Catalysts
Moderators: Chem_Mod, Chem_Admin
-
megangeorge-1K
- Posts: 59
- Joined: Thu Feb 01, 2018 3:01 am
-
AnnaYan_1l
- Posts: 96
- Joined: Fri Apr 06, 2018 11:05 am
- Been upvoted: 1 time
Re: Catalysts
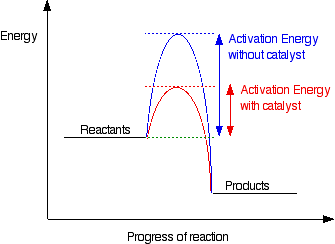
Catalysts reduce the activation energy between reactants to products, meaning it takes less energy to make the forward reaction occur
-
Michelle Wang 4I
- Posts: 63
- Joined: Fri Sep 28, 2018 12:27 am
Re: Catalysts
Catalysts work by giving an entirely new pathway for the reaction to occur with a lower activation energy. Therefore, the curve would be lower.
-
Rogelio Bazan 1D
- Posts: 64
- Joined: Tue Nov 14, 2017 3:01 am
Re: Catalysts
A catalyst brings down the activation energy, effectively increasing the speed at which the reaction occurs. Graphically this would bring down the very top peak on the graph as the activation energy is lowered to make the reaction occur. Hope this helps.
-
Jacqueline Duong 1H
- Posts: 61
- Joined: Fri Sep 28, 2018 12:28 am
Re: Catalysts
A catalyst would lower the peak on a curve since it can speed up the rate of a chemical reaction by providing a faster route.
-
Phil Timoteo 1K
- Posts: 101
- Joined: Fri Apr 06, 2018 11:05 am
-
sallina_yehdego 2E
- Posts: 75
- Joined: Sun Apr 29, 2018 3:00 am
-
Claudia Luong 4K
- Posts: 59
- Joined: Fri Sep 28, 2018 12:25 am
-
Gillian Ward 1F
- Posts: 61
- Joined: Fri Sep 28, 2018 12:27 am
-
Rosa Munoz 2E
- Posts: 105
- Joined: Wed Sep 18, 2019 12:21 am
-
Andres Merlos 2L
- Posts: 46
- Joined: Wed Sep 18, 2019 12:17 am
-
kristi le 2F
- Posts: 102
- Joined: Thu Jul 11, 2019 12:15 am
Re: Catalysts
Catalysts lower the activation barrier, so a greater fraction of reactants can cross the new lower barrier and convert into products.
-
Chantel_2I
- Posts: 109
- Joined: Sat Sep 07, 2019 12:19 am
Re: Catalysts
The way a catalyst lowers the activation energy is by creating a completely new path to get to the products, which has a lower activation energy.
-
CameronDis2K
- Posts: 51
- Joined: Wed Feb 20, 2019 12:18 am
Re: Catalysts
A catalyst will lower the activation energy (Ea) in a reaction, causing the curve of the rxn process vs energy graph curve to be lower than without a catalyst.
Re: Catalysts
AnnaYan_1l wrote:Catalysts reduce the activation energy between reactants to products, meaning it takes less energy to make the forward reaction occur
does this affect the reverse reaction too?
-
JohannaPerezH2F
- Posts: 94
- Joined: Wed Sep 18, 2019 12:18 am
-
Jacob_Eberson_2D
- Posts: 50
- Joined: Mon Jan 03, 2022 8:40 pm
-
Saatvika Nair
- Posts: 49
- Joined: Wed Feb 17, 2021 12:17 am
Re: Catalysts
Catalysts lower the activation energy, so the way I like to think about it, on a graph, the reaction with a catalyst will have a smaller "hump" than the reaction without a catalyst. This is what speeds up the reaction.
-
Srikar_Chintala_1E
- Posts: 105
- Joined: Fri Sep 24, 2021 6:33 am
Re: Catalysts
Catalysts reduce the amount of activation energy required for the reaction to proceed.
-
Zoe Dhalla 3I
- Posts: 104
- Joined: Fri Sep 24, 2021 5:44 am
Re: Catalysts
A catalyst speeds up a chemical reaction, without being consumed by the reaction. It increases the reaction rate by lowering the activation energy for a reaction.
-
Violet Mbela 2B
- Posts: 98
- Joined: Fri Sep 24, 2021 5:15 am
Re: Catalysts
Hi,
Since catalysts speed up a reaction, the activation energy is lowered. So graphically, the addition of a catalyst lowers the overall curve.
Since catalysts speed up a reaction, the activation energy is lowered. So graphically, the addition of a catalyst lowers the overall curve.
-
Lesley Kim 1K
- Posts: 50
- Joined: Mon Jan 03, 2022 11:06 am
Re: Catalysts
Catalysts lower the activation energy needed in order for a reaction to take place. By doing so, the reaction requires less energy in order to occur which helps to speed up the reaction rate of a reaction.
-
alexjung1A
- Posts: 101
- Joined: Fri Sep 24, 2021 7:07 am
Re: Catalysts
Catalysts work be reducing the activation energy of a reaction, so it occurs "earlier" than if it was uncatalyzed, speeding up reaction rate.
-
JennyZhu1K
- Posts: 101
- Joined: Fri Sep 24, 2021 7:12 am
Re: Catalysts
Both catalysts and enzymes decrease the activation energy of a reaction so they will make the reaction faster that way.
-
Zechuan Rao 1K
- Posts: 99
- Joined: Fri Sep 24, 2021 5:26 am
Re: Catalysts
catalysts redirect the steps of the reaction, enable a path with intermediate complexes that are more thermodynamically favorable than if the catalyst is not involved. This means at any given time at a specific temperature, more complex can be formed, and more reaction can be carried out, making the rate of reaction higher.
-
Michelle Gong
- Posts: 104
- Joined: Fri Sep 24, 2021 7:10 am
Re: Catalysts
Hellp!
Catalysts lower the activation energy and therefore make the "hump" in the curve appear earlier.
Catalysts lower the activation energy and therefore make the "hump" in the curve appear earlier.
-
Matthew Li 1B
- Posts: 118
- Joined: Fri Sep 24, 2021 6:40 am
-
Gabrielle Malte 2G
- Posts: 102
- Joined: Fri Sep 24, 2021 6:53 am
Return to “Arrhenius Equation, Activation Energies, Catalysts”
Who is online
Users browsing this forum: No registered users and 5 guests