Hi I'm wondering how you can find the q of a reaction when there is no temperature change? I thought of this while looking at the q = nC*deltaT equation.
When you use this equation, and input 0 for delta T, q can only equal 0.
Thank you.
q value when no change in Temp
Moderators: Chem_Mod, Chem_Admin
-
Chinyere Okeke 2J
- Posts: 102
- Joined: Wed Sep 30, 2020 10:05 pm
- Been upvoted: 1 time
Re: q value when no change in Temp
I think that if there is no change in temperature then that equation will give you a q of 0 since that equation tells us the heat due to temperature. If you want to get a q value not equal to 0 when there is no temp change you might need to use a different equation.
-
Peter DePaul 1E
- Posts: 101
- Joined: Wed Sep 30, 2020 9:55 pm
- Been upvoted: 1 time
Re: q value when no change in Temp
Q is the value of the net heat transfer. If 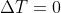 , then there's no heat transfer. Therefore q = 0
, then there's no heat transfer. Therefore q = 0
-
Anthony_Sandoval_1D
- Posts: 101
- Joined: Wed Feb 19, 2020 12:15 am
-
Aanya Tanti 3C
- Posts: 52
- Joined: Wed Nov 25, 2020 12:19 am
-
Charmaine Ng 2D
- Posts: 108
- Joined: Wed Sep 30, 2020 9:37 pm
Re: q value when no change in Temp
Q would be zero, since no change in temperature also means no heat is transferred
-
Lauren Strickland 1B
- Posts: 97
- Joined: Wed Sep 30, 2020 9:47 pm
-
Queena Chu 3E
- Posts: 96
- Joined: Wed Sep 30, 2020 10:09 pm
Return to “Thermodynamic Definitions (isochoric/isometric, isothermal, isobaric)”
Who is online
Users browsing this forum: No registered users and 4 guests

