.ai/client/q_glossy,ret_img,w_596/https://vivadifferences.com/wp-content/uploads/2019/06/Sigma-Vs-Pi-Bond.png
- Fri Nov 27, 2020 3:33 pm
- Forum: Properties of Electrons
- Topic: Difference between electrons and light properties
- Replies: 5
- Views: 773
Re: Difference between electrons and light properties
Although they can be considered to have both particle and waves like motions, I think that you shouldn't see them as very similar. The negativity of the electron and the mass-less light greatly differentiate them from each other and have very different implications in chemical reactions.
- Sun Nov 22, 2020 12:50 pm
- Forum: Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding)
- Topic: Boiling Point/vapor pressure
- Replies: 5
- Views: 449
Re: Boiling Point/vapor pressure
I don't know if this will help but an easy way to think of vapor pressure is to think of it as the urge to become a vapor. So the higher the vapor pressure, the higher the urge to become a vapor. In terms of boiling point, the lower the vapor pressure, the higher the boiling temperature.
- Sun Nov 22, 2020 12:37 pm
- Forum: Shape, Structure, Coordination Number, Ligands
- Topic: Expanded Octets
- Replies: 9
- Views: 452
Re: Expanded Octets
Like those above, the atoms after period 3 can all have expanded octets. The periodic table is always expanding as we find more atoms, so there is likely an 8d that just hasn't been discovered yet. I think for this course we will only need to know that the atoms after neon can have octets because n=3.
- Sun Nov 22, 2020 12:24 pm
- Forum: Dipole Moments
- Topic: Dipole Moments Cancelling out
- Replies: 10
- Views: 1164
Re: Dipole Moments Cancelling out
I think looking at the forces in terms of vectors may help. Note that the example below works because the atoms surrounding the dipole moments are exactly the same, thus having the same forces acted upon it (just in a different direction). https://www.andrew.cmu.edu/course/09-105/GIFs/DIPOLE.08.GIF
- Sun Nov 22, 2020 12:16 pm
- Forum: Dipole Moments
- Topic: Dipole Moments on cis-dichloroethene
- Replies: 3
- Views: 369
Re: Dipole Moments on cis-dichloroethene
To add on, I think that looking at the general direction of the forces will be helpful in determining whether they are polar or not. The trans-dicholorethene are nonpolar because they point the opposite way while cisdichloroethene is polar because they point towards a general direction. https://www....
- Sat Nov 21, 2020 8:46 pm
- Forum: Determining Molecular Shape (VSEPR)
- Topic: VSEPR
- Replies: 4
- Views: 394
Re: VSEPR
I also don't think we need to know it but I think this website is quite informative for learning electrostatic potential maps. link: https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_(Physical_and_Theoretical_Chemistry)/Chemical_Bonding/Fun...
- Sat Nov 14, 2020 7:21 pm
- Forum: Bond Lengths & Energies
- Topic: Energy released
- Replies: 3
- Views: 205
Re: Energy released
In general intramolecular interactions will require more energy to break than intermolecular interaction since intramolecular bonds are usually a lot stronger.
- Sat Nov 14, 2020 6:59 pm
- Forum: Sigma & Pi Bonds
- Topic: Triple bond?
- Replies: 21
- Views: 941
Re: Triple bond?
Catherine Bubser 2C wrote:What is an example of a molecule that exhibits a delta bond?
I don't think we need to know this but apparently Re2Cl8 2- ion has a delta bond .
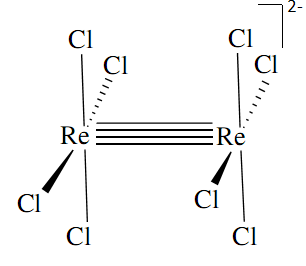
- Sat Nov 14, 2020 6:51 pm
- Forum: Ionic & Covalent Bonds
- Topic: Cation vs Anion
- Replies: 5
- Views: 632
Re: Cation vs Anion
Hello. Like Emily said, I think you should follow the trends of the periodic table. Think about how many electrons they need or don't need to become stable. As a general trend the atoms on the left side are usually cations while the atoms on the right side are anions. If you need additional help you...
- Sat Nov 14, 2020 6:41 pm
- Forum: Dipole Moments
- Topic: Interaction potential energy
- Replies: 2
- Views: 132
Re: Interaction potential energy
The negative sign represents that the energy is being released outside the system.
- Sat Nov 14, 2020 5:10 pm
- Forum: Resonance Structures
- Topic: Finding The Most Plausible Resonance Structure
- Replies: 3
- Views: 541
Re: Finding The Most Plausible Resonance Structure
To add on to what Edward said, the formal charge exaggerates the covalent character of the bond by assuming that all the bonds are equally shared while the oxidation number exaggerates the ionic character by assuming that the more electronegative gets the e-. So in the scope of this question, since ...
- Sat Nov 07, 2020 4:31 pm
- Forum: Electron Configurations for Multi-Electron Atoms
- Topic: Electron configurations of f state
- Replies: 4
- Views: 368
Re: Electron configurations of f state
If I remember correctly, in one of the lectures the professor said that we will be focusing on s, p, and d blocks so I don't think the f block is necessary. However, if you want to learn it just incase I recommend watching this video: https://youtu.be/KJLgnAUVALc
- Sat Nov 07, 2020 4:18 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: Inner Core
- Replies: 4
- Views: 270
Re: Inner Core
The inner core would be the electrons that are already filled in. I like to think of it as the electrons that aren't touched in a reaction so this will usually be the closest noble gas.
- Sat Nov 07, 2020 4:06 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: S vs P electrons
- Replies: 5
- Views: 418
Re: S vs P electrons
The s-electrons have lower energies because they are closer to the nucleus than the p-electrons. This protects the p-electrons from the nucleus's charge (aka shielding). You can probably get a better explanation here: https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textboo...
- Sat Nov 07, 2020 2:42 pm
- Forum: Electron Configurations for Multi-Electron Atoms
- Topic: Isoelectric atoms
- Replies: 9
- Views: 710
Re: Isoelectric atoms
To add on to those above me, you should focus on how many protons they have to determine their electronegativities and ionization.
- Sat Nov 07, 2020 2:28 pm
- Forum: Trends in The Periodic Table
- Topic: Easy Way to Remember Trends
- Replies: 5
- Views: 562
Re: Easy Way to Remember Trends
Hello,
I don't have an acronym to remember the trends but I found this website very helpful in doing so.
Website: https://chem.libretexts.org/Bookshelves ... _Chemistry)/Descriptive_Chemistry/Periodic_Trends_of_Elemental_Properties/Periodic_Trends
I don't have an acronym to remember the trends but I found this website very helpful in doing so.
Website: https://chem.libretexts.org/Bookshelves ... _Chemistry)/Descriptive_Chemistry/Periodic_Trends_of_Elemental_Properties/Periodic_Trends
- Sat Oct 31, 2020 9:33 pm
- Forum: Ionic & Covalent Bonds
- Topic: Non Metals
- Replies: 10
- Views: 811
Re: Non Metals
To add on to those above me, the high ionization energy of these materials are also the reason why they are bad conductors of electricity.
- Sat Oct 31, 2020 9:15 pm
- Forum: Octet Exceptions
- Topic: Octet Exceptions
- Replies: 10
- Views: 605
Re: Octet Exceptions
To add on to those above me, I think there is a correlation between the charge of the electrons and the nucleus. I don't think the charges of the nucleus of these atoms (+1 H, +2 H, +3 Li, +4 B) are strong enough to hold all 8 electrons together.
- Sat Oct 31, 2020 9:02 pm
- Forum: Lewis Structures
- Topic: Lewis Structures
- Replies: 7
- Views: 588
Re: Lewis Structures
You will need to find the atom with the lowest electromagnetic and then surround it based on how many valence electrons it needs. For more complex molecules, there are isomers (molecules with same number of molecules but different structures) so we may have to build our structures based on the infor...
- Sat Oct 31, 2020 8:53 pm
- Forum: Ionic & Covalent Bonds
- Topic: Homework due date
- Replies: 49
- Views: 2584
Re: Homework due date
You should finish it by 11:59pm on Sunday but I assume that you can still do the problems past the time since he didn't seem to set an "available from date" on the website itself.
- Sat Oct 31, 2020 8:40 pm
- Forum: Ionic & Covalent Bonds
- Topic: Ionic Vs. Covalent
- Replies: 7
- Views: 454
Re: Ionic Vs. Covalent
To add on, ionic bonds are held together by the charges of the whole atom while covalent bonds are held together by their shared electron.
- Sun Oct 25, 2020 6:43 pm
- Forum: Limiting Reactant Calculations
- Topic: M.7
- Replies: 6
- Views: 475
Re: M.7
The LR for this problem would be Mg because the weight of 3 Mg's are greater than the weight of B2O3. The book seems to also use Mg as the LR so you may have done your calculations wrong.
- Sun Oct 25, 2020 5:58 pm
- Forum: Wave Functions and s-, p-, d-, f- Orbitals
- Topic: Advice for studying
- Replies: 92
- Views: 8952
Re: Advice for studying
To add onto those ahead of me, if you are having problems with a specific topic, I recommend that you watch The Organic Chemistry Tutor on youtube. I find him really helpful because he goes over each problem step by step.
- Sun Oct 25, 2020 5:10 pm
- Forum: Heisenberg Indeterminacy (Uncertainty) Equation
- Topic: Rearranging uncertainty in velocity
- Replies: 5
- Views: 180
Re: Rearranging uncertainty in velocity
In order to solve for the delta V, we have to divide each side by M (Delta V= delta P/M). I think it will help if you think of the deltas (delta V and Delta P in this case)as a variable that cannot be split.
- Sun Oct 25, 2020 4:33 pm
- Forum: Properties of Light
- Topic: J/mol or J/photon
- Replies: 2
- Views: 1808
Re: J/mol or J/photon
The problems will typically be in J/photons unless they tell you to convert to moles. In this problem, instead of changing it to moles, you had to divide 36.14 J by the joules of the photon with the wavelength of 6.18×10−4 cm.
- Sun Oct 25, 2020 2:57 pm
- Forum: Properties of Light
- Topic: Midterm
- Replies: 42
- Views: 1791
Re: Midterm
So we're going to be in a zoom call with our whole discussion group? Doesn't that mean that we'll be able to see each other's work? Yes, I'm pretty sure we will be in a zoom call with our group, however I think it will be quite hard to cheat off of each other since the TA will be watching your scre...
- Sat Oct 17, 2020 1:59 pm
- Forum: Heisenberg Indeterminacy (Uncertainty) Equation
- Topic: Module Question 14
- Replies: 2
- Views: 96
Re: Module Question 14
Yes, you are correct. Although every object has a wave-like property, for classical objects like baseballs we often consider them as particle-like objects since the lambda is too small to be noticed or have substantial effects.
- Sat Oct 17, 2020 1:49 pm
- Forum: DeBroglie Equation
- Topic: Wavelike Properties
- Replies: 4
- Views: 145
Re: Wavelike Properties
To add to the post above, while all objects have wave-like properties, we consider classical objects like baseballs to have particle-like properties since the lambda is too small to consider.
- Sat Oct 17, 2020 1:36 pm
- Forum: Heisenberg Indeterminacy (Uncertainty) Equation
- Topic: IB27
- Replies: 2
- Views: 120
Re: IB27
Hi. It is 10m/s because you have to consider both the negative and positive uncertainty of the equation. For example, if a ball had a velocity of 7.00 ± 3.0 m/s. The uncertainty will be 6.
- Sat Oct 17, 2020 12:49 pm
- Forum: *Black Body Radiation
- Topic: Black Body Radiation
- Replies: 6
- Views: 599
Re: Black Body Radiation
The black body radiation is a item that can absorb all frequencies of light and is purely theoretical. Scientist still haven't found any material able to do so. But the reason why black gets hotter than the other colors is because it can absorb more light frequencies than them.
- Sat Oct 17, 2020 12:29 pm
- Forum: Student Social/Study Group
- Topic: How are you studying?
- Replies: 204
- Views: 24984
Re: How are you studying?
To add onto the posts above, I personally think the Audio-Visual Focus-Topics really straightened-out a lot of topics for me. If you are struggling with some of the topics there, you can re-watch and redo the post-assessment. I plan on redoing the post-assessment test and re-watching the video befor...
- Fri Oct 09, 2020 4:04 pm
- Forum: Limiting Reactant Calculations
- Topic: Determining Limiting Reactant
- Replies: 4
- Views: 111
Re: Determining Limiting Reactant
You can do either, but I think it is safer to find the moles of the product since it will always consider the moles ratios between the product and reactant unlike the first option.
- Fri Oct 09, 2020 3:46 pm
- Forum: Limiting Reactant Calculations
- Topic: Determining the Limiting Reactant
- Replies: 4
- Views: 209
Re: Determining the Limiting Reactant
Yes, you are correct C14H18N205 is the limiting reactant in this case since it has less moles, meaning the reaction runs less. If you are unsure, you can always check your answer by just converting it to the moles of the product. Hope this helps!
- Fri Oct 09, 2020 3:29 pm
- Forum: Molarity, Solutions, Dilutions
- Topic: Week 1 HW Q#10
- Replies: 5
- Views: 335
Re: Week 1 HW Q#10
No, you don't need to know what 2-butanone or 1-bromopropane are. You can determine their elemental composition by counting the provided Lewis structures. The Hydrogens aren't usually shown in Lewis structures so you will have to add hydrogens based on how many electrons each element needs for a sta...
- Thu Oct 08, 2020 9:24 pm
- Forum: Balancing Chemical Reactions
- Topic: Textbook Problem L35.
- Replies: 8
- Views: 386
Re: Textbook Problem L35.
After changing the 2.5 t into moles of NaBr, you need to convert the moles of NaBr to Fe3Br8 (1mole of Fe3Br8/8 moles of NaBr); then, convert moles of Fe3Br8 to moles of FeBr2 (3 moles of FeBr2/1 mole Fe3Br8); then, FeBr2 to Fe (1 mole of Fe/1 mole of FeBr2). As you're doing this, you should find th...
- Thu Oct 08, 2020 6:30 pm
- Forum: Balancing Chemical Reactions
- Topic: Question H 7d
- Replies: 3
- Views: 180
Question H 7d
The question: "the reaction of ammonia gas with oxygen gas at high temperatures in the presence of a copper metal catalyst produces the gases water and nitrogen dioxide." If the copper metal is a catalyst of the formula, is the copper metal included in the chemical equation? If not, how do...

