15.23
Moderators: Chem_Mod, Chem_Admin
-
donnanguyen1d
- Posts: 64
- Joined: Fri Sep 29, 2017 7:04 am
15.23
For Part C, why did you have to subtract (2(.034molB)) from (.153 molA) to get [A]t, I thought calculating [A]t would only require multiplying .034molB by 2?
-
Deborah Cheng 1F
- Posts: 50
- Joined: Thu Jul 13, 2017 3:00 am
Re: 15.23
I believe it's because the equation of the reaction given is 2A 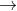 B + C so we need to account for the ratio of 2 mol A used to 1 mol B produced.
B + C so we need to account for the ratio of 2 mol A used to 1 mol B produced.
-
ConnorThomas2E
- Posts: 57
- Joined: Fri Sep 29, 2017 7:04 am
Re: 15.23
You start with the initial concentration of A, and you need to find how much b is produced to see how much the concentration of A decreases. Therefore, you would subtract 2*b from the initial concentration of A
-
Kyra LeRoy 1E
- Posts: 52
- Joined: Sat Jul 22, 2017 3:00 am
Re: 15.23
For product to form, there must be something taken from the reactant. Therefore, you take the initial concentration of A and subtract the concentration that has been used to create B at time t.
Return to “First Order Reactions”
Who is online
Users browsing this forum: No registered users and 1 guest

