Order
Moderators: Chem_Mod, Chem_Admin
-
Rhea Shah 2F
- Posts: 97
- Joined: Thu Jul 25, 2019 12:17 am
-
Juliana Chopelas 1A
- Posts: 50
- Joined: Sat Aug 24, 2019 12:16 am
Re: Order
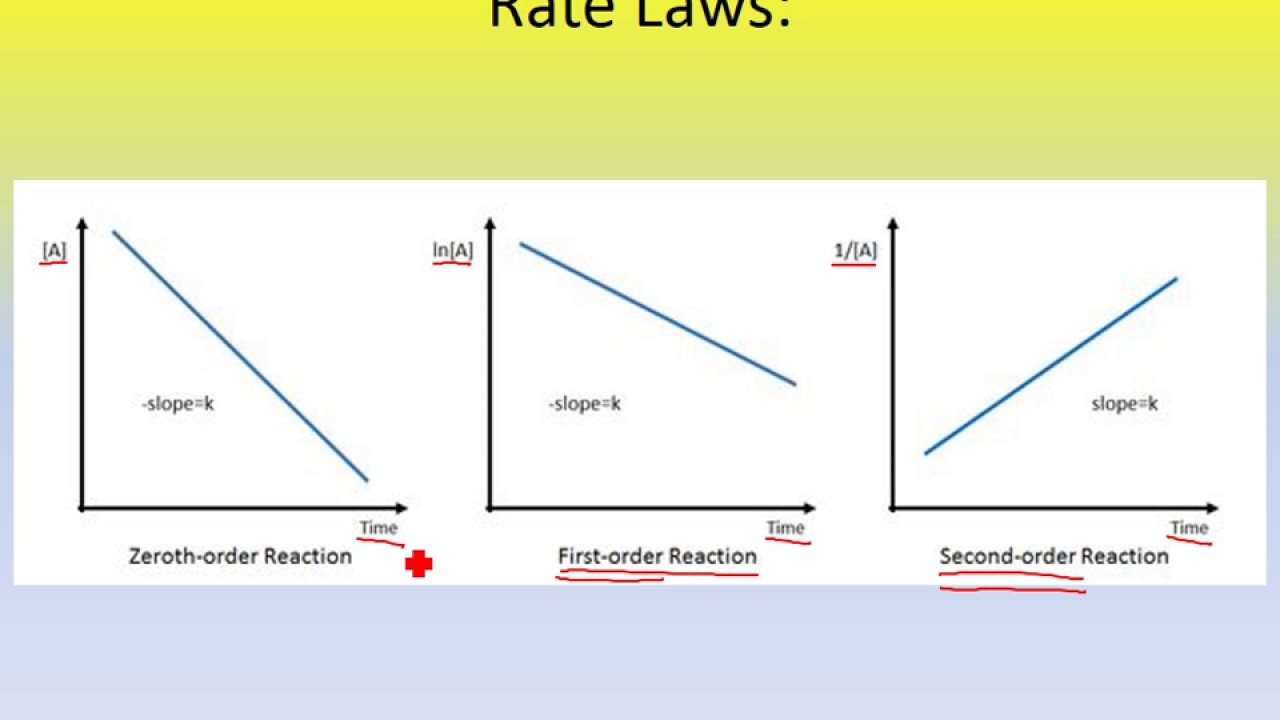
You can look at the reactions graph. Which ever graph produces a straight line will give you the reaction order. For zero order the graph would be (A) v time and would give a straight line with a -slope. First order reaction would be ln(A) v time and would give a straight line with a -slope. Second order would be 1/(A) v time and would give a straight line with a +slope.
-
205154661_Dis2J
- Posts: 109
- Joined: Wed Sep 18, 2019 12:21 am
Re: Order
You can look at the graphs provided or you can look at the units used in a specific problem if they do not specifically tell you which order it is. That is why it is important to know and memorize the units for the orders!
-
Eva Zhao 4I
- Posts: 101
- Joined: Sun Sep 29, 2019 12:16 am
Re: Order
Juliana Chopelas 1A wrote:You can look at the reactions graph. Which ever graph produces a straight line will give you the reaction order. For zero order the graph would be (A) v time and would give a straight line with a -slope. First order reaction would be ln(A) v time and would give a straight line with a -slope. Second order would be 1/(A) v time and would give a straight line with a +slope.
are these graphs always labeled to clearly see that? because the line for zero and first both have -slope
-
Jack Riley 4f
- Posts: 100
- Joined: Sat Aug 24, 2019 12:17 am
Re: Order
you look at the graph and compare it with the 3 possible orders to determine which one best fits the graph shown
Who is online
Users browsing this forum: No registered users and 4 guests