Equation use
Moderators: Chem_Mod, Chem_Admin
-
Matt Sanruk 2H
- Posts: 131
- Joined: Wed Sep 18, 2019 12:21 am
Re: Equation use
I don't think the question would directly tell you to use this equation, but I know when we lectured about the photoelectric effect, this equation was required to complete the worked example. So if you have those notes, I would take a look back at those! We also reference this equation when we derived the De Broglie Equation as well.
-
Kelvin Chung 1C
- Posts: 50
- Joined: Sat Aug 24, 2019 12:15 am
Re: Equation use
If you have one of the two unknown values of this equation (E or v) and need to solve for the other, use this equation.
-
Amy Pham 1D
- Posts: 103
- Joined: Fri Aug 09, 2019 12:15 am
Re: Equation use
Additionally, the question would have to be dealing with electromagnetic radiation, not electron wave properties. A question relating to the latter would likely use the De Broglie equation or Schrodinger's wave function equation.
-
Eesha Sohail 1D
- Posts: 100
- Joined: Sat Aug 17, 2019 12:16 am
Re: Equation use
The equation is used when referring to the energy of light/electromagnetic radiation, not with matter. v refers to the frequency of the light; if the problem gives you wavelength, you can use 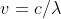 to find that frequency. Then you can use Planck's constant to find the energy of the particle. Alternatively, if the energy is given, the E=hv equation can be used in reverse to find the light's frequency.
to find that frequency. Then you can use Planck's constant to find the energy of the particle. Alternatively, if the energy is given, the E=hv equation can be used in reverse to find the light's frequency.
-
Callum Guo 1H
- Posts: 134
- Joined: Fri Aug 02, 2019 12:15 am
- Been upvoted: 1 time
Re: Equation use
you would just have to see what you're looking for and what you're given. I think the best way to learn is through practice.
-
Jennifer Yang 3F
- Posts: 56
- Joined: Wed Sep 11, 2019 12:17 am
Re: Equation use
I believe they would give you two of three variables (frequency, Planck's variable, and energy), asking you to find the one last variable. Also, you might have to use other equations to find one of those variables.
Who is online
Users browsing this forum: No registered users and 9 guests

