How deltaG affects product/reactant formation
Moderators: Chem_Mod, Chem_Admin
-
Isabelle Hales 1J
- Posts: 100
- Joined: Wed Sep 30, 2020 9:41 pm
- Been upvoted: 1 time
How deltaG affects product/reactant formation
In last Friday’s lecture, Lavelle mentioned that when K > 1, deltaG under standard conditions is negative. He said that there are more products that reactants which will cause product formation to be favored. I am a bit confused by this. I thought that if there are more products than reactants, the formation of reactants would be favored. Can someone help me understand this?
-
AnjikaFriedman-Jha2D
- Posts: 101
- Joined: Wed Sep 30, 2020 9:55 pm
Re: How deltaG affects product/reactant formation
He might be saying that when there are more products than reactants the delta G of the reverse reaction is negative and thus that is what is spontaneous
-
Sami Siddiqui 1J
- Posts: 165
- Joined: Wed Sep 30, 2020 9:58 pm
- Been upvoted: 6 times
Re: How deltaG affects product/reactant formation
In the long run, to have a negative delta G value means that K (the equilibrium constant) will be greater than 1, meaning that the equilibrium concentrations will favor the products over the reactants as the reaction approaches equilibrium. This is given by the fact that, on a conceptual level, K is essentially [P]/[R] (while factoring in all the stoichiometric coefficients as exponents for each substance). To have a positive delta G value means that K will be less than 1, meaning that vice versa will be true. In the context of chemical equilibrium, if we took a system that was already at equilibrium and then added more product to it, then the reactants would be favored according to Le Chatelier's principle.
-
Aydin Karatas 1F
- Posts: 141
- Joined: Wed Sep 30, 2020 9:37 pm
Re: How deltaG affects product/reactant formation
If we look at the equation for 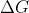 at standard condition, we get
at standard condition, we get ) . For
. For 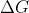 to be negative, thus meaning the reaction favors the products, we need
to be negative, thus meaning the reaction favors the products, we need ) to produce a positive number. When K > 1,
to produce a positive number. When K > 1, ) < 0. When 0 < K < 1,
< 0. When 0 < K < 1, ) > 0. Since R is a positive constant and T is given in Kelvin, we can see that the favorability of a reaction is depended on
> 0. Since R is a positive constant and T is given in Kelvin, we can see that the favorability of a reaction is depended on ) in this expression.
in this expression.
-
Victor Qiu 1C
- Posts: 106
- Joined: Wed Sep 30, 2020 9:59 pm
- Been upvoted: 2 times
Re: How deltaG affects product/reactant formation
It is not true that if there are more products than reactants, the formation of reactants would be favored. You can just think of an example of dissociation of a strong acid.
HCl(aq) + H2O(l) → H3O+(aq) + Cl-(aq)
The products are favored even when there are more products than reactants.
HCl(aq) + H2O(l) → H3O+(aq) + Cl-(aq)
The products are favored even when there are more products than reactants.
-
Grace_Remphrey_2J
- Posts: 100
- Joined: Wed Sep 30, 2020 9:54 pm
Re: How deltaG affects product/reactant formation
I was also confused about this and these explanations were super helpful. Thank you!!
-
SavannahScriven_1F
- Posts: 107
- Joined: Wed Sep 30, 2020 9:37 pm
Re: How deltaG affects product/reactant formation
When K is > 1, the equilibrium lies to the right (i.e. at equilibrium, there is a higher concentration of products than reactants). Delta G would be negative because the forward reaction is favored (product form is more stable than reactant form, so rxn is spontaneous).
Return to “Van't Hoff Equation”
Who is online
Users browsing this forum: No registered users and 2 guests

