The answer in the solutions manual says:
Cd(s)|Cd(OH)2(s)|KOH(aq) || Ni(OH)3(s)|Ni(OH)2(s)|Ni(s)
First of all how did they determine this order? I saw in an earlier post Chem.mod said that cell diagrams are typically written to where the reaction proceeds from the outside towards the middle, that doesn't seem to be the case here. Does it really matter what order the anode and cathode are written in?
Also, why is the OH- included in the anode but not the cathode for the cell diagram? OH- appears in both of the half reactions but in the cell diagram they take it out of the cathode but leave in it the anode.
Also the addition of the Ni(s) as the conducting electrode for the cathode makes sense, but how would you know to do this? Typically we add Pt(s) as the conducting electrode, when do you know to use something different? Or does it really matter?
Thanks
14.15 c [ENDORSED]
Moderators: Chem_Mod, Chem_Admin
-
Chem_Mod
- Posts: 23858
- Joined: Thu Aug 04, 2011 1:53 pm
- Has upvoted: 1253 times
Re: 14.15 c
Typically anodes are on the left and cathodes are on the right in a cell diagram. If you balance the complete reaction, OH- is net in the reaction with Cd so it is written on that side. It is best to use an electrode that is a species in your cell. Since you have 2 Ni compounds and Ni is a conductive metal, it is easier to use Ni to avoid possible side reactions.
-
Alisha Clark
- Posts: 20
- Joined: Fri Sep 25, 2015 3:00 am
Re: 14.15 c
Where does the potassium come from in the cell diagram? If it is from the salt bridge, why can't we just write 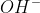 instead of KOH?
instead of KOH?
-
Chem_Mod
- Posts: 23858
- Joined: Thu Aug 04, 2011 1:53 pm
- Has upvoted: 1253 times
Re: 14.15 c [ENDORSED]
The K does come from the salt bridge. You need to include the K to show that the negative OH is bound to something since all of the other compounds are in the solid state.
Return to “Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams”
Who is online
Users browsing this forum: No registered users and 8 guests

