7th edition 7B.3
Posted: Sun Mar 10, 2019 3:58 pm
by Alyssa Bryan 3F
Determine the rate constant for each of the following first order reactions, in each case expressed for the rate loss of A:
how do you determine the rate constant for part c) 2A
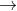
B + C, given that [Ao] = 0.153 mol/L and that after 115 s the concentration of B rises to 0.34 mol/L
Re: 7th edition 7B.3
Posted: Sun Mar 10, 2019 4:13 pm
by Andrew Bennecke
This type of problem where we are given information about both A and B, A being a reactant and B being a product occurs several times in the 7B exercises of the 7th edition of the textbook. I also don't know which equations to use that involve information about both A and B, I assume it is setting 2 rate laws equal to each other, but i am unable to figure which ones to do so with.
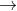 B + C, given that [Ao] = 0.153 mol/L and that after 115 s the concentration of B rises to 0.34 mol/L
B + C, given that [Ao] = 0.153 mol/L and that after 115 s the concentration of B rises to 0.34 mol/L