Calorimeter
Moderators: Chem_Mod, Chem_Admin
Calorimeter
What is the difference between a calorimeter and a bomb calorimeter? Thank you in advance.
Re: Calorimeter
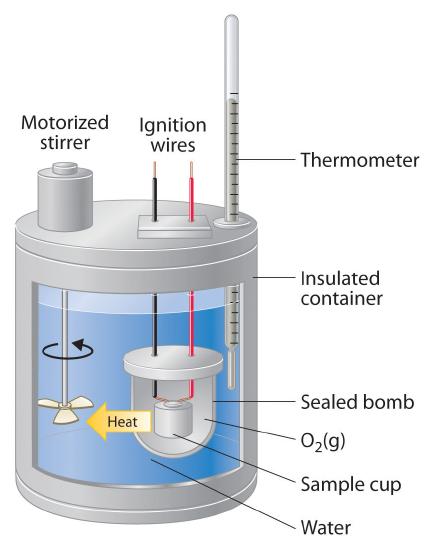
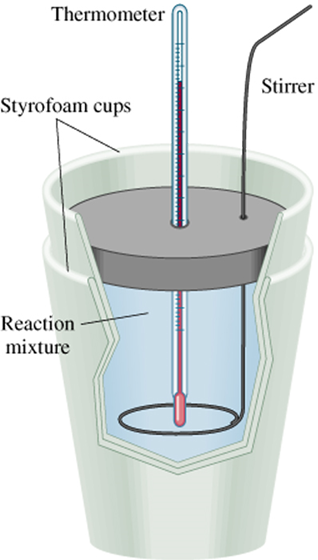
A calorimeter is just a "thermally insulated container" to measure changes of heat while a bomb calorimeter is the same thing but can withstand more pressure.
-
Juliana Chopelas 1A
- Posts: 50
- Joined: Sat Aug 24, 2019 12:16 am
Re: Calorimeter
a calorimeter is a normal insulated container while a bomb calorimeter consists of a sealed metal container that is in another sealed container immersed in water to measure the heat combustion of a reaction
-
Rebekah Alfred 1J
- Posts: 102
- Joined: Thu Jul 11, 2019 12:15 am
- Been upvoted: 1 time
-
CMaduno_1L
- Posts: 102
- Joined: Wed Sep 18, 2019 12:18 am
Re: Calorimeter
Although both are structured differently, it is important to remember that they serve the same function-- to measure specific heat capacity (often referred to as specific heat).
-
stephaniekim2K
- Posts: 79
- Joined: Fri Aug 09, 2019 12:16 am
-
Jasmine 2C
- Posts: 184
- Joined: Wed Sep 18, 2019 12:18 am
Re: Calorimeter
Bomb calorimeter is just a specific type of calorimeter. It works best when trying to keep volume and moles of the gas constant bc it's perfectly sealed and isolated.
-
Omar Selim 1D
- Posts: 108
- Joined: Sat Jul 20, 2019 12:16 am
Re: Calorimeter
A bomb calorimeter is a more robust and accurate calorimeter. It can withstand more pressure, so it is typically more often used
-
Jesse Anderson-Ramirez 3I
- Posts: 54
- Joined: Thu Sep 26, 2019 12:18 am
Return to “Phase Changes & Related Calculations”
Who is online
Users browsing this forum: No registered users and 4 guests