Wave and Particle Properties of Electrons and Photons
Moderators: Chem_Mod, Chem_Admin
-
RichardValdez1L
- Posts: 59
- Joined: Fri Apr 06, 2018 11:05 am
Wave and Particle Properties of Electrons and Photons
Are we just supposed to understand that electrons and photons act sometimes as waves and sometimes as particles because when using two beams the particles do not collide and the interference patterns for waves? Any clarification would be much appreciated ! :)
-
Chem_Mod
- Posts: 23858
- Joined: Thu Aug 04, 2011 1:53 pm
- Has upvoted: 1253 times
Re: Wave and Particle Properties of Electrons and Photons
Hello Richard,
For the purposes of this class, we are to understand that massless photons as well as matter with mass (electrons, protons, a baseball) all have wave-particle duality to some degree.
For a photon, its wavelength is defined via
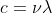
And for particle with mass (eg. electron), its wavelength is defined via De Broglie,
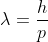
Hope this helps!
For the purposes of this class, we are to understand that massless photons as well as matter with mass (electrons, protons, a baseball) all have wave-particle duality to some degree.
For a photon, its wavelength is defined via
And for particle with mass (eg. electron), its wavelength is defined via De Broglie,
Hope this helps!
-
RichardValdez1L
- Posts: 59
- Joined: Fri Apr 06, 2018 11:05 am
-
vivianndo_1L
- Posts: 30
- Joined: Fri Apr 06, 2018 11:02 am
Re: Wave and Particle Properties of Electrons and Photons
How does constructive/destructive interference relate to the wave-particle duality then? I'm still a little confused
-
Chem_Mod
- Posts: 23858
- Joined: Thu Aug 04, 2011 1:53 pm
- Has upvoted: 1253 times
Re: Wave and Particle Properties of Electrons and Photons
The constructive/destructive interference patterns show that they have wave-like properties because you can see diffraction.
For an understanding of diffraction, see this post:
viewtopic.php?f=27&t=30885&p=97373&hilit=diffraction&sid=cb3a739b3b5986fdd7a628d9ad2d6fcc#p97373
For an understanding of diffraction, see this post:
viewtopic.php?f=27&t=30885&p=97373&hilit=diffraction&sid=cb3a739b3b5986fdd7a628d9ad2d6fcc#p97373
Return to “Bohr Frequency Condition, H-Atom , Atomic Spectroscopy”
Who is online
Users browsing this forum: No registered users and 5 guests

