Electromotive force (emf)
Moderators: Chem_Mod, Chem_Admin
-
Matthew Tsai 2H
- Posts: 101
- Joined: Wed Sep 18, 2019 12:20 am
Electromotive force (emf)
Can someone explain what emf is exactly and if we need to know anything about it other than that it represents max work?
Re: Electromotive force (emf)
the book mentions that EMF is the same as the cell potential of a cell.
-
Sanjana Borle 2K
- Posts: 111
- Joined: Fri Aug 09, 2019 12:15 am
Re: Electromotive force (emf)
I think all we need to know is that EMF is the same thing as the cell potential, or the variable E, so if a problem refers to emf it is just referring to E.
Re: Electromotive force (emf)
And also, maximum nonexpansion work is the same as Gibbs free energy, so emf directly relates to 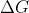
-
Jocelyn Thorp 1A
- Posts: 103
- Joined: Wed Sep 18, 2019 12:20 am
-
Miriam Villarreal 1J
- Posts: 105
- Joined: Sat Aug 17, 2019 12:16 am
Re: Electromotive force (emf)
Emf is given a positive value if the cell reaction is spontaneous. This implies that the electrode on the left forces electrons into the external circuit and the electrode on the right withdraws them
On the other hand, Emf is given a negative value if the cell reaction is nonspontaneous. This implies that if the half-cell on the right side of the shorthand cell notation is releasing electrons, making the right-hand terminal of the voltmeter negative
On the other hand, Emf is given a negative value if the cell reaction is nonspontaneous. This implies that if the half-cell on the right side of the shorthand cell notation is releasing electrons, making the right-hand terminal of the voltmeter negative
Re: Electromotive force (emf)
if its the same as cell potential I would assume we don't need to know much more about it
-
Julia Holsinger_1A
- Posts: 50
- Joined: Tue Feb 26, 2019 12:16 am
Re: Electromotive force (emf)
005384106 wrote:Why do we measure the voltage difference of two cells?
The difference in potential voltage is what gives rise to electric current
Return to “Work, Gibbs Free Energy, Cell (Redox) Potentials”
Who is online
Users browsing this forum: No registered users and 10 guests

