Gibbs Free Energy Equilibrium Equation
Moderators: Chem_Mod, Chem_Admin
-
Reynaldo Fernandez 3L
- Posts: 41
- Joined: Mon Jan 09, 2023 10:15 am
Gibbs Free Energy Equilibrium Equation
How does the equation Delta G = -RTln(k) let us solve for the equilibrium constant. How does the change in free energy at all relate to equilibrium? And how are there 2 ways to solve for the equilibrium constant with the ice table and now with this equation? It just doesn’t conceptually make full sense to me.
-
Nick Kraemer 2J
- Posts: 34
- Joined: Mon Jan 09, 2023 9:27 am
Re: Gibbs Free Energy Equilibrium Equation
Hi! Using the equation delta G = -RTlnK you can first rearrange it to be lnK = delta G/-RT and then raise both sides by e to undo the ln giving you the final equation of K = e^(delta G/-RT). This equilibrium constant is the same as the one find by the ice tables, just there's two different ways to find it.
Re: Gibbs Free Energy Equilibrium Equation
Gibbs free energy is the potential energy. So when Gibbs free is negative, then the reaction has the potential to lose energy meaning it is spontaneous.
The equation for standard Gibbs free energy is = -RTlnK. K being greater than 1(Products>reactants at Eq) makes lnK positive, making G negative(according to the equation). K being greater than one means reaction is product favored so forward reaction is favored(spontaneous reaction). K being less than 1 does the exact opposite, and makes it not spontaneous.
= -RTlnK. K being greater than 1(Products>reactants at Eq) makes lnK positive, making G negative(according to the equation). K being greater than one means reaction is product favored so forward reaction is favored(spontaneous reaction). K being less than 1 does the exact opposite, and makes it not spontaneous.
The equation for standard Gibbs free energy is
Re: Gibbs Free Energy Equilibrium Equation
The relationship between ΔG and equilibrium is fundamental to understanding chemical reactions. At equilibrium, the forward and reverse reactions are occurring at the same rate, so there is no net change in the concentrations of the reactants and products. ΔG is related to the difference in the Gibbs free energy between the reactants and products, and at equilibrium, this difference is zero. Therefore, ΔG is also zero at equilibrium.
The ICE table method is typically used when you are given the concentrations of reactants and products, along with change in concentrations.
the formula is typically used when you are given the
formula is typically used when you are given the 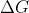 to find K.
to find K.
The ICE table method is typically used when you are given the concentrations of reactants and products, along with change in concentrations.
the
Return to “Gibbs Free Energy Concepts and Calculations”
Who is online
Users browsing this forum: No registered users and 2 guests

