Kelvin or Celsius?
Moderators: Chem_Mod, Chem_Admin
-
Veronica Lu 2H
- Posts: 89
- Joined: Wed Sep 18, 2019 12:18 am
- Been upvoted: 1 time
Re: Kelvin or Celsius?
I think you use Kelvin because the gas constant R always has Kelvin in the denominator so they cancel out!
-
Timmy Nguyen Dis 1I
- Posts: 107
- Joined: Sat Aug 17, 2019 12:17 am
-
Shrayes Raman
- Posts: 129
- Joined: Sat Jul 20, 2019 12:15 am
Re: Kelvin or Celsius?
Kelvin... The gas constant R is listed in Kelvin so Kelvin is used in equation.
-
Alexis Robles 2k
- Posts: 100
- Joined: Wed Sep 18, 2019 12:18 am
Re: Kelvin or Celsius?
Whenever it is T alone, you should use kelvin but when it is deltaT, you can use Celcius or kelvin.
-
Venus_Hagan 2L
- Posts: 104
- Joined: Fri Aug 02, 2019 12:16 am
Re: Kelvin or Celsius?
for the ideal gas equation use Kelvin but if you ever need the change in temperature it doesn't matter because both are scaled the same way
Re: Kelvin or Celsius?
Kelvins are always used when applying the ideal gas law 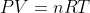
The reason this is done is because Kelvins are more mathematically accurate in terms of chemically applying the excited state of the molecules in a gas to your calculations. zero K is the same as zero energy in the molecules, and is therefore the same as zero movement of the molecules. This is more mathematically correct than -273.15oC
If you really want, I'm sure you could look up values for the Ideal Gas Constant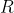 , but most are given in K because they're better for representing how the energy of the molecules affects the context.
, but most are given in K because they're better for representing how the energy of the molecules affects the context.
The reason this is done is because Kelvins are more mathematically accurate in terms of chemically applying the excited state of the molecules in a gas to your calculations. zero K is the same as zero energy in the molecules, and is therefore the same as zero movement of the molecules. This is more mathematically correct than -273.15oC
If you really want, I'm sure you could look up values for the Ideal Gas Constant
-
Rosa Munoz 2E
- Posts: 105
- Joined: Wed Sep 18, 2019 12:21 am
Re: Kelvin or Celsius?
Kelvin. it only doesn't matter if we are talking about change. If it is a capital T then it must be Kelvin
-
Juana Abana 1G
- Posts: 100
- Joined: Wed Sep 18, 2019 12:15 am
-
Aarushi Solanki 4F
- Posts: 107
- Joined: Sat Jul 20, 2019 12:17 am
-
Jesse Anderson-Ramirez 3I
- Posts: 54
- Joined: Thu Sep 26, 2019 12:18 am
-
Anish Natarajan 4G
- Posts: 51
- Joined: Thu Jul 25, 2019 12:16 am
Re: Kelvin or Celsius?
Technically it's all dependent on the value of R that you use, but in most cases you'll be given a value that incorporates Kelvin rather than Celsius
-
Orrin Zhong 4G
- Posts: 51
- Joined: Sat Jul 20, 2019 12:16 am
-
AronCainBayot2K
- Posts: 101
- Joined: Fri Aug 30, 2019 12:17 am
-
CalvinTNguyen2D
- Posts: 102
- Joined: Thu Jul 25, 2019 12:16 am
Re: Kelvin or Celsius?
You will almost always be using Kelvin in equations and calculations; in terms of the gas constant, it is measured in Kelvin rather than Celsius.
-
Sean Tran 2K
- Posts: 65
- Joined: Sat Aug 17, 2019 12:17 am
-
AveryAgosto
- Posts: 76
- Joined: Wed Sep 18, 2019 12:16 am
-
Esha Chawla 2E
- Posts: 108
- Joined: Thu Jul 25, 2019 12:17 am
Re: Kelvin or Celsius?
Veronica Lu 2H wrote:when using PV=nRT is the temperature supposed to be celsius or kelvin?
The units for R are L*atm/mol*K. Based on this information, we can determine that R should be in Kelvin.
In general, most chemical reactions use temperature in Kelvin.
-
Aarushi Solanki 4F
- Posts: 107
- Joined: Sat Jul 20, 2019 12:17 am
Re: Kelvin or Celsius?
It's supposed to be in Kelvin. Generally, we use Kelvin, but it is okay to use Celsius in equations involving the "change of T" since the scaling is the same for Kelvin and Celsius.
-
Gurmukhi Bevli 4G
- Posts: 49
- Joined: Wed Nov 14, 2018 12:20 am
Re: Kelvin or Celsius?
Since the STP (standard temperature and pressure) and R constant are in units of Kelvin, you should be using Kelvin for your calculations. If the problem does ask for the answer in units of Celsius, just do the calculations in Kelvin and then convert to Celsius after completing the calculations so you don't mess up unit calculations.
-
Madeline Phan 1E
- Posts: 103
- Joined: Sat Aug 17, 2019 12:18 am
-
Yailin Romo 4G
- Posts: 109
- Joined: Wed Feb 20, 2019 12:16 am
-
Areli C 1L
- Posts: 95
- Joined: Wed Nov 14, 2018 12:19 am
-
Justin Quan 4I
- Posts: 104
- Joined: Sat Sep 14, 2019 12:17 am
Re: Kelvin or Celsius?
Veronica Lu 2H wrote:when using PV=nRT is the temperature supposed to be celsius or kelvin?
To add on, you use kelvin especially since the constant R is in kelvin, not celsius. However, if you use PV=nR∆T, the you may use either celsius or kelvin since the change in temperature would be the same.
-
Alfred Barrion 2H
- Posts: 100
- Joined: Sat Jul 20, 2019 12:16 am
Re: Kelvin or Celsius?
You should use Kelvin since it matches with the gas constant R to cancel out.
Re: Kelvin or Celsius?
You use Kelvin because you must match the units within the R (gas constant) which uses kelvin.
-
William Chan 1D
- Posts: 102
- Joined: Sat Sep 14, 2019 12:15 am
Re: Kelvin or Celsius?
Kelvin. If the reaction happened at below 0°C, then the subsequent equation would be negative, having some weird consequences (i.e., negative pressure, negative volume).
-
Bryan Chen 1H
- Posts: 58
- Joined: Mon Jun 17, 2019 7:24 am
Re: Kelvin or Celsius?
it depends on the unit of the gas constant. if its .08206 Latm/Kmol then use Kelvin, but another gas constant COULD have its units in celcius for whatever reason and then you should use celcius (but it probably will never ever come up in real life or a problem)
-
Leonardo Le Merle 1D
- Posts: 56
- Joined: Wed Feb 27, 2019 12:16 am
Re: Kelvin or Celsius?
Kelvin = Celsius + 273.15; the two are on the same scale so converting between the two isn't too tricky.
-
Tanmay Singhal 1H
- Posts: 143
- Joined: Sat Jul 20, 2019 12:16 am
Re: Kelvin or Celsius?
kelvin because the gas constant R is in K. sometimes it is in C and in that case u would use C
-
Kavya Immadisetty 2B
- Posts: 106
- Joined: Sat Aug 24, 2019 12:17 am
- Been upvoted: 1 time
-
Alan Cornejo 1a
- Posts: 98
- Joined: Fri Aug 30, 2019 12:15 am
-
Ayushi2011
- Posts: 101
- Joined: Wed Feb 27, 2019 12:17 am
-
Charlene Datu 2E
- Posts: 62
- Joined: Wed Sep 11, 2019 12:16 am
-
BAlvarado_1L
- Posts: 49
- Joined: Thu Jul 25, 2019 12:16 am
-
SVajragiri_1C
- Posts: 115
- Joined: Thu Jul 11, 2019 12:16 am
-
EllieSchmidtke_4I
- Posts: 51
- Joined: Wed Feb 20, 2019 12:16 am
-
Bella Townsend
- Posts: 50
- Joined: Wed Feb 20, 2019 12:18 am
Re: Kelvin or Celsius?
Typically, you only ever use celsius is there is a change in temperature (delta T) because then the units don't matter. The difference will be the same.
-
Susan Chamling 1F
- Posts: 101
- Joined: Wed Sep 30, 2020 9:42 pm
-
Ariel Guan 1H
- Posts: 107
- Joined: Wed Sep 30, 2020 9:36 pm
- Been upvoted: 1 time
-
Will Skinner
- Posts: 94
- Joined: Wed Sep 30, 2020 9:28 pm
-
Rajshree 1F
- Posts: 108
- Joined: Wed Sep 30, 2020 9:32 pm
-
Emma Strassner 1J
- Posts: 102
- Joined: Wed Sep 30, 2020 9:50 pm
Re: Kelvin or Celsius?
Hi, you would use Kelvin and make sure to convert from other units like Celsius.
-
Mursall M 2A
- Posts: 60
- Joined: Wed Nov 18, 2020 12:20 am
Re: Kelvin or Celsius?
You should be using Kelvin so it can cancel with the units for the R constant.
-
Keeryth Sandhu 1H
- Posts: 101
- Joined: Wed Sep 30, 2020 10:04 pm
-
Gwirnowski 3B
- Posts: 58
- Joined: Wed Nov 18, 2020 12:29 am
Re: Kelvin or Celsius?
Kelvin is always used with the ideal gas law as the units need to cancel out in the end.
-
Michelle Nguyen 3F
- Posts: 104
- Joined: Wed Sep 30, 2020 10:03 pm
-
Katelyn_Ortega_1D
- Posts: 102
- Joined: Wed Sep 30, 2020 10:00 pm
-
Taylor Newville 1C
- Posts: 51
- Joined: Wed Nov 18, 2020 12:21 am
Re: Kelvin or Celsius?
You would use Kelvin. If you are ever unsure about what units to use, look at the units of the equation's constant to see what needs to be canceled out. For the Ideal Gas Law, the gas constant R= 8.314 J/mol·K, so the temperature needs to be in Kelvins too.
-
Crystal Pan 2G
- Posts: 118
- Joined: Wed Sep 30, 2020 10:04 pm
- Been upvoted: 2 times
-
Darlene Lien 3E
- Posts: 132
- Joined: Wed Sep 30, 2020 9:37 pm
Re: Kelvin or Celsius?
You would use Kelvin. If you look at the constants and equations sheet, the units for the rate are in Kelvin. Hope this helps!
-
Joshua Chung 2D
- Posts: 108
- Joined: Wed Sep 30, 2020 9:32 pm
-
Lauren Strickland 1B
- Posts: 97
- Joined: Wed Sep 30, 2020 9:47 pm
-
Rayna Irving 2C
- Posts: 103
- Joined: Wed Sep 30, 2020 9:33 pm
Re: Kelvin or Celsius?
Kelvin because in this case the rate, R, is a value that corresponds with the unit Kelvin.
-
Jack Kettering 3D
- Posts: 101
- Joined: Wed Sep 30, 2020 9:42 pm
-
Bai Rong Lin 2K
- Posts: 101
- Joined: Wed Sep 30, 2020 9:54 pm
-
Jiapeng Han 1C
- Posts: 101
- Joined: Wed Sep 30, 2020 9:50 pm
-
Brandon Le 3C
- Posts: 99
- Joined: Wed Sep 30, 2020 9:49 pm
Re: Kelvin or Celsius?
Using Kelvin is more beneficial because it is a more accurate measurement of the movement of molecules that determine the temperature of a system.
-
LeanneBagood_2F
- Posts: 101
- Joined: Wed Sep 30, 2020 9:32 pm
Re: Kelvin or Celsius?
as everyone has already said, temperature is always in Kelvin. this is not limited to just the PV = nRT equation as we will see throughout the remaining weeks
-
Sejal Parsi 3K
- Posts: 101
- Joined: Wed Sep 30, 2020 10:03 pm
-
Joshua Swift
- Posts: 100
- Joined: Wed Sep 30, 2020 9:50 pm
-
Violet Kwan 3H
- Posts: 132
- Joined: Wed Sep 30, 2020 9:55 pm
- Been upvoted: 1 time
Re: Kelvin or Celsius?
Kelvin is the SI unit for temperature, so I believe you would use that. Also, the conversion is just adding 273.15 to Celsius, so better just to be safe and convert to Kelvin.
-
Ethan Goode 2H
- Posts: 136
- Joined: Wed Sep 30, 2020 9:51 pm
-
Edison Tham 3D
- Posts: 50
- Joined: Mon Jun 17, 2019 7:25 am
- Been upvoted: 1 time
Re: Kelvin or Celsius?
Typically, since the gas constant R is given in Kelvin, we would use temperature in Kelvin when looking at the equation PV=nRT.
-
Will Skinner
- Posts: 94
- Joined: Wed Sep 30, 2020 9:28 pm
-
Chinyere Okeke 2J
- Posts: 102
- Joined: Wed Sep 30, 2020 10:05 pm
- Been upvoted: 1 time
Re: Kelvin or Celsius?
In this class and most Thermodynamic classes in the US, we are doing our calculations in Kelvin, this is because a lot of values/constants have Kelvin in their units not Celsius. The only time you wouldn't need to change the degrees is when you are doing change in temperature since change in 1 K = change in 1 degree-Celsius.
-
Claudia_Danysh_2B
- Posts: 99
- Joined: Wed Sep 30, 2020 9:58 pm
-
Rose_Malki_3G
- Posts: 143
- Joined: Wed Sep 30, 2020 10:02 pm
-
Charmaine Ng 2D
- Posts: 108
- Joined: Wed Sep 30, 2020 9:37 pm
-
Jordan Tatang 3L
- Posts: 125
- Joined: Wed Sep 30, 2020 9:31 pm
- Been upvoted: 3 times
-
jasmineculilap_3F
- Posts: 101
- Joined: Wed Sep 30, 2020 9:40 pm
-
Ximeng Guo 2K
- Posts: 114
- Joined: Wed Sep 30, 2020 9:54 pm
Return to “Thermodynamic Systems (Open, Closed, Isolated)”
Who is online
Users browsing this forum: No registered users and 5 guests

