polar vs nonpolar molecules
Moderators: Chem_Mod, Chem_Admin
-
KiaraTenorio_14B
- Posts: 102
- Joined: Fri Sep 24, 2021 5:38 am
polar vs nonpolar molecules
In the lecture notes the example used for polar and non-polar molecules is dichloroethene, what is the difference between cis-dichloroethene and trans-dichloroethene?
-
azurexrose
- Posts: 103
- Joined: Fri Sep 24, 2021 5:29 am
- Been upvoted: 2 times
Re: polar vs nonpolar molecules
Cis-dichloroethane has the two chlorine atoms on the same side of their respective carbon atoms, kind of like
Cl Cl
C C
H H
Therefore, there's a dipole moment on the Cl side because it's a lot more electronegative than carbon and they aren't orientated in a way that they cancel each other out. Therefore cis dichloroethane is polar.
Trans-dichloroethane have the two chlorine atoms opposite to each other, like
Cl H
C C
H Cl
The dipole moments between C and Cl cancel out, so there is no net dipole moment and trans dichloroethane is nonpolar.
Cl Cl
C C
H H
Therefore, there's a dipole moment on the Cl side because it's a lot more electronegative than carbon and they aren't orientated in a way that they cancel each other out. Therefore cis dichloroethane is polar.
Trans-dichloroethane have the two chlorine atoms opposite to each other, like
Cl H
C C
H Cl
The dipole moments between C and Cl cancel out, so there is no net dipole moment and trans dichloroethane is nonpolar.
-
William_Hora_2H
- Posts: 104
- Joined: Fri Sep 24, 2021 7:27 am
Re: polar vs nonpolar molecules
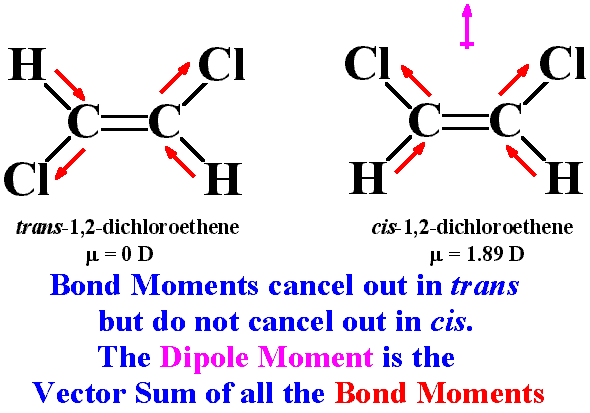
In the cis orientation, the dipoles DO NOT cancel out (see red arrows). In the trans orientation, the dipoles DO cancel out. For dipoles (red arrows) to cancel out, two red arrows must be pointing directly away from one another (see trans orientation, specifically, C-Cl bonds); notice this is NOT the case for the C-Cl bonds in the cis orientation.
-
Rena Wu 3E
- Posts: 101
- Joined: Fri Sep 24, 2021 6:46 am
Re: polar vs nonpolar molecules
Hi Kiara, cis-dichloroethene would have the two Cl atoms next to each other in the molecular structure, and the two H atoms next to each other as well. Meanwhile the trans-dichloroethene would have the the two Cl atoms diagonal from one another, and the same for the two H atoms. Since there is no net dipole moment in the trans-dichloroethene structure, we can classify this isomer as a nonpolar structure. On the other hand, there is a net dipole moment in the cis-dichloroethene structure, meaning the isomer is polar. Hope this helps!
Who is online
Users browsing this forum: No registered users and 6 guests

