So for the psuedo equilibrium constant K dagger, you would use this equation
deltaG dagger=-RT ln(K dagger) with dagger referring to the transition state
On my quiz you solved for delta G in the previous question and he temperature was given, so you would just have to solve for K
Search found 26 matches
- Fri Mar 17, 2017 10:14 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Quiz 2 question
- Replies: 3
- Views: 743
- Sat Mar 11, 2017 3:32 pm
- Forum: *Electrophiles
- Topic: Nucleophile vs Electrophile
- Replies: 7
- Views: 2827
Re: Nucleophile vs Electrophile
Also, it is useful to note electronegativity in a molecule that can cause it to become an electrophile. For example, CO2 is an electrophile because of the dipole moments created by the negative charge of the O with the positive charge of the C, and the electronegativity of the Oxygens cause the Carb...
- Sat Mar 04, 2017 11:07 am
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Slow Reaction
- Replies: 11
- Views: 1981
Re: Slow Reaction
When the question asks you to find the rate equation, what do you do when your equation requires you to know the concentration of an intermediate species? Are you supposed to replace it by looking at the fast reaction and setting the forward rate equal to the reverse rate? Or is there another method...
- Sat Feb 25, 2017 12:32 pm
- Forum: Reaction Mechanisms, Reaction Profiles
- Topic: Slow Reaction
- Replies: 11
- Views: 1981
Re: Slow Reaction
I believe that the fast step is at equilibrium and the slow step is not at equilibrium. Also do you know if you use the pre-equilibrium approach if the fast step comes before the slow step or after the slow step?
- Sun Feb 19, 2017 2:13 pm
- Forum: First Order Reactions
- Topic: Concept of Half Life [ENDORSED]
- Replies: 3
- Views: 689
Re: Concept of Half Life [ENDORSED]
Is there a general trend that relates temperature and half-life? Does a higher temperature correlate to a lower half live or vice versa?
- Sat Feb 11, 2017 1:45 pm
- Forum: Balancing Redox Reactions
- Topic: highest possible charge [ENDORSED]
- Replies: 3
- Views: 702
Re: highest possible charge [ENDORSED]
How do the standard reaction potential reactions translate to oxidizing agents? Does a higher voltage mean that a reaction is a better oxidizing agent and vice versa?
- Sat Feb 04, 2017 12:45 pm
- Forum: Balancing Redox Reactions
- Topic: Salt bridge
- Replies: 3
- Views: 727
Re: Salt bridge
I have a follow up question, what allows for a current to flow: the electrons themselves or the electron movement from anode to cathode? Also, during a redox reaction, does the salt bridge allow for the reaction to occur multiple times so that a continuous charge is being generated?
- Sat Jan 28, 2017 2:29 pm
- Forum: Concepts & Calculations Using First Law of Thermodynamics
- Topic: Reversibly & Isothermally
- Replies: 3
- Views: 835
Re: Reversibly & Isothermally
In reversible reactions, reactants react with other reactants to form products, and the products react with other products to form more reactants. Essentially, it is a reaction cycle. In an irreversible reaction, reactants react with other reactants to form products, and the products do not form rea...
- Sat Jan 28, 2017 2:22 pm
- Forum: Entropy Changes Due to Changes in Volume and Temperature
- Topic: Assuming Constant Pressure?
- Replies: 2
- Views: 729
Re: Assuming Constant Pressure?
I would think that if the problem doesn't mention pressure change, you can assume that it is under conditions of constant pressure.
I may be wrong though
I may be wrong though
- Sat Jan 21, 2017 3:09 pm
- Forum: Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation)
- Topic: Energy of the system during expansion [ENDORSED]
- Replies: 2
- Views: 611
Re: Energy of the system during expansion [ENDORSED]
Is it true that energy can only exist in the system or surroundings? or is it somehow lost when the piston is moved and work is done?
- Fri Jan 13, 2017 8:22 am
- Forum: Thermodynamic Systems (Open, Closed, Isolated)
- Topic: Most Stable Form
- Replies: 4
- Views: 2509
Re: Most Stable Form
Would it be necessary to draw out the Lewis structure with a formal charge of 0 in order to determine a molecule's most stable form? Or is there another method that can be used?
- Fri Dec 02, 2016 5:47 pm
- Forum: Calculating the pH of Salt Solutions
- Topic: Henderson-Hasselbalch Equation: Solving for acid
- Replies: 2
- Views: 3242
Re: Henderson-Hasselbalch Equation: Solving for acid
The Henderson-Hasselbach Equation is pH=pKa + log ([A-]/[HA]) [HA] is the molar concentration of the weak acid, [A⁻] is the molar concentration of this acid's conjugate base, and pKa is −log10 Ka where Ka is the acid dissociation constant. So if we are given the pH, pKa, and the concentration of [HA...
- Fri Dec 02, 2016 5:32 pm
- Forum: Conjugate Acids & Bases
- Topic: Conjugate Acids/Bases Proton Acceptors/Donors
- Replies: 1
- Views: 515
Re: Conjugate Acids/Bases Proton Acceptors/Donors
I assume its the Hydrogen furthest away from the "center of the molecule" because it can easily be transferred whereas the H in the H3 isn't easily transferred, but I may be mistaken.
- Tue Nov 22, 2016 1:49 pm
- Forum: Bronsted Acids & Bases
- Topic: J Section Question 1
- Replies: 2
- Views: 886
Re: J Section Question 1
I guess you can use this as a strategy:
A Bronsted acid donates protons, A Lewis acid accepts electrons
A Bronsted base accepts protons, A Lewis base donates electrons
Also, you can visualize the potential reaction and see if protons are donated or accepted.
Hope this helped!
A Bronsted acid donates protons, A Lewis acid accepts electrons
A Bronsted base accepts protons, A Lewis base donates electrons
Also, you can visualize the potential reaction and see if protons are donated or accepted.
Hope this helped!
- Fri Nov 18, 2016 11:56 am
- Forum: Applying Le Chatelier's Principle to Changes in Chemical & Physical Conditions
- Topic: Neglecting solids and liquids
- Replies: 1
- Views: 2894
Re: Neglecting solids and liquids
You ignore the concentration of solids and liquids because they have a constant concentration. Liquids and solids cannot be compressed because the molecules are very close together; without compression there is no change is concentration (like there is when gasses are compressed.) For these reasons,...
- Mon Nov 14, 2016 12:08 am
- Forum: *Molecular Orbital Theory (Bond Order, Diamagnetism, Paramagnetism)
- Topic: Quiz #3 [ENDORSED]
- Replies: 7
- Views: 1580
Re: Quiz #3 [ENDORSED]
Yes. I'm pretty sure that MO theory will be on the exam. We covered it in class and it wasn't on the midterm so its safe to say that its going to be on there
- Sun Nov 06, 2016 5:49 pm
- Forum: Hybridization
- Topic: Hybridizaton Textbook equation
- Replies: 2
- Views: 601
Re: Hybridizaton Textbook equation
I assume that the equations must mean that the first hybridization for the atom has one s orbital and 3 p orbitals (hence px, py, pz). Therefore, you can conclude that the hybridization is sp3. You can also determine the hybridization of an atom by finding how many regions of electron density there ...
- Sun Oct 30, 2016 4:46 pm
- Forum: Quantum Numbers and The H-Atom
- Topic: Midterm 2007 review question [ENDORSED]
- Replies: 1
- Views: 616
Re: Midterm 2007 review question [ENDORSED]
When n=5, l can equal 0,1,2,3,4 which corresponds to the s,p,d,f, and (theoretically) g subshells. 5s has one orbital (maximum of 2 electrons), 5p has three orbitals (maximum of 6 electrons), 5d has five orbitals (maximum of 10 electrons), 5f has 7 orbitals (maximum of 14 electrons), and 5g would th...
- Sun Oct 23, 2016 7:43 pm
- Forum: Electron Configurations for Multi-Electron Atoms
- Topic: Unpaired Electrons
- Replies: 1
- Views: 385
Re: Unpaired Electrons
Whether or not an element has an unpaired electron depends on the total number of electrons in the molecule. So you would first need to find the total number of valence electrons and then you would draw a lewis structure (try to follow the octet rule). You would then figure out how many electrons wo...
- Mon Oct 10, 2016 6:14 pm
- Forum: Balancing Chemical Reactions
- Topic: Naming Chemical Formula and Balancing Chemical Equations [ENDORSED]
- Replies: 5
- Views: 2033
Re: Naming Chemical Formula and Balancing Chemical Equations [ENDORSED]
When writing out the chemical equation, should we include the state that the chemical is in? so would it be N2(g) + O2(g) ==> 2NO(g)
- Sun Oct 09, 2016 9:03 pm
- Forum: Significant Figures
- Topic: All students read this sig fig post [ENDORSED]
- Replies: 170
- Views: 35071
Re: All students read this sig fig post [ENDORSED]
For testing purposes, is there a difference between writing 1.8 x 10^-2 and 0.018? I know they're synonymous, but I've seen both used in the textbook and workbook so I was wondering if there is a preference for either when it comes to giving a final answer. I would use 1.8 x 10^-2 because it is in ...
- Sun Oct 02, 2016 11:40 pm
- Forum: Bohr Frequency Condition, H-Atom , Atomic Spectroscopy
- Topic: Atomic Spectra AV post assessment #31 [ENDORSED]
- Replies: 8
- Views: 1055
Re: Atomic Spectra AV post assessment #31 [ENDORSED]
Oh ok thank you very much for the clarification
- Sun Oct 02, 2016 11:20 pm
- Forum: Bohr Frequency Condition, H-Atom , Atomic Spectroscopy
- Topic: Atomic Spectra AV post assessment #31 [ENDORSED]
- Replies: 8
- Views: 1055
Re: Atomic Spectra AV post assessment #31 [ENDORSED]
Wouldnt you get a different answer if you use a different constant?
- Sat Oct 01, 2016 4:18 pm
- Forum: Bohr Frequency Condition, H-Atom , Atomic Spectroscopy
- Topic: Atomic Spectra AV post assessment #31 [ENDORSED]
- Replies: 8
- Views: 1055
Re: Atomic Spectra AV post assessment #31 [ENDORSED]
Why do you use 1.097 x 10^7 instead of 3.29 x 10^15 for R?
- Fri Sep 30, 2016 2:18 pm
- Forum: Bohr Frequency Condition, H-Atom , Atomic Spectroscopy
- Topic: Atomic Spectra AV post assessment #24
- Replies: 3
- Views: 628
Re: Atomic Spectra AV post assessment #24
I noticed in your work that you equated frequency (v) to 1/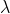 but actually, frequency (v) is equal to
but actually, frequency (v) is equal to  /
/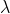
- Fri Sep 30, 2016 1:43 pm
- Forum: Bohr Frequency Condition, H-Atom , Atomic Spectroscopy
- Topic: Atomic Spectroscopy Experiment Setup
- Replies: 1
- Views: 297
Atomic Spectroscopy Experiment Setup
In an atomic spectroscopy experiment, what exactly is detected? Is it all light or just emitted light?

