Ionic and Covalent Character
Moderators: Chem_Mod, Chem_Admin
-
Tiffany Wu 1K
- Posts: 19
- Joined: Wed Sep 21, 2016 2:56 pm
Ionic and Covalent Character
What is the difference between ionic and covalent character and how do we determine strength of each of them? Do we use the periodic table?
-
Vivian Wang 3J
- Posts: 29
- Joined: Wed Sep 21, 2016 2:57 pm
- Been upvoted: 1 time
Re: Ionic and Covalent Character
Ionic and covalent character describe the nature of the bond between atoms.
For example, a molecule with a higher ionic character than covalent character means that the atoms have more of a give-and-take relationship for the electrons that bond them.
If the bond were described to have higher covalent character, the atoms are sharing the electrons.
Covalent bonds do have some ionic character, though it is very low. As seen in this chart, covalently bonded molecules (HCl, HBr, and HI) have lower ionic character than ionically bonded molecules.
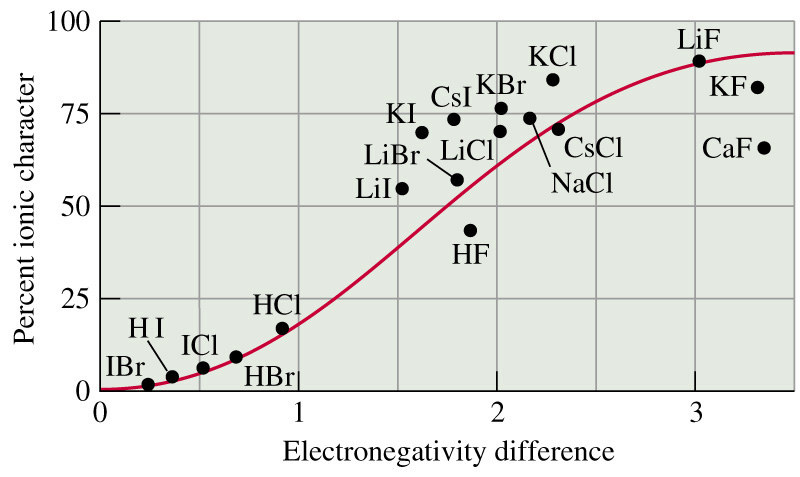
Likewise, ionic bonds can have covalent character, though it will be low.
The ionic/covalent character can be determined using the periodic table. The greater the difference in electronegativity between the atoms, the greater the ionic character of the bond.

For example, a molecule with a higher ionic character than covalent character means that the atoms have more of a give-and-take relationship for the electrons that bond them.
If the bond were described to have higher covalent character, the atoms are sharing the electrons.
Covalent bonds do have some ionic character, though it is very low. As seen in this chart, covalently bonded molecules (HCl, HBr, and HI) have lower ionic character than ionically bonded molecules.
Likewise, ionic bonds can have covalent character, though it will be low.
The ionic/covalent character can be determined using the periodic table. The greater the difference in electronegativity between the atoms, the greater the ionic character of the bond.
-
Mirna_Velasco_1L
- Posts: 19
- Joined: Wed Sep 21, 2016 3:00 pm
Re: Ionic and Covalent Character
Vivian Wang, You said that the ionic/covalent character can be determined using the periodic table. And that the greater the difference in electronegativity between the atoms, the greater the ionic character of the bond, so does this means that if there is not much difference in electronegativity between the atoms, than it should have covalent character instead. Or how do we decide if the atoms have covalent character. I am still confused on how to distinguish between ionic and covalent character? I will appreciate if someone explains this to me?
-
Vivian Wang 3J
- Posts: 29
- Joined: Wed Sep 21, 2016 2:57 pm
- Been upvoted: 1 time
Re: Ionic and Covalent Character
"Ionic and covalent character represent points along a continuum."
Based on this statement, we can assume that as ionic character of a bond increases, covalent character of the same bond decreases.
And, as covalent character of a bond increases, ionic character of that bond decreases.
Based on this statement, we can assume that as ionic character of a bond increases, covalent character of the same bond decreases.
And, as covalent character of a bond increases, ionic character of that bond decreases.
-
Mizuno_Mikaela_1D
- Posts: 29
- Joined: Wed Sep 21, 2016 3:00 pm
Re: Ionic and Covalent Character
How do we determine covalent character of an ionic bond? I know in the course reader it says the more polarizable the anion, the more covalent character the ionic bond has due to a larger distortion, however I still do not quite understand what that means or how that makes the bond have more covalent character.
-
NinaSheridan
- Posts: 30
- Joined: Wed Sep 21, 2016 2:58 pm
Re: Ionic and Covalent Character
So based on this logic, any bond with small ionic character (such as elements of the same group, or the same element bonding with another atom of the same element) would have the highest covalent character, and elements far apart from one another on the periodic table would have low covalent character.
Return to “Ionic & Covalent Bonds”
Who is online
Users browsing this forum: No registered users and 2 guests

