15.17
Moderators: Chem_Mod, Chem_Admin
-
Mia Navarro 1D
- Posts: 52
- Joined: Fri Sep 29, 2017 7:04 am
-
Caroline LaPlaca
- Posts: 30
- Joined: Sat Jul 22, 2017 3:01 am
-
Johann Park 2B
- Posts: 51
- Joined: Thu Jul 27, 2017 3:01 am
- Been upvoted: 1 time
Re: 15.17
With respect to [B], the reaction is second-order. You use the different values for the rates and concentrations to solve for the exponents (x, y, z) on the concentrations.
-
OliviaShearin2E
- Posts: 37
- Joined: Fri Sep 29, 2017 7:05 am
Re: 15.17
Johann is right but to be clearer, first order in B means that the rate is proportional to 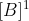 , second order in B means that the rate is proportional to
, second order in B means that the rate is proportional to 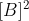 . and third order in B means that the rate is proportional to
. and third order in B means that the rate is proportional to 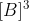 .
.
-
Sandhya Rajkumar 1C
- Posts: 50
- Joined: Fri Jun 23, 2017 11:40 am
Re: 15.17
The reaction is second order with respect to B because when the amount of B is doubled (increased by a factor of 2), the rate increases by a factor of 4, so instead of it being a 1:1 ratio, which would make it first order, it's a 1:2 ratio, which makes it second order.
Who is online
Users browsing this forum: No registered users and 15 guests

