Collision Cross Section
Moderators: Chem_Mod, Chem_Admin
-
Sarah Sharma 2J
- Posts: 33
- Joined: Fri Sep 29, 2017 7:05 am
Collision Cross Section
Can someone please explain the importance of knowing the collision cross section? Why do we need to know the area at which a molecule presents itself as a target. Also, how do we know if a molecule IS a target? When a collision happens aren't they both considered targets?
-
Michelle Chernyak 1J
- Posts: 33
- Joined: Fri Sep 29, 2017 7:06 am
Re: Collision Cross Section
if a molecule is not at the proper orientation when a collision happens, then the reaction will not occur
-
Julian Krzysiak 2K
- Posts: 49
- Joined: Fri Sep 29, 2017 7:07 am
Re: Collision Cross Section
The collision cross section is important because it makes up a component of A, the pre-exponential factor. 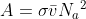
The pre-exponential factor tells us the rate at which molecules collide with each other. The collision cross section plays into this because it gives us the area of the molecule available to be a target for other molecules. In other words, it tells us how "big" the molecules are- the bigger the molecule is, the more likely it will collide with another molecule, which would increase the collision frequency and A. Therefore, this, along with the mean relative speed of the atoms and Avagadro's number combine to tell us the rate at which the molecules/atoms collide.
Whenever we are talking about a specific molecule, we assume that it is the target, as everything else is relative to it. Technically, every single molecule in a certain container is a potential target for each other, but saying an atom/molecule is a target is just an easier way for you to conceptualize what is happening on the individual molecular scale.
The pre-exponential factor tells us the rate at which molecules collide with each other. The collision cross section plays into this because it gives us the area of the molecule available to be a target for other molecules. In other words, it tells us how "big" the molecules are- the bigger the molecule is, the more likely it will collide with another molecule, which would increase the collision frequency and A. Therefore, this, along with the mean relative speed of the atoms and Avagadro's number combine to tell us the rate at which the molecules/atoms collide.
Whenever we are talking about a specific molecule, we assume that it is the target, as everything else is relative to it. Technically, every single molecule in a certain container is a potential target for each other, but saying an atom/molecule is a target is just an easier way for you to conceptualize what is happening on the individual molecular scale.
Return to “Reaction Mechanisms, Reaction Profiles”
Who is online
Users browsing this forum: No registered users and 4 guests

