However, for the example
Melting Point
Moderators: Chem_Mod, Chem_Admin
-
Juliana Misirian
- Posts: 80
- Joined: Fri Sep 24, 2021 5:30 am
Melting Point
Hello! I'm having a hard time determining melting points. Example: Which has the higher melting point, NaF or KCl. For this question, we need to look at their size because their charge is the same. So, by looking at their charge, I see that NaF is smaller than KCl, which makes the melting point of NaF higher. For this example, as size increases, the melting point decreases.
However, for the example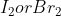 , the molecule with the higher melting point is
, the molecule with the higher melting point is 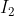 , even though I is larger than Br. So, in this case, as size increases, the melting point increases. How do you know when to use which rule?
, even though I is larger than Br. So, in this case, as size increases, the melting point increases. How do you know when to use which rule?
However, for the example
-
Romi Moore 3k
- Posts: 100
- Joined: Fri Sep 24, 2021 5:21 am
Re: Melting Point
I think it is a combination of a few different factors. As the strength of the bond increasing, the melting point will increase as well. That is most likely the case in the example you mentioned. Br2 might contain bonds other that single bonds. If it contains any double or triple bonds, the strength of the bond increases along with its melting point.
-
Maleeha Zaman 1K
- Posts: 49
- Joined: Fri Sep 24, 2021 6:46 am
Re: Melting Point
Hi Juliana, size is actually not the determining factor for the reason why NaF has a higher melting point than KCl. Recall that physical properties of a molecule are dependent on their intermolecular forces. Since flourine is highly electronegative, the dipole-dipole forces that NaF experiences are stronger than that of KCl. You would only use size as a comparison if the two molecules you are comparing are both nonpolar. For instance, I2 and Br2 are nonpolar molecules that only experience London dispersion forces. Since iodine has more electrons than bromine, it has a more easily distortable e- cloud and thus experiences stronger London dispersion forces, suggesting I2 has a higher melting point than Br2.
-
azurexrose
- Posts: 103
- Joined: Fri Sep 24, 2021 5:29 am
- Been upvoted: 2 times
Re: Melting Point
The boiling point of ionic compounds like NaF or KCl is determined by the strength of attraction between the ions, which can be found by looking at the difference in electronegativities. Ionic compounds aren't really grouped in separate molecules like covalently bonded molecules are, so there aren't really things like IMFs. The boiling point just depends on the electrostatic force between the ions. Since NaF have a higher difference in electronegativity than KCl, the NaF bond is more ionic in character and the ions have stronger connections to each other, so the boiling point is lower.
However, for I2 and Br2, which are atoms, the molecules have IMFs with each other. The only IMFs between I2 and Br2 molecules are LDFs, and since I2 is larger than Br2, it's more polarizable and therefore has stronger LDFs. Therefore, I2 has a larger boiling point than Br2.
However, for I2 and Br2, which are atoms, the molecules have IMFs with each other. The only IMFs between I2 and Br2 molecules are LDFs, and since I2 is larger than Br2, it's more polarizable and therefore has stronger LDFs. Therefore, I2 has a larger boiling point than Br2.
-
RJ Lopez 2l
- Posts: 104
- Joined: Fri Sep 24, 2021 5:21 am
Re: Melting Point
The dipole dipole forces between NaF are greater due to F being the most electronegative element. Dipole Dipole forces are stronger than the London Dispersion Forces in KCl. It important to look at the difference in the electronegativity as well as size of an atom when determine melting point
-
Abhinav Amanaganti 3K
- Posts: 54
- Joined: Fri Sep 24, 2021 6:20 am
Re: Melting Point
Intermolecular forces and bond strength are the main factors influencing melting point. Thus, you should look for dipole moments, hydrogen bonding, and other forms of intermolecular forces to determine the relative attractive forces in a substance, while also observing the bonds: double bonds would be stronger than single bonds b/c increasing number of bonds decreases bond length and this increases bond strength.
-
Rachel Bartley 2B
- Posts: 104
- Joined: Fri Sep 24, 2021 6:06 am
Re: Melting Point
When comparing melting/boiling points between two compounds, it is important to look at both the size of the compounds as well as the difference in electronegativities depending on the type of compounds you are comparing. So when looking at ionic compounds, such as NaF and KCl, the difference in electronegativities will tell you that KCl has a greater difference in electronegativity than NaF and therefore has a higher boiling point because its ion-ion bond is stronger. But when looking at covalent compounds, such as I2 and Br2 with only LDFs, you would use the size of the atoms in the compounds to determine which has a greater melting/boiling point. In this case, diatomic iodine is larger than diatomic bromine, so I2 has stronger intermolecular forces than Br2, making it require more energy to pull apart and melt.
Re: Melting Point
Melting point is determined by the strength of the bonds within the molecule. This is why the bigger the atom, the smaller the melting point, as the bonds are weaker. However, size is not the only factor that contributes to bond strength, you also need to look at the type of bonding occurring as well as the electronegativity. Hope that helps!
-
Rebekah Jung 1C
- Posts: 101
- Joined: Fri Sep 24, 2021 6:40 am
- Been upvoted: 1 time
Re: Melting Point
Melting point is heavily dependent on intermolecular forces. Looking at the types of bonds and their respective strengths will be helpful in determining which molecule will boil at higher temperatures.
Who is online
Users browsing this forum: No registered users and 4 guests

