Delocalization
Moderators: Chem_Mod, Chem_Admin
-
Carissa Young 1K
- Posts: 65
- Joined: Fri Sep 28, 2018 12:17 am
Re: Delocalization
Delocalization means that the electron pair is distributed over several pairs of atoms and can't be identified with just one pair of atoms.
-
danicatran4
- Posts: 35
- Joined: Fri Sep 28, 2018 12:17 am
Re: Delocalization
Does delocalization have anything to do with hybridization? Like does in happen in conjunction with only pi bonds?
-
Dana Wilks 3I
- Posts: 30
- Joined: Fri Sep 28, 2018 12:23 am
Re: Delocalization
I believe delocalization has to do with resonance. When we draw a lewis structure for a compound with resonance we indicate single and double bonds. In actuality the molecule doesn't have clear single or double bonds, rather it has something in between (which makes it more stable).
-
Ivan Tadeja 1G
- Posts: 81
- Joined: Fri Sep 28, 2018 12:28 am
Re: Delocalization
In resonance, a lone pair can sometimes be in different locations with respect to the configuration. For example, drawing the lewis structure for 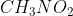 would result in resonance between two different structures in which the lone electron pair can be placed on either oxygen atom.
would result in resonance between two different structures in which the lone electron pair can be placed on either oxygen atom.
-
Ethan McCarthy 1F
- Posts: 31
- Joined: Wed Feb 27, 2019 12:17 am
-
KaitlynBali_4B
- Posts: 51
- Joined: Fri Aug 02, 2019 12:16 am
Re: Delocalization
Resonance is term used to describe a molecule that has a blend of structures that results from delocalized electrons. I am drawing the conclusion that if electrons in a molecule are delocalized, the molecule is immediately described to have resonance.
Return to “Resonance Structures”
Who is online
Users browsing this forum: No registered users and 3 guests

