bar conversion
Moderators: Chem_Mod, Chem_Admin
-
nshahwan 1L
- Posts: 100
- Joined: Fri Aug 30, 2019 12:18 am
bar conversion
Could someone more clearly explain how to change the units to bar? Example 5I.1 show it but it is not very clear and I am confused
-
Isha_Maniyar_Dis2E
- Posts: 110
- Joined: Thu Jul 11, 2019 12:16 am
Re: bar conversion
The conversion of bar to atm will be given in the formula handout we are given in each test. It is mainly a difference in values of R. If you are given values in bar, use the value for R involving bar, and then convert to atm using the formula sheet.
-
Justin Sarquiz 2F
- Posts: 106
- Joined: Fri Aug 30, 2019 12:15 am
Re: bar conversion
The formula sheet should say how to convert from atm to bar or bar to torr, so you should not worry too much about that.
-
Julie Park 1G
- Posts: 100
- Joined: Thu Jul 25, 2019 12:15 am
Re: bar conversion
In example 5I.1, they want you convert the partial pressures PH2 and PCl2 from mPa (which is given) to bar.
Partial pressure of H2 = 4.2 mPa and Cl2 = 8.3 mPa
It's also given that 1 bar = 10^5 Pa
However, we are given partial pressures in mPa. Knowing that there are 1000 mPa in 1 Pa, you can convert 4.2 mPa and 8.3 mPa to 4.2x10^-3 Pa and 8.3x10^-3 Pa.
Now that you know the Pa, you can convert the units to bar.
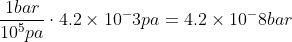
Partial pressure of H2 = 4.2 mPa and Cl2 = 8.3 mPa
It's also given that 1 bar = 10^5 Pa
However, we are given partial pressures in mPa. Knowing that there are 1000 mPa in 1 Pa, you can convert 4.2 mPa and 8.3 mPa to 4.2x10^-3 Pa and 8.3x10^-3 Pa.
Now that you know the Pa, you can convert the units to bar.
-
nshahwan 1L
- Posts: 100
- Joined: Fri Aug 30, 2019 12:18 am
-
Manav Govil 1B
- Posts: 104
- Joined: Sat Sep 07, 2019 12:19 am
Re: bar conversion
Also, if you are given the concentration and need to convert into bar, torr, etc., you would use the PV = nRT equation with the corresponding R.
Return to “Equilibrium Constants & Calculating Concentrations”
Who is online
Users browsing this forum: No registered users and 7 guests

