Meaning of K
Moderators: Chem_Mod, Chem_Admin
Meaning of K
I just wanted to check, when using K in the thermodynamics equations, does it always stand for the equilibrium constant? Thank you!
-
Madilyn Schindler 3E
- Posts: 109
- Joined: Wed Sep 30, 2020 9:52 pm
- Been upvoted: 1 time
Re: Meaning of K
The only things I can think of that K could stand for is potassium, Kelvin, or the equilibrium constant haha. However, a lowercase k could stand for the Boltzmann constant. Hope that helps!
-
Tiao Tan 3C
- Posts: 109
- Joined: Wed Sep 30, 2020 9:59 pm
Re: Meaning of K
I agree with Madilyn! So far, I've only seen K representing the equilibrium constant in thermodynamics questions. The lowercase k representing the Boltzmann's constant will appear in the future.
-
Alan Huang 1E
- Posts: 101
- Joined: Wed Sep 30, 2020 9:37 pm
Re: Meaning of K
In most problems I think the K would be for Kelvin. However, if the problems ask for an equilibrium constant than K could be that as well. It depends on context so it could be case-by-case.
-
Jasmine Ho 3I
- Posts: 102
- Joined: Wed Sep 30, 2020 9:41 pm
Re: Meaning of K
Yes, in all of the equations Professor Lavelle has given us with a capital K, the K is the equilibrium constant.
-
Brandon Pham 1H
- Posts: 82
- Joined: Wed Sep 30, 2020 9:59 pm
- Been upvoted: 1 time
-
Uyen Trinh 3C
- Posts: 56
- Joined: Thu Nov 14, 2019 12:17 am
Re: Meaning of K
It depends on what the question is asking for but in most cases it is the equilibrium constant.
-
Muskaan Abdul-Sattar
- Posts: 101
- Joined: Wed Nov 13, 2019 12:19 am
Re: Meaning of K
K when used as a unit is likely referring to Kelvin. But, in problems regarding equilibrium constants, K is the equilibrium constant.
-
Brian Bui 3H
- Posts: 101
- Joined: Wed Sep 30, 2020 9:42 pm
Re: Meaning of K
If K is next to a number (ex. 298 K), then K = Kelvin
If K equals something (ex. K = 1.0 x 10-8), then K = equilibrium constant
If k is lowercase, then it's the Boltzmann's constant
Hope this helps!
If K equals something (ex. K = 1.0 x 10-8), then K = equilibrium constant
If k is lowercase, then it's the Boltzmann's constant
Hope this helps!
-
Annika Tamaki 1E
- Posts: 102
- Joined: Wed Sep 30, 2020 10:03 pm
- Been upvoted: 1 time
-
Xinying Wang_3C
- Posts: 105
- Joined: Wed Sep 30, 2020 9:39 pm
-
Nicoli Peiris 1B
- Posts: 110
- Joined: Wed Sep 30, 2020 10:02 pm
Re: Meaning of K
Sometimes it is a unit meaning kelvin but otherwise for our purposes it is the equilibrium constant.
-
MCalcagnie_ 1D
- Posts: 86
- Joined: Wed Sep 30, 2020 10:08 pm
Re: Meaning of K
I think you're referring to the recent homework problems and examples that we have been given, and in those, yes K is representing the equilibrium constant.
-
Vanessa Perez
- Posts: 118
- Joined: Wed Sep 30, 2020 9:56 pm
Re: Meaning of K
Yes! I was getting a little confused on this too! But K is equilibrium constant. Also look at for Q too!
-
Samantha Lee 1A
- Posts: 108
- Joined: Wed Sep 30, 2020 10:05 pm
- Been upvoted: 1 time
Re: Meaning of K
If you are referencing K in terms of the equation, ) , then yes, K is the equilibrium constant. K alone can mean lots of things. If it is attached to a temperature, it stands for Kelvin. If it is a variable in a
, then yes, K is the equilibrium constant. K alone can mean lots of things. If it is attached to a temperature, it stands for Kelvin. If it is a variable in a 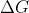 equation, there is a good chance that it is referencing the equilibrium constant. Another thing to hint at K being the equilibrium constant is if Q (is also apart of the equation). I mean Q for the non equilibrium constant, ie. Q>K, etc, and not q, meaning heat.
equation, there is a good chance that it is referencing the equilibrium constant. Another thing to hint at K being the equilibrium constant is if Q (is also apart of the equation). I mean Q for the non equilibrium constant, ie. Q>K, etc, and not q, meaning heat.
-
Akriti Ratti 1H
- Posts: 100
- Joined: Wed Sep 30, 2020 9:36 pm
-
Kimiya Aframian IB
- Posts: 131
- Joined: Wed Sep 30, 2020 9:34 pm
Re: Meaning of K
EmilyC_3D wrote:I just wanted to check, when using K in the thermodynamics equations, does it always stand for the equilibrium constant? Thank you!
Hi! I think that K must be put into context for thermodynamics because it can be the unit of temperature K (Kelvin), k (also written as kb) for Boltzmann's constant, or K which is the equilibrium constant. Hope this helps!
-
Gustavo_Chavez_1K
- Posts: 101
- Joined: Wed Sep 30, 2020 9:46 pm
Re: Meaning of K
Yeah for thermodynamics K can either be used as an abbreviation for the units Kelvin or as the equilibrium constant!
-
Ava Nickman
- Posts: 101
- Joined: Wed Sep 30, 2020 9:54 pm
Re: Meaning of K
Yup! equilibrium constant or I guess Kelvin... but the indication should be clear in the question
-
Colin Squire 3B
- Posts: 100
- Joined: Wed Sep 30, 2020 9:35 pm
Re: Meaning of K
In the context of thermochemistry and thermodynamics, I have only seem K used as the equilibrium constant. The only other option I can think of is Kelvin which is also K.
-
RyanKopeikin_2I
- Posts: 81
- Joined: Fri Aug 30, 2019 12:17 am
Re: Meaning of K
I would assume K stands for the equilibrium constant, because k (or kB) is Boltzmann's Constant, and if it is temperature in Kelvin the "K" will be accompanied by a number, like in 300K.
-
Carly_Lipschitz_3H
- Posts: 104
- Joined: Wed Sep 30, 2020 9:56 pm
Re: Meaning of K
K most usually means the equilibrium constant. As I'm sure you know K can also stand for Kelvins. It will definitely be clear whether the K given in a question is an equilibrium constant or if it is the value of temperature.
-
Leyla Anwar 3B
- Posts: 101
- Joined: Wed Sep 30, 2020 10:03 pm
Re: Meaning of K
If K is in the units it is Kelvin and if K is a variable you are solving for, such as within the deltaG equation, then it is the equilibrium constant!
-
Keon Amirazodi 3H
- Posts: 107
- Joined: Wed Sep 30, 2020 9:59 pm
-
Kayla Booker 1F
- Posts: 99
- Joined: Wed Sep 30, 2020 9:43 pm
-
Olivia Smith 2E
- Posts: 101
- Joined: Wed Sep 30, 2020 9:43 pm
Re: Meaning of K
It will stand for the equilibrium constant if in an equation. If used after a temperature it is kelvin.
-
Arnav Saud 2C
- Posts: 127
- Joined: Wed Sep 30, 2020 9:51 pm
Re: Meaning of K
I think the best way to think about it is the way it's situated.
If K is used as a unit, then it's most likely kelvin. If K is equaling some kind of value or is involved in an equilibrium problem, then it's probably referring to the equilibrium constant. A lowercase k refers to the Boltzmann Constant.
If K is used as a unit, then it's most likely kelvin. If K is equaling some kind of value or is involved in an equilibrium problem, then it's probably referring to the equilibrium constant. A lowercase k refers to the Boltzmann Constant.
-
Mari Williams 1K
- Posts: 104
- Joined: Wed Sep 30, 2020 9:53 pm
Re: Meaning of K
Temperature is represented in equations by T, so an uppercase K in the equation refers to the equilibrium constant :)
-
Lauren Mungo 1K
- Posts: 100
- Joined: Wed Sep 30, 2020 10:08 pm
- Been upvoted: 1 time
Re: Meaning of K
Yes in all equations K is the equilibrium constant. The only other thing you could confuse it with is K for kelvin so just make sure to read the problem carefully !
-
Kyla Villarama 1E
- Posts: 101
- Joined: Wed Nov 20, 2019 12:18 am
-
Alejandro Gonzalez 2G
- Posts: 82
- Joined: Wed Sep 30, 2020 10:04 pm
Re: Meaning of K
Depending on the context of the problem, K can either refer to the equilibrium constant or the temperature in Kelvin.
-
Joshua Chung 2D
- Posts: 108
- Joined: Wed Sep 30, 2020 9:32 pm
-
Kelly Ha 1K
- Posts: 109
- Joined: Wed Sep 30, 2020 9:52 pm
- Been upvoted: 1 time
Re: Meaning of K
K is the equilibrium constant. However, if it is mentioned in the context of units, K most likely refers to temperature in Kelvin.
-
Can Yilgor 2D
- Posts: 104
- Joined: Wed Sep 30, 2020 10:09 pm
Re: Meaning of K
From a thermodynamics standpoint, I believe K generally refers to the equilibrium constant and the absolute temperature in equations.
-
Crystal Pan 2G
- Posts: 118
- Joined: Wed Sep 30, 2020 10:04 pm
- Been upvoted: 2 times
Re: Meaning of K
Uppercase K means the equilibrium constant, or it could mean Kelvins. lowercase k would be the boltzmann constant.
-
Queena Chu 3E
- Posts: 96
- Joined: Wed Sep 30, 2020 10:09 pm
-
Mehreen 3I
- Posts: 99
- Joined: Wed Sep 30, 2020 9:36 pm
Re: Meaning of K
K always refers to the equilibrium constant in that context. K is the constant of a certain reaction when it is in equilibrium, while Q is the quotient of activities of products and reactants at any stage of a reaction.
-
Ranen_Chang_2G
- Posts: 44
- Joined: Wed Nov 25, 2020 12:21 am
-
Shreyank Kadadi 3K
- Posts: 50
- Joined: Thu Dec 17, 2020 12:19 am
-
Emily_Stenzler_2H
- Posts: 197
- Joined: Wed Sep 30, 2020 10:02 pm
Re: Meaning of K
Yes! Especially in this class, if we are finding K this is most always referring to the equilibrium constant. If we are meant to find temperature, we would likely be told to calculate temperature in K, rather than to calculate K.
-
Taylor Newville 1C
- Posts: 51
- Joined: Wed Nov 18, 2020 12:21 am
Re: Meaning of K
The problem will tell you what is meant by K and its value will make it obvious too. Usually it is either the equilibrium constant or temperature in kelvins.
-
Kaleb Tuliau 3E
- Posts: 68
- Joined: Tue Nov 05, 2019 12:18 am
-
Alison Perkins 2B
- Posts: 105
- Joined: Wed Sep 30, 2020 9:57 pm
- Been upvoted: 1 time
Re: Meaning of K
K can either stand for Kelvin or the equilibrium constant, which should be fairly easy to distinguish. The Boltzman constant is the other k I can think of appearing, but it's lowercase and often has a B subscript. Be careful with kB for the Boltzman constant and KB for an equilibrium constant considering basicity.
-
Lizbeth Garcia 1F
- Posts: 102
- Joined: Wed Sep 30, 2020 9:39 pm
Re: Meaning of K
You can use it for Kelvin or the equilibrium constant, but the K for Kelvin is usually shown regular in the books while the K for the equilibrium constant is usually italicized.
-
Evonne Hsu 1J
- Posts: 108
- Joined: Wed Sep 30, 2020 9:35 pm
Re: Meaning of K
Generally, K would stand for Kelvin. It can also stand for the equilibrium constant K, if stated in the question.
-
Kathy_Li_1H
- Posts: 100
- Joined: Wed Sep 30, 2020 9:31 pm
-
Sable Summerfield
- Posts: 52
- Joined: Tue Nov 17, 2020 12:18 am
Re: Meaning of exp
What exactly does exp mean? I saw it in a problem and was not sure what to do with it.
-
Caelin Brenninkmeijer 1G
- Posts: 100
- Joined: Wed Sep 30, 2020 9:50 pm
Re: Meaning of K
I think it's safe to assume that K will usually signify the equilibrium constant when not addressing temperature (as in Kelvin).
-
Sreeram Kurada 3H
- Posts: 103
- Joined: Wed Sep 30, 2020 10:00 pm
-
Susanna Givan 2B
- Posts: 144
- Joined: Thu Feb 27, 2020 12:16 am
Re: Meaning of K
It is either the equilibrium constant or temperature in Kelvin. But usually, it is the equilibrium constant.
-
LovepreetSran_3H
- Posts: 103
- Joined: Wed Sep 30, 2020 9:33 pm
-
Susan Chamling 1F
- Posts: 101
- Joined: Wed Sep 30, 2020 9:42 pm
Return to “Gibbs Free Energy Concepts and Calculations”
Who is online
Users browsing this forum: No registered users and 7 guests

