Le Chatelier's Principle on Temperature
Moderators: Chem_Mod, Chem_Admin
-
Victoria Luu - 1C
- Posts: 60
- Joined: Fri Sep 28, 2018 12:15 am
Le Chatelier's Principle on Temperature
Which is favored when the heat is lowered in a reaction? Would it move towards the exothermic reaction?
-
armintaheri
- Posts: 68
- Joined: Fri Sep 28, 2018 12:26 am
Re: Le Chatelier's Principle on Temperature
If the reaction is exothermic, heat is released as a result of the reaction. Therefore, lowering heat is like removing a product and the equilibrium shifts toward the products.If the reaction is endothermic, removing heat is like removing a reactant, so the equilibrium shifts toward the reactants.
-
Luc Lorain 1L
- Posts: 59
- Joined: Fri Sep 28, 2018 12:18 am
Re: Le Chatelier's Principle on Temperature
Though it is not technically correct scientific notation, you can think about heat like its own species in an equilibrium equation. If a reaction is endothermic, ( H>0), think of heat as a reactant, and when exothermic ( H<0), consider heat a product. This makes the application of Le Chatelier's Principle much more straightforward, as a change in temperature can now be contextualized as a change in a reactant or product, which I personally find easier to contextualize.
For instance, take the endothermic reaction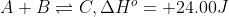 . This can be rewritten in the terms explained above to be:
. This can be rewritten in the terms explained above to be: 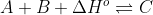 .
.
For this new equation, an increase in temperature --> an increase in heat; thus product formation is favored. If temperature is decreased, heat decreases--> reactant formation. It's that easy!
For instance, take the endothermic reaction
For this new equation, an increase in temperature --> an increase in heat; thus product formation is favored. If temperature is decreased, heat decreases--> reactant formation. It's that easy!
-
Ahmed Mahmood 4D
- Posts: 72
- Joined: Fri Sep 28, 2018 12:28 am
Re: Le Chatelier's Principle on Temperature
Heat can be seen as part of the reaction, with an exothermic reaction having heat in the products and an endothermic reaction having heat in the reactants. Thus, lowering temperature for an exothermic reaction will cause the reaction to sit to the right.
-
Yiting_Gong_4L
- Posts: 69
- Joined: Fri Sep 28, 2018 12:25 am
Re: Le Chatelier's Principle on Temperature
When the reaction is exothermic that means heat is being released as a product. If you are lowering the temperature you are essentially removing heat and that means the reaction will favor the product.
Return to “Applying Le Chatelier's Principle to Changes in Chemical & Physical Conditions”
Who is online
Users browsing this forum: No registered users and 6 guests

