In this problem, we are given one of the following options for four different scenarios: frequency, wavelength, and photon energy, and have to determine which option matches to which scenario, each of which is a different type of radiation.
Is it viable to just use the 2 formulas of c=λv and e=hv to calculate the data needed to complete the table and determine the sources?
Also, in c=λv, the correct unit for λ is meters, correct?
Problem 1A.9 (7th edition)
Moderators: Chem_Mod, Chem_Admin
-
Minsub Lee 3E
- Posts: 32
- Joined: Fri Sep 28, 2018 12:25 am
Re: Problem 1A.9 (7th edition)
Yes, use c=λv and e=hv to find the source. The correct unit for wavelength (λ) are meters.
-
Henri_de_Guzman_3L
- Posts: 88
- Joined: Fri Sep 28, 2018 12:25 am
Re: Problem 1A.9 (7th edition)
Isn't it dumb how we need E=hv to solve this problem, yet that isn't given until 1B? Am I tripping?
Re: Problem 1A.9 (7th edition)
Yea I thought that was strange too because 1A doesn't address Energy of the photon much apart from Table 1A.1
-
Diana Bibireata 1B
- Posts: 60
- Joined: Fri Sep 28, 2018 12:23 am
Re: Problem 1A.9 (7th edition)
In the equation c=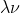 you do use meters (because the unit for c is meters per second)
you do use meters (because the unit for c is meters per second)
But it is important to note that wavelengths in the visible spectrum are usually given in nanometers (which is 10-9m) so just also know this conversion.
But it is important to note that wavelengths in the visible spectrum are usually given in nanometers (which is 10-9m) so just also know this conversion.
-
Summer de Vera 2C
- Posts: 65
- Joined: Fri Sep 28, 2018 12:16 am
Re: Problem 1A.9 (7th edition)
Yes, you can use both equations. You may also want to combine them for some, though I don't know if it's needed.
Return to “Properties of Light”
Who is online
Users browsing this forum: No registered users and 8 guests

