Uncertainty
Moderators: Chem_Mod, Chem_Admin
-
Kyle Thorin
- Posts: 50
- Joined: Sat Aug 24, 2019 12:16 am
-
Eugene Chung 3F
- Posts: 142
- Joined: Wed Nov 15, 2017 3:03 am
-
Julie Park 1G
- Posts: 100
- Joined: Thu Jul 25, 2019 12:15 am
Re: Uncertainty
https://www.youtube.com/watch?v=BNYz5EKXVeI
This video does a pretty good job at explaining the concepts behind the Heisenberg Uncertainty Principle, and it also provides examples using the uncertainty equation (including an example that translates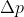 from the uncertainty equation to
from the uncertainty equation to 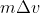 )
)
This video does a pretty good job at explaining the concepts behind the Heisenberg Uncertainty Principle, and it also provides examples using the uncertainty equation (including an example that translates
-
SVajragiri_1C
- Posts: 115
- Joined: Thu Jul 11, 2019 12:16 am
-
Madeline Ogden 3B
- Posts: 112
- Joined: Wed Sep 30, 2020 10:03 pm
- Been upvoted: 1 time
Re: Uncertainty
Yes, uncertainty and indeterminacy are used interchangeably in this equation. The reason he often says "indeterminacy" instead of "uncertainty" is because uncertainty makes it sound like we are unsure of whether or not the equation is accurate even though it is. Indeterminacy is a better word because it instead connotes the fact that we know the object is there and that the equation is accurate but are simply unsure/indeterminate of its exact momentum and/or position.
Return to “Heisenberg Indeterminacy (Uncertainty) Equation”
Who is online
Users browsing this forum: No registered users and 6 guests

