Clarification on Frequency
Moderators: Chem_Mod, Chem_Admin
-
Melvin Reputana 1L
- Posts: 55
- Joined: Sat Aug 17, 2019 12:16 am
Clarification on Frequency
I understand that frequency is essentially the number of times a point on the wave passes a fixed area in one second. However, how can you manipulate the frequency of a wave? Is it by increasing the intensity of the light?
-
Justin Sarquiz 2F
- Posts: 106
- Joined: Fri Aug 30, 2019 12:15 am
Re: Clarification on Frequency
We can manipulate frequency by changing the wavelength since frequency and wavelength are inversely proportional [c=lambda (v)]. Increasing the intensity only increases the amplitude of the wave and has no effect on the frequency or wavelength.
Re: Clarification on Frequency
Also just to add on!
Shorter wavelengths have a higher frequency and a greater energy (this would be things like purple light)
Longer wavelengths have a lesser frequency and not as strong of energy (this would be red light)
As energy increases, frequency increases since they are proportional to each other
Shorter wavelengths have a higher frequency and a greater energy (this would be things like purple light)
Longer wavelengths have a lesser frequency and not as strong of energy (this would be red light)
As energy increases, frequency increases since they are proportional to each other
-
Andrew Liang 1I
- Posts: 105
- Joined: Fri Aug 30, 2019 12:18 am
Re: Clarification on Frequency
We cannot really change the frequency of a wave unless we completely change the type of the electromagnetic wave. Frequency of an electromagnetic wave normally remains the same because each electromagnetic wave has its own unique driven oscillation.
When we increase the intensity of the light, we are only increasing the amplitude of the wave or in other words adding more light photons with the same energy.
When we increase the intensity of the light, we are only increasing the amplitude of the wave or in other words adding more light photons with the same energy.
-
Shrayes Raman
- Posts: 129
- Joined: Sat Jul 20, 2019 12:15 am
Re: Clarification on Frequency
Increasing the intensity is not the same as changing the frequency. When we change frequency we are changing the energy of each photon of the incoming em radiation. When we change intensity we change the number of the photons coming in.
-
Caroline Zepecki
- Posts: 101
- Joined: Fri Aug 09, 2019 12:16 am
Do photons change with wavelength?
I'm trying to visualize some of what we went over in lecture about how shorter, less intense wavelengths have enough energy in a single photon to displace elections. I was wondering what about photons changed with wavelength (is it intensity?), and if there's an equation that relates them?
-
Julia Mazzucato 4D
- Posts: 64
- Joined: Fri Aug 30, 2019 12:17 am
Re: Clarification on Frequency
To answer 905289082:
The energy of the light is related to the frequency of it, not the intensity per se. Amplitude is intensity (the brighter the light, the higher the amplitude), but the amplitude of the light has no bearing on the equation of the energy. The wavelength of the light has a direct effect on the frequency because of the equation c=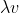 - we know that as wavelength gets shorter, the frequency gets higher. It's easier to visualize this if you look at a graph. The shorter the distance between a wave's peaks means that the wave is more "compressed", so more complete waves pass by in a second. Because E=hv, a higher frequency will increase the energy per photon. But changing the amplitude doesn't do that cause you're just making the light "brighter", not higher energy. So the energy per photon won't increase, you'll just have more of those low-energy photons. An analogy to visualize this: you have a machine that will requires you to insert one (and only one) coin with the value of 10 cents. You only have nickels. You can increase the amount of nickels you have (increase the intensity), but having more coins won't help because no single coin you have is 10 cents. And you can't combine two nickels to make ten cents because you specifically can only use one coin (one photon to one electron interaction). You need a higher value (higher energy) dime.
- we know that as wavelength gets shorter, the frequency gets higher. It's easier to visualize this if you look at a graph. The shorter the distance between a wave's peaks means that the wave is more "compressed", so more complete waves pass by in a second. Because E=hv, a higher frequency will increase the energy per photon. But changing the amplitude doesn't do that cause you're just making the light "brighter", not higher energy. So the energy per photon won't increase, you'll just have more of those low-energy photons. An analogy to visualize this: you have a machine that will requires you to insert one (and only one) coin with the value of 10 cents. You only have nickels. You can increase the amount of nickels you have (increase the intensity), but having more coins won't help because no single coin you have is 10 cents. And you can't combine two nickels to make ten cents because you specifically can only use one coin (one photon to one electron interaction). You need a higher value (higher energy) dime.
The energy of the light is related to the frequency of it, not the intensity per se. Amplitude is intensity (the brighter the light, the higher the amplitude), but the amplitude of the light has no bearing on the equation of the energy. The wavelength of the light has a direct effect on the frequency because of the equation c=
-
ValerieChavarin 4F
- Posts: 99
- Joined: Wed Sep 18, 2019 12:18 am
Re: Clarification on Frequency
Increasing the intensity of light will not change the frequency of a wave, it will only change the amplitude of the wave.
-
stephaniekim2K
- Posts: 79
- Joined: Fri Aug 09, 2019 12:16 am
Re: Clarification on Frequency
Since C=λν, frequency and wavelength are inversely proportional, meaning that as one increases, the other decreases since the speed of light, c, is constant. By decreasing the wavelength, you can increase the frequency.
Return to “Properties of Light”
Who is online
Users browsing this forum: No registered users and 13 guests

