Q<K?
Moderators: Chem_Mod, Chem_Admin
-
Emily Engelkemier 1E
- Posts: 105
- Joined: Fri Sep 24, 2021 5:10 am
Re: Q<K?
If Q<K, the reaction will proceed in the forward direction and produce more products. The concentration of reactants is relatively greater than the concentration of products compared to their equilibrium concentrations.
-
KatieFrancisco1F
- Posts: 105
- Joined: Fri Sep 24, 2021 5:43 am
Re: Q<K?
Likewise, If Q>K then the concentrations of products is greater than reactants with regards to the reaction's equilibrium constant. This means the reverse reaction would occur in order to reach equilibrium
-
William_Hora_2H
- Posts: 104
- Joined: Fri Sep 24, 2021 7:27 am
Re: Q<K?
The following helps me envision a Q/K comparison:
Since Q=[P]/[R] and Q<K
If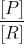 <K and K is a constant (doesn't change), the only way such that
<K and K is a constant (doesn't change), the only way such that  could be less than K is if denominator (R) was large. It helped to plug in some easy numbers (ex. 1) so that I could get a really firm grasp of the concept.
could be less than K is if denominator (R) was large. It helped to plug in some easy numbers (ex. 1) so that I could get a really firm grasp of the concept.
Since Q=[P]/[R] and Q<K
If
-
daniellediem1k
- Posts: 109
- Joined: Fri Sep 24, 2021 6:00 am
-
Michael Vigman 2D
- Posts: 111
- Joined: Fri Sep 24, 2021 5:00 am
- Been upvoted: 2 times
Re: Q<K?
Recall that Q is the ratio (in M or partial P) of reactants and products at any time during a given reaction. Meanwhile, K is the ratio when the reactants and products are at equilibrium.
When comparing Q to K, I tend to think of the comparison when comparing K to itself. If K > 1E3, then the reaction strongly lies to the right and product formation is favored. If K < 1E-3, then reaction strongly lies to the left and reactant formation is favored.
The same holds true for comparing Q to K:
If Q < K at any time during the reaction then [R] > [P] and the forward reaction (shift to the right) is favored and more product is formed until equilibrium is achieved again.
If Q > K at any time during the reaction then [R] < [P] and the reverse reaction (shift to the left) is favored and more reactant is formed until equilibrium is achieved again.
On a side note, I liked how William set up the comparison.
When comparing Q to K, I tend to think of the comparison when comparing K to itself. If K > 1E3, then the reaction strongly lies to the right and product formation is favored. If K < 1E-3, then reaction strongly lies to the left and reactant formation is favored.
The same holds true for comparing Q to K:
If Q < K at any time during the reaction then [R] > [P] and the forward reaction (shift to the right) is favored and more product is formed until equilibrium is achieved again.
If Q > K at any time during the reaction then [R] < [P] and the reverse reaction (shift to the left) is favored and more reactant is formed until equilibrium is achieved again.
On a side note, I liked how William set up the comparison.
-
Terrence Chi
- Posts: 99
- Joined: Fri Sep 24, 2021 6:06 am
Re: Q<K?
If Q<K at some time during the reaction, then the concentration of reactant is greater than the concentration of products therefore the forward reaction is favored. Hope this helps!
-
Elena Chan 2L
- Posts: 100
- Joined: Fri Sep 24, 2021 5:25 am
Re: Q<K?
If O<K then this means that the reaction quotient, Q, is less than the value of the equilibrium constant, K, meaning that [R] is greater than [P] thus they want to favor the forward reaction in order to bring the R/P ratio back to equilibrium and reach the equilibrium constant once again. Hope this helps.
-
Madison Chan 3B
- Posts: 50
- Joined: Mon Jan 03, 2022 9:17 pm
- Been upvoted: 1 time
Re: Q<K?
When Q < K, the system favors the forward reaction. This means that there are more reactants than products so the system moves in a forward fashion to try and reach equilibrium.
-
Sophie Cresitello 1B
- Posts: 50
- Joined: Mon Jan 03, 2022 10:31 am
Re: Q<K?
If Q<K, then this means the concentration of the reactants are greater than the concentrations of the products ( [R] > [P] ) and the forward reaction is favored.
Conversely, if Q>K, then [P] > [R] and the reverse reaction is favored.
Conversely, if Q>K, then [P] > [R] and the reverse reaction is favored.
-
Preethika Praveen 2G
- Posts: 100
- Joined: Fri Sep 24, 2021 6:25 am
Re: Q<K?
When Q<K, there is an excess of reactants, so the value of Q would be less than the equilibrium constant. Therefore, in order for the reaction to go to equilibrium, we would need a forward reaction that would increase the amount of product and allow for the reaction to reach the equilibrium constant.
-
taline krumian 1L
- Posts: 50
- Joined: Mon Jan 03, 2022 10:58 am
Re: Q<K?
If Q<K, [R]>[P], meaning the forward reaction is favored. The concentration of reactants is greater than the concentration of the products, so the reactants move towards the products in order to reach equilibrium.
-
Gianna Sciole 2F
- Posts: 101
- Joined: Fri Sep 24, 2021 7:06 am
Re: Q<K?
If Q<K, the reaction will proceed in the forward direction because there are more reactants than products in the equation than there are when the reaction is at equilibrium. If Q>K, the reverse reaction occurs because there are more products than reactants in the reaction than are when it is at equilibrium. And lastly, if Q=K, the reaction is at equilibrium!
-
Mia Orr 3B
- Posts: 103
- Joined: Fri Sep 24, 2021 7:11 am
-
Jessica Sun 2I
- Posts: 102
- Joined: Fri Sep 24, 2021 7:18 am
Re: Q<K?
When Q<K, the reaction will make more products in order to bring the system to equilibrium. In other words, it will favor the forward reaction.
-
Nithya Narapa Reddy
- Posts: 100
- Joined: Fri Sep 24, 2021 6:47 am
Re: Q<K?
If Q is less than K then the reaction proceeds in the forward reaction and the products are favored.
-
Rebecca Preusch 2C
- Posts: 101
- Joined: Fri Sep 24, 2021 6:26 am
Re: Q<K?
If Q<K the reaction will shift right/proceed forward. If Q>K the reaction shifts left/the reverse reaction is favored.
-
Renga Rengappa 1D
- Posts: 102
- Joined: Fri Sep 24, 2021 5:34 am
-
Ramya_Paravastu_1H
- Posts: 54
- Joined: Mon Jan 03, 2022 10:55 am
Re: Q<K?
If Q<K, the reaction favors the product side and shifts right (acting in the forward direction) to achieve equilibrium.
-
Zoe Dhalla 3I
- Posts: 104
- Joined: Fri Sep 24, 2021 5:44 am
Re: Q<K?
If Q<K , then the reaction favors the products. The ratio of products to reactants is less than that for the system at equilibrium—the concentration or the pressure of the reactants is greater than the concentration or pressure of the products. Because the reaction tends toward reach equilibrium, the system shifts to the RIGHT to make more products.
-
Jordyn Lee 1J
- Posts: 106
- Joined: Fri Sep 24, 2021 7:08 am
Re: Q<K?
Q=[P]/[R], so if Q<K the concentration of reactants is greater than products. This means that the reaction will proceed in the forward direction until it reaches the equilibrium constant again.
-
Hannah Carsey 1B
- Posts: 100
- Joined: Fri Sep 24, 2021 6:05 am
Re: Q<K?
When Q<K, that means the reaction quotient is a smaller value than the equilibrium constant, so when comparing Q to the ratio of K, there is too much reactant and not enough product given that Q is less than K, meaning that the reaction will shift to the right (the forward reaction) and produce product until the system reaches equilibrium (when Q is equal to K). (Q=[P]/[R], K=[P]/[R]).
-
Alex Luong 3H
- Posts: 108
- Joined: Fri Sep 24, 2021 7:18 am
Re: Q<K?
It means that there were more reactants than usual, so the reaction will favor the products (forward rxn).
-
Amy Jordan 2A
- Posts: 104
- Joined: Fri Sep 24, 2021 6:23 am
-
Caleb_Mei_1J
- Posts: 105
- Joined: Fri Sep 24, 2021 7:09 am
Re: Q<K?
Hey there!
When K>Q, this means that the reaction will shift right and favor the formation of products (the forward reaction) since at this particular point in time there are more reactants than there would be at equilibrium.
I hope this helps!
When K>Q, this means that the reaction will shift right and favor the formation of products (the forward reaction) since at this particular point in time there are more reactants than there would be at equilibrium.
I hope this helps!
-
Cadence Chang
- Posts: 135
- Joined: Fri Sep 24, 2021 6:05 am
- Been upvoted: 1 time
Re: Q<K?
Q < K means that in that instant, the forward reaction is favored since the ratio of products to reactants is less than normal. Because of Le Chatlier's principle, the system works to re-establish equilibrium which, in the case of Q < K, means making more products.
-
Brooke Gushiken 1B
- Posts: 101
- Joined: Fri Sep 24, 2021 6:48 am
Re: Q<K?
If Q<K, then at the moment Q is being calculated, there are too many reactants and not enough products to reach the equilibrium value, since Q and K both = products/reactants. Therefore, the reaction will need to shift right/proceed forward to produce more products, therefore raising the Q value to reach equilibrium.
-
Michael 1G
- Posts: 101
- Joined: Fri Sep 24, 2021 6:16 am
- Been upvoted: 1 time
Re: Q<K?
When Q is less than K it means that there are significantly more reactants than products, favoring the forward reaction. This means more product will be formed.
-
Jessica Servoss 1H
- Posts: 92
- Joined: Fri Sep 24, 2021 7:07 am
- Been upvoted: 1 time
Re: Q<K?
When Q<K, the forward reaction is favored, meaning that there is a greater concentration of reactants, which are creating more products.
-
Vashe Sundar 3H
- Posts: 109
- Joined: Fri Sep 24, 2021 6:09 am
- Been upvoted: 1 time
Re: Q<K?
When Q<K, the products are favored. Since, Q/K = [P]/[R], the situation where Q<K would mean that the reaction quotient has less products or too many reactants needed to establish equilibrium. Meaning, in order to establish equilibrium, the forward reaction would be favored to produce more products, so that Q = K.
-
Darren Apostol 2L
- Posts: 102
- Joined: Fri Sep 24, 2021 5:12 am
- Been upvoted: 1 time
Re: Q<K?
Q<K ⮞ 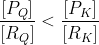
The fraction representing Q must increase its numerator/decrease its denominator (product⭧/reactant⭨) to reach K, so the reaction will favor products (shift right).
The fraction representing Q must increase its numerator/decrease its denominator (product⭧/reactant⭨) to reach K, so the reaction will favor products (shift right).
-
Macy_Anderson_2F
- Posts: 100
- Joined: Fri Sep 24, 2021 5:02 am
Re: Q<K?
If Q > K the forward reaction is favored because there was a higher concentration of reactants than products. Conversely, if Q < K the reverse reaction is favored for the opposite reasoning.
-
Sabira Mohammed 3I
- Posts: 100
- Joined: Fri Sep 24, 2021 5:34 am
Re: Q<K?
if Q is less than K, then rate of the forward reaction is faster and more products than reactants are being made (reaction shifts left)
-
Alice Weber 3I
- Posts: 94
- Joined: Fri Sep 24, 2021 7:27 am
Re: Q<K?
When Q is less than K, the concentration of the reactants in greater than the concentration of the products and a forward reaction is favored.
-
N Kanuri 2E
- Posts: 109
- Joined: Fri Sep 24, 2021 5:41 am
- Been upvoted: 1 time
Re: Q<K?
If Q<K, the reaction currently has more reactants/less products than the reaction would at equilibrium. Thus, the reaction will proceed in the forward direction.
-
Madelyn_Rios_2c
- Posts: 110
- Joined: Fri Sep 24, 2021 5:54 am
Re: Q<K?
If Q<K at some point during the reaction, this means that the concentration of reactants is greater than the concentration of products. This makes the forward reaction be favored meaning more products will be made.
-
Ameerah Hameed 3B
- Posts: 112
- Joined: Fri Sep 24, 2021 5:32 am
Re: Q<K?
When Q is less than K, the reaction has a greater concentration of reactants compared to that of the products. Additionally, this means a forward reaction is favored.
-
Arden Napoli 1E
- Posts: 100
- Joined: Fri Sep 24, 2021 5:19 am
Re: Q<K?
When Q<K, that means that the concentration of reactants is higher than the products, therefore the reaction will shift to the right in the forward direction.
-
Natalie Quilala 1I
- Posts: 105
- Joined: Fri Sep 24, 2021 5:28 am
-
Karen Harrison 1L
- Posts: 105
- Joined: Fri Sep 24, 2021 7:07 am
Re: Q<K?
Hello! A visual that really helped me understand the relationship between reaction quotients and equilibrium constants is the image below.
If Q is less than K, the reaction is going in the forward direction because there are fewer products formed than the system would like to be at. Because of this, the system will shift towards the production of products to compensate for the lack of products.
If Q is more than K, the reaction is going in the reverse direction because there are more products formed than the system would like to be at. Because of this, the system will shift towards the formation of reactants to compensate.
If Q is equal to K, there is no shifting taking place.
The image shows that K is the ideal state of the system, and if Q is above or below that, the system will naturally proceed in a way to achieve the state of equilibrium.
If Q is less than K, the reaction is going in the forward direction because there are fewer products formed than the system would like to be at. Because of this, the system will shift towards the production of products to compensate for the lack of products.
If Q is more than K, the reaction is going in the reverse direction because there are more products formed than the system would like to be at. Because of this, the system will shift towards the formation of reactants to compensate.
If Q is equal to K, there is no shifting taking place.
The image shows that K is the ideal state of the system, and if Q is above or below that, the system will naturally proceed in a way to achieve the state of equilibrium.
-
Rishab_Haldar_3B
- Posts: 112
- Joined: Fri Sep 24, 2021 7:26 am
Re: Q<K?
Hi, Q<K means that the reaction will shift to the right in the forward direction since the concentration of reactants is higher than the products.
-
Abu Zhang 2D
- Posts: 37
- Joined: Mon Jan 03, 2022 8:42 pm
Re: Q<K?
Q and K are both ratios of reactant to product, and the only difference is that Q is not under equilibrium. Therefore, because the product is the numerator and the reactant is the denominator, if Q<K, it means that the current product is less than the product concentration under equilibrium. Thus, the reaction will shift right, the side that favors more product formation.
-
Daniela G 2C
- Posts: 92
- Joined: Fri Sep 24, 2021 5:06 am
Re: Q<K?
When Q<K, the reactants are still going to products during the RxN. The forward reaction is favored.
-
Skylar Lo 2C
- Posts: 110
- Joined: Fri Sep 24, 2021 5:10 am
Re: Q<K?
When Q<K that means the reaction favors the products and the forward reaction will be more favored, causing production of products.
-
Sarah Hong 2K
- Posts: 102
- Joined: Fri Sep 24, 2021 5:48 am
-
Talia Tam 3L
- Posts: 107
- Joined: Fri Sep 24, 2021 5:38 am
-
Nicole Friday 1E
- Posts: 51
- Joined: Mon Jan 03, 2022 10:45 am
-
Chris Van 2J
- Posts: 102
- Joined: Fri Sep 24, 2021 5:28 am
- Been upvoted: 1 time
-
JasmineReyes-2K
- Posts: 97
- Joined: Fri Sep 24, 2021 6:12 am
Re: Q<K?
When K is greater than Q , our forward reaction is favored, meaning there are more products than reactants
-
Chris Korban 1D
- Posts: 107
- Joined: Fri Sep 24, 2021 6:53 am
Re: Q<K?
When Q is less than K it means that the Reactant concentration increased and therefore the forward reaction will be favored
-
RobinFong_2B
- Posts: 53
- Joined: Mon Jan 03, 2022 8:44 pm
Re: Q<K?
If the reactant quotient(Q) is less than the equilibrium constant (K), then the current ratio of products to reactants is less than what is expected when the reaction reached equilibrium. Thus, the chemical equation will favor the forward direction to increase the concentration of products in order to reach equilibrium.
-
Amanda Nguyen dis 2E
- Posts: 102
- Joined: Fri Sep 24, 2021 6:36 am
-
sidneypalacios
- Posts: 101
- Joined: Fri Sep 24, 2021 5:30 am
Re: Q<K?
If Q is greater than K than that means there is a higher concentration of reactants vs. products and so the reaction will proceed in the forward direction (toward the products).
-
Isabella Perez Dis3L
- Posts: 100
- Joined: Fri Sep 24, 2021 6:38 am
Re: Q<K?
Hi! Q<K means during RxN, R>P (reactants > products), and the forward reaction is favored & proceeds toward products. When Q>K during RxN, then P>R and the reverse reaction is favored & proceeds toward reactants.
-
Alaura Dis 1H
- Posts: 66
- Joined: Fri Sep 24, 2021 7:24 am
Re: Q<K?
The relationship between Q and K determines the direction in which the reaction will proceed. When Q>K, then the products are favored, and when Q<K, the reactants are favored.
-
Bobak Pourrahimi 2L
- Posts: 108
- Joined: Fri Sep 24, 2021 6:09 am
Re: Q<K?
Q determines the current state of the reaction, while K displays the ideal product-reactant concentration ratio for equilibrium. If Q<K, it indicates there are more reactants and fewer products than there should be at equilibrium, and the reaction would adjust. Similarly, the opposite is occurring if Q>K.
-
Polo Morales 3C
- Posts: 103
- Joined: Fri Sep 24, 2021 7:01 am
Re: Q<K?
If Q<K, it means that the reaction has not yet reached equilibrium, and more products have yet to be created. Remember that Q=K at equilibrium and Q = [P]/[R] at any given time. Hope this helps!
-
Veronica Larson- 1I
- Posts: 103
- Joined: Wed Nov 11, 2020 12:18 am
Re: Q<K?
If Q<K, the concentration of reactants is greater than the concentration of products. This means that the reaction will proceed in the forward direction and favor the formation of products.
-
amara ajon 1d
- Posts: 109
- Joined: Fri Sep 24, 2021 6:34 am
Re: Q<K?
Hi,
When Q<K, the reaction is not in equilibrium and we will have more products formed as the forward reaction is favored.
When Q<K, the reaction is not in equilibrium and we will have more products formed as the forward reaction is favored.
-
Collin Le 3I
- Posts: 52
- Joined: Mon Jan 03, 2022 9:35 pm
Re: Q<K?
If Q is less than K, that means the equilibrium state has a larger product to reactant ratio than the current state Q. This means the reaction will proceed in the forward direction.
-
Ruirui Lan
- Posts: 50
- Joined: Mon Jan 03, 2022 9:15 pm
Re: Q<K?
K represents the constant when the chemical equation reaches a dynamic equilibrium, and Q indicates the instant status of a chemical reaction. Therefore, when Q is smaller than K, we can infer based on this information in which direction the chemical reaction would proceed in. The K constant is calculated using products over reactants, so if Q is smaller than K, it means that Q has more reactants than products in its product to reactant ratio compared to K. Therefore, in order to proceed to chemical equilibrium which is represented by K. it will orient itself in favor of aligning its value with K. Therefore, it will make its reactants go down and products go up, which means the chemical reaction will proceed in the forward direction, decreasing reactants and increasing products.
-
Kaitlyn_Urquilla_1I
- Posts: 102
- Joined: Fri Sep 24, 2021 7:15 am
Re: Q<K?
When Q<K, that means that the reaction favors the forward reaction because there is still more reactant to form products with.
-
oliviahelou
- Posts: 103
- Joined: Fri Sep 24, 2021 6:36 am
-
Cynthia_L_2C
- Posts: 99
- Joined: Fri Sep 24, 2021 7:25 am
Re: Q<K?
Q<K means that the forward reaction is favored. There is still more reactants to form products.
-
Vikram Sharma
- Posts: 105
- Joined: Fri Sep 24, 2021 5:32 am
Re: Q<K?
When Q is less than K, there are more reactants than products than there should be at equilibrium. This means the equilibrium should shift to the products.
-
Diana Avalos
- Posts: 55
- Joined: Tue Feb 16, 2021 12:15 am
Re: Q<K?
if you remember, Kc or Qc= [products]/[reactants]. If Q<K, this means there are more reactants and the formation of products would be favored in order to reach equilibrium. Hope this helps!
-
Muryam_Hasan_2I
- Posts: 100
- Joined: Fri Sep 24, 2021 6:52 am
- Been upvoted: 1 time
Re: Q<K?
Since Q and K are both calculated by the equation products/reactants, Q being less than K means that there are more reactants (greater denominator) in the current reaction than there is at equilibrium, shown by K. This means that the reaction still has to form more products, such that the forward reaction is favored.
-
Minoo Bastani 2J
- Posts: 103
- Joined: Fri Sep 24, 2021 6:51 am
Re: Q<K?
If Q is less than K, this means that the reaction is not at equilibrium and that the concentration of the products at that point in the reaction is less than what the concentration of products is at equilibrium, and that the forward reaction is favored as that reaction continues towards equilibrium.
-
Ruben Adamov 1E
- Posts: 105
- Joined: Fri Sep 24, 2021 5:56 am
-
Santiago Cortes 2J
- Posts: 49
- Joined: Mon Jan 03, 2022 9:04 pm
Re: Q<K?
If Q<K then this means that there are more reactants in relation to the products. Thus the reaction shifts to the right.
-
Santiago Cortes 2J
- Posts: 49
- Joined: Mon Jan 03, 2022 9:04 pm
Re: Q<K?
If Q>K then this means that there are more products in relation to reactants. Thus the reaction will shift to the left and favor the reactants.
-
Abigail Tran 14a
- Posts: 126
- Joined: Fri Sep 24, 2021 7:12 am
Re: Q<K?
when q<k the amount of products at the time is greater than the amount of products at equilibrium so the reaction will shift left towards reactants to reach equillibrium
-
Marie Khijniak 2E
- Posts: 101
- Joined: Fri Sep 24, 2021 6:22 am
Re: Q<K?
Hi!
If Q<K, this means that in the current state of the system, there are more reactants than there should be at equilibrium, so the system will favor products and shift to the right to produce more products to reach equilibrium.
Hope this helps!
If Q<K, this means that in the current state of the system, there are more reactants than there should be at equilibrium, so the system will favor products and shift to the right to produce more products to reach equilibrium.
Hope this helps!
-
davis sandberg 2H
- Posts: 104
- Joined: Fri Sep 24, 2021 7:30 am
-
RobinFong_2B
- Posts: 53
- Joined: Mon Jan 03, 2022 8:44 pm
Re: Q<K?
It means that the reaction will favor the production of products to move towards equilibrium. Because the reaction quotient measures [P]/[R] of the reaction not in equilibrium it is telling of the direction that the reaction will move in to achieve equilibrium. Having a value less than K means that its concentration of products is less than what it should be in equilibrium. Thus the reaction will make more products so that it moves towards equilibrium.
Re: Q<K?
When Q is less than K, that means the reaction will be proceeding in the forward direction, where products are being formed. When Q>K, that means the process is going in the reverse direction, where products will go towards reactants.
-
Esther Kim
- Posts: 99
- Joined: Fri Sep 24, 2021 6:07 am
Re: Q<K?
Q<K it means then it will proceed towards the forward direction while as if Q>K then the reaction will proceed in the reverse direction
-
Parinita Jithendra 2A
- Posts: 101
- Joined: Fri Sep 24, 2021 6:57 am
Re: Q<K?
When Q is less than K, the reaction will go in the forward direction which means that reaction will favor the production of products.
-
Palig Kechichian 3F
- Posts: 102
- Joined: Fri Sep 24, 2021 5:26 am
Re: Q<K?
When Q<K, you have more reactants so the reactions will shift in the forward direction to produce more products
-
Miranda Hess 3I
- Posts: 100
- Joined: Fri Sep 24, 2021 6:15 am
Return to “Non-Equilibrium Conditions & The Reaction Quotient”
Who is online
Users browsing this forum: No registered users and 8 guests

