ice
Moderators: Chem_Mod, Chem_Admin
-
jonathan chi 1J
- Posts: 105
- Joined: Fri Sep 24, 2021 6:02 am
ice
How does ICE strategy work when given intial concentrations and one equilibrium concentration? Does this help you get the equilibrium constant?
-
Ella Henrickson 3I
- Posts: 101
- Joined: Fri Sep 24, 2021 6:24 am
Re: ice
Hello! From what I understand you can rearrange the equilibrium constant equation in order to find whatever value is missing. For this example, if you don't know K but you have the initial conditions and the equilibrium concentration of at least one species, you can find the change in concentration from that and apply it to the rest of the species that you know the initial concentrations for to find all of the equilibrium concentrations. Once you have that information you can solve for the equilibrium constant (K).
Re: ice
Hi! For the species where you know both the initial and equilibrium concentration, you can calculate the change in concentration. This value is the value of 'X' we would usually use here if we didn't know any of the equilibrium concentrations. You can use this value to calculate the change in concentration for the other reactants and products using their respective stoichiometric coefficients and, in turn, the equilibrium concentrations. Hope this helps!
-
Jeffrey Yang 3I
- Posts: 50
- Joined: Mon Jan 03, 2022 9:37 pm
Re: ice
To add on to Ella's response, most problems will give you initial and final concentrations of all reactants and products, to find K; or you will be given K, and be asked to find a final concentration.
-
William_Hora_2H
- Posts: 104
- Joined: Fri Sep 24, 2021 7:27 am
Re: ice
I've included an example in which your scenario may be applicable.
A+ B ⇌ C, intial concen of A is 4 mol/L and B is 4 mol/L. equil concen of C is 3 mol/L. Find K.
Information gathered (solve for x):
A: 4-x = 4-x (x would cancel)
B: 4-x = 4-x (x would cancel)
C: 0+x=3 (x=3)
Since x=3 plug into 4-x to get 4-3=1 thus equilibrium cocnen of A and B are 1.
Plug values into equil expression to find K (particularly, Kc).
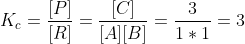
A+ B ⇌ C, intial concen of A is 4 mol/L and B is 4 mol/L. equil concen of C is 3 mol/L. Find K.
Information gathered (solve for x):
A: 4-x = 4-x (x would cancel)
B: 4-x = 4-x (x would cancel)
C: 0+x=3 (x=3)
Since x=3 plug into 4-x to get 4-3=1 thus equilibrium cocnen of A and B are 1.
Plug values into equil expression to find K (particularly, Kc).
-
Riya Sawhney 1C
- Posts: 104
- Joined: Fri Sep 24, 2021 5:07 am
Re: ice
If we are given the initial and the equilibrium concentrations of a species in a reaction, we know the difference is the change, C in the ICE table. Depending on value of x we choose based on moles, this change is some multiple of x. We can use this x to find the equilibrium concentrations of the other species based off of the value of K.
-
Abu Zhang 2D
- Posts: 37
- Joined: Mon Jan 03, 2022 8:42 pm
Re: ice
Hi! Just reading from your posts, I have another question with applying ICE strategy to problems. Sometimes, when we plug our results into the K quotient, to facilitate calculation, we can omit a slight change X so that we can solve the equation. However, when can we omit a value that is small enough? If it is decided by the value of Kc, how small does Kc have to be such that we can omit the x that is being reduced?
-
Ashley Presnell 1C
- Posts: 112
- Joined: Fri Sep 24, 2021 6:06 am
Re: ice
If we use the ICE box to figure out the change in concentrations based on the balanced chemical equation, we can usually isolate a single variable x that is causing the change from the initial to equilibrium concentrations. Using the calculated change x from the initial and known equilibrium concentration, we can plug that into the change in the other reagents to find the remaining equilibrium concentrations. Once we know all the equilibrium concentrations, we can plug it into the Kc equation and solve for the equilibrium constant by plugging the values at equilibrium in.
-
Maggie Clark
- Posts: 100
- Joined: Fri Sep 24, 2021 7:34 am
Re: ice
Yes it does work in that instance, you should be able to calculate the constant from deduction from the given information
-
Aaron Kwan 3B
- Posts: 101
- Joined: Fri Sep 24, 2021 6:07 am
-
Ethan Mai 1D
- Posts: 105
- Joined: Fri Sep 24, 2021 6:03 am
Re: ice
You can definitely use ICE boxes when given both initial concentrations and an equilibrium concentration! If anything, it makes the problem easier because you can use the equilibrium concentration to derive the change in concentration for a species and find the other final equal concentrations using stoichiometric relationships.
Re: ice
For the initial concentration, if you are only given one for a side with one molecule then you can assume that the other molecules in the ICE table have an initial concentration for 0. As for the other values, you need to substitute x in and then plug them into the equation to solve for x. this will allow you to get the concentrations of all molecules.
Hope this helps!
Hope this helps!
Return to “Non-Equilibrium Conditions & The Reaction Quotient”
Who is online
Users browsing this forum: No registered users and 2 guests

